Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.

a heating element in miniature ovens for quartz crystals acting as constant-
frequency sources. It has also been made use of in hair-driers and space-heaters.
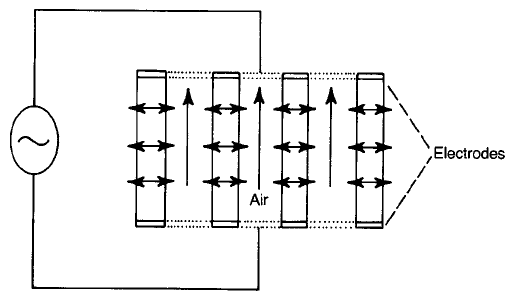
Air is blown through the perforations in a block of ceramic (Fig. 4.24) that is
designed to heat the emergent air to a suitable temperature. Even when the fan is
switched off the heater temperature only rises by about 20 8C so that no damage
results.
PTC elements make useful temperature indicators because the sharp rise in
resistivity above the Curie point is very easily detected or used to operate a
control mechanism. Small tubular elements a millimetre in diameter and a few
millimetres long can be inserted in motor or transformer windings and used to
detect overheating or to control it directly by increasing the overall resistance of
the winding when it becomes too hot.
The non-equilibrium properties of PTC elements can also be utilized. If the
room temperature resistance is low there will be a high current surge when a
voltage is applied and this will fall to 1% or less of its initial value when the
element heats up above its Curie point. Such devices are used in demagnetizing
(‘degaussing’) coils in T.V. monitors and in place of capacitors in motor starters.
Just as in the case of NTC thermistors (see Section 4.4.1), the temperature
distribution within a PTC thermistor can be far from uniform in certain non-
equilibrium applications. For example, thin discs in motor-starter units have
172 CERAMIC CONDUCTORS
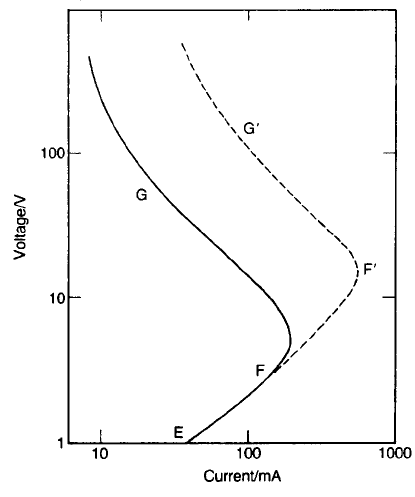
Fig. 4.23 Current–voltage characteristic for a PTC thermistor in thermal equilibrium.
TEAMFLY
Team-Fly
®
been known to split along a plane parallel to their electrodes. Highly
homogeneous flaw-free ceramics are needed to survive under such severe
conditions.
It can be seen that PTC elements give rise to a greater diversity of applications
than NTC units, but their production in high yield to meet close specifications
demands great care and attention.
4.5 Fuel Cells and Batteries
4.5.1 The stimulus for developing fuel cells and batteries
Increasing awareness of environmental factors and limited energy resources have
led to a profound evolution in the way we view the generation and supply of
energy. Although fossil and nuclear sources will remain the most important
energy providers for many more years, flexible technological solutions which
involve alternative means of energy supply and storage need to be developed
urgently.
The search for cleaner, cheaper, smaller and more efficient energy technologies
has been driven by recent developments in materials science and engineering. The
aim of this collection of reviews is therefore to focus on what materials-based
solutions can offer and to show how the rational design and improvement of
chemical and physical properties of these materials can lead to energy alternatives
that can compete with existing technologies [3].
The treatment in the present overview places emphasis on the roles ceramics and
their processing are playing in two of the various alternative energy technologies.
There is an extensive literature dealing with fuel cell and battery technologies
and the interested reader has much choice. The monograph by N.Q. Minh and T.
Takahashi [4] deals in depth with the science and technology of ceramic fuel cells,
especially with the electroceramics aspects. The comprehensive texts by K.
FUEL CELLS AND BATTERIES 173
Fig. 4.24 Section through a PTC ceramic hair-drier heating element: horizontal arrows
indicate heat flow out of the ceramic.
Kordesch and G. Simander [5] and J. Larminie and A. Dicks [6] deal more
generally with fuel cell technologies and applications and that by D.A.J. Rand
et al. [7] with batteries for electric vehicles. The web offers much information;
other recommended publications are identified during the course of the
discussion.
The imperative to lower ‘greenhouse’ gas emissions and to achieve cleaner city
and, indeed, global environments has stimulated the intensive research and
development efforts directed to reducing reliance on the internal combustion
engine for automotive power, and fossil fuel-powered electrical power
generators. The effort is not only driven by the need to reduce the harmful
effects of burning fossil fuels, but also by the knowledge that supply of these fuels
is limited.
An important stimulus to battery development was the 1989 Los Angeles
legislation which has as its objective an increasing proportion of new cars sold
in California to be ‘Zero Emission Vehicles’ which means, in effect, that cars
must be powered by hydrogen fuel cells or batteries. The legislation requires
that at least 10% of all new vehicles marketed in California must be ZEVs by
2003. The California Air Resources Board (CARB) initiative was followed by
similar legislation in New York and Massachusetts. Many hundreds of
thousands of electric vehicles are expected to be on the road in California by
2010.
Students are reminded of the upper thermodynamic limit set on the
efficiency of a heat engine, for example the internal combustion and gas-
turbine engines. The ideal and totally unrealistic engine would operate on the
so-called Carnot cycle where the ‘working substance’ (e.g. the gas) is taken in
at the high temperature (T
h
) and pressure and after doing external work is
exhausted at the lower temperature (T
c
) and lower pressure. The Carnot
efficiency, Z, is given by
Z ¼ 1 T
c
=T
h
(4:29Þ
As an example, if the hot temperature is 1273 K (1000 8C) and the cold
temperature 373 K (100 8C) then the efficiency is approximately 70%. In
practice the operation of a real engine does not follow the Carnot cycle and
the efficiency is considerably lower. For a medium sized motor car with an
internal combustion engine the fuel efficiency is about 12%, much of the
wasted 88% demanding water cooling. There are continuous improvements
made in petrol and diesel engine technologies and in the fuels and
projections suggest that thermal efficiencies a little over 50% will eventually
be achieved.
For a modern gas-fired power station in the UK the electrical efficiency is
about 45% and for a coal-fired station about 37%. There are combined cycle
plants in which the waste heat from the gas turbines is used to produce steam
that in turn drives a steam turbine generator. Such combinations, which are
174 CERAMIC CONDUCTORS
expensive, can deliver overall (electricalþuseful heat) efficiencies of up to 55%.
Another approach to increasing overall thermal efficiencies is via combined heat
and power installations where the waste heat is used typically for district heating
or for an industrial process. For such CHP plants efficiencies of 80% are
achieved.
It is important to recognize that all the potential improvements to increase the
thermal efficiencies of heat engines do nothing to reduce emission of the
‘greenhouse’ gases CO
2
and NO
x
. The achievement of this goal probably lies
with the fuel cell and rechargeable battery. But then there is a range of competing
fuel cell and battery types and choices will depend on many complex factors, not
least the impact new technologies will have on global economies, for instance the
oil industries, the infrastructures geared to the present day automotive internal
combustion engine and, not least, political considerations.
The subject is too complex to attempt an intelligent summary here; over the
next decade or two the scenario will unfold with the various technologies most
probably first finding their own particular niche markets and eventually
dominating the scene.
Although the research and development effort is focused on a range of fuel
cell and electrical storage battery types the discussion here is confined to
those whose function depends on conducting ceramics. To appreciate the
technological challenges involved in developing them to a commercially
viable reality it is necessary to have an understanding of the relevant basic
science.
Electrochemical cells are devices that convert chemical energy directly into
electrical energy thus circumventing the fundamental efficiency limit set by the
Carnot cycle. This is the case whether the device is the familiar battery or the less
familiar fuel cell.
A fuel cell is a device in which the reactants, for example hydrogen and
oxygen, are each continuously supplied to opposite sides of a suitable membrane
and converted to electrical energy.
A ‘secondary’ electrochemical cell is simply one that can be recharged as in the
case of the Na/S cell discussed below (in contrast a ‘primary’ cell, such as the
common torch battery, is exhausted after use and cannot be recharged). During
charging the chemical reaction is driven in reverse by applying an e.m.f. in the
sense to oppose the forward direction e.m.f.
The voltages developed by individual electrochemical cells are small, typically
in the range 1–2 V and for them to constitute useful power sources they must be
connected in series so that the voltage is multiplied by the number of cells, and in
parallel to increase current availability.
The fuel cells and batteries we are concerned with here (and the sensors to be
discussed later) rely on electroceramics having high electrical conductivities and,
ideally, charge-carriers that are either ions or electrons but not a mix of the two.
They are discussed later.
FUEL CELLS AND BATTERIES 175
4.5.2 Basics of fuel cells and batteries
Fuel cells
Overview The English chemist Humphrey Davy wrote in 1812 ‘If a piece of
zinc and a piece of copper be brought in contact with each other, they will form a
weak electrical combination, of which the zinc will be positive, and the copper
negative . . .’ so initiating the history of the electrochemical cell. But it was
Michael Faraday who, in 1834, laid the foundations of quantitative electro-
chemistry by relating the quantity of a substance electrolysed to the amount of
electrical charge involved.
It is from these foundations that ‘electrochemistry’ has evolved and which now
provides the scientific basis to the technology of electrochemical cells.
Fuel cells are not new, the concept dating back to 1839 and the observations of
a Welsh physicist, William Grove. One of the first practical cells was developed
at Cambridge University in the UK by an engineer, ‘Tom’ Bacon, a descendant
of the 17th century philosopher, Francis Bacon. Bacon constructed a stack of 40
alkaline fuel cells (AFCs) which could develop 5 kW of electrical power. Interest
continued at a low level and in 1958 General Electric developed a polymer
electrolyte membrane (PEM) cell which was used in the Gemini space
programme; in the 1960s Pratt and Whitney developed Bacon’s AFC technology
which was used in the Apollo space programme. During the 1990s interest in fuel
cell technology grew at a rapid pace, particularly in the automotive industry, and
the growth in interest is unabated.
There are many obstacles to the replacement of the internal combustion engine
for automotive power by fuel cell technology, a major one being the existing
infrastructure designed to service the petrol- and diesel-driven vehicles. As far as
reducing greenhouse gases is concerned the favoured fuel is hydrogen since water
is the only reaction product. However, because of the existing infrastructure (e.g.
petrol service stations and garages) there would be short-term economic
advantages to be gained through using cells fuelled by methanol or petrol;
unfortunately one of the reaction products is the greenhouse gas CO
2
, although
the higher efficiency of the fuel cell would mean a pro rata reduction in these.
The almost limitless abundance of hydrogen on the planet in the form of
water has stimulated intensive research effort, especially in Japan and the US,
directed towards developing the so-called ‘hydrogen economy’. This demands
economically viable routes to producing adequate supplies of hydrogen, for
example via the electrolysis of water. There are various ‘renewable’ ways of
powering the electrolysis of water to obtain hydrogen and oxygen, including for
example, wind power, tidal and wave power. There is also photoelectrolysis which
harnesses the sun’s energy via the photovoltaic effect to electrolyse water directly.
The challenges are real but the potential benefits immeasurable. To achieve the
‘hydrogen economy’ would be an unquestionable ‘giant step for mankind’.
176 CERAMIC CONDUCTORS
The situation is an extremely complex one and future evolution of the large-
scale exploitation of fuel cell technology impossible to predict. But there are
significant developments. For example, DaimlerChrysler announced in 2000 its
plans to build a fleet of hydrogen-fuelled buses, the hydrogen being stored in
tanks under pressure. This is an initial but very significant step. There are
alternative and far more convenient ways of storing hydrogen ‘on board’, for
example the metal hydrides can store hydrogen within their crystal structure and
release it on heating [3].
One of the important operational considerations is the range the car can travel
on a single fuelling (or, in the case of the battery, on a single charging) and the
power availability. It is clear that if fuel cell and battery technologies are to be
acceptable to the motorist then they must compete favourably with the internal
combustion engine in these respects. The vehicle has, of course, to transport its
own ‘engine’ and fuel as well as passengers. The comparisons are made with the
help of the power-to-weight and energy-to-weight ratios, the former determining
the acceleration and hill-climbing capabilities of the vehicle and the latter its
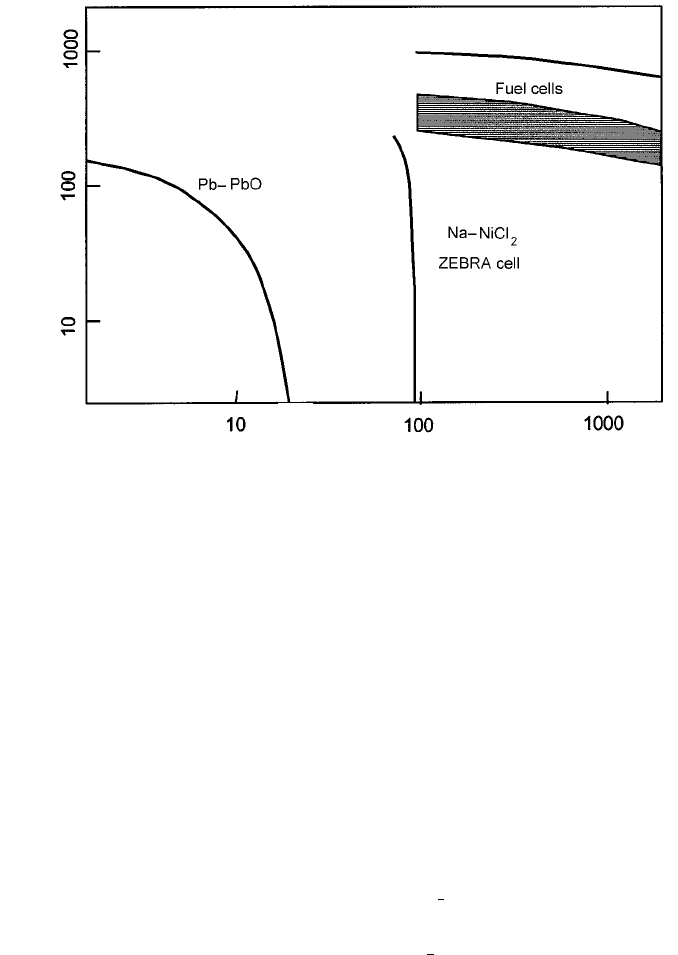
range. On these bases Fig. 4.25 shows fuel cells’ characteristics to be very
competitive even though they are unlikely to match internal combustion engines
when it comes to ‘high performance’ characteristics.
The various types of fuel cell currently under development and described by
Larminie and Dicks [6] are listed below.
1. The polymer electrolyte membrane or proton exchange membrane (PEM) cell.
2. The alkaline electrolyte cell (AFC).
3. The phosphoric acid cell (PAFC).
4. The molten carbonate cell (MCFC).
5. The solid oxide ceramic cell (SOFC).
The SOFC will be the major concern here although it will be helpful to consider
first the elements of the PEM cell since not only is it a very strong contender for
large-scale use, but its basic science is simple.
Elementary fuel cell science The essential of a fuel cell is the electrolyte, a
material which conducts electricity by the transport of an ionic species. On one
side of the electrolyte membrane there is a source of the ion species at a
particular chemical potential and on the other side a ‘sink’ for the ions at a
relatively lower chemical potential.
The PEM cell is the cleanest fuel cell since the fuel is hydrogen, the oxidant
oxygen and the product water. Although it clearly falls outside the scope of a text
focused on electroceramics there are good reasons for prefacing the present
discussion with a brief outline of those elements of the science and technology
basic to it and common to the ceramics-based fuel cells. Also, for an intelligent
FUEL CELLS AND BATTERIES 177
appreciation of the status of ceramics-based cells, the ‘solid oxide fuel cells’ (or
SOFCs), they must be viewed in the context of fuel cell technology as a whole
which includes the PEM cell as an important part.
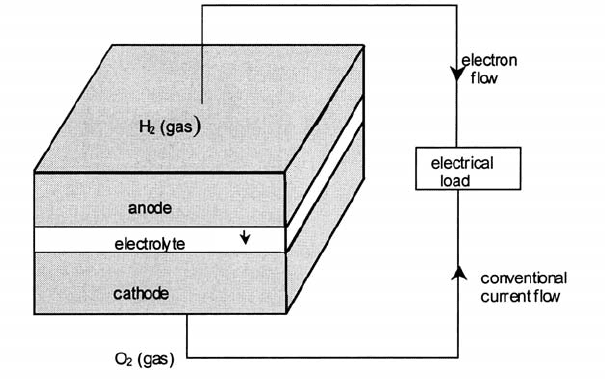
The elements of the PEM cell are shown in Fig. 4.26 together with the
reactions occurring at the anode and cathode.
The student will recall that the anode is the electrode to which the anions (the
negatively charged species) in the electrolyte migrate and the cathode the
electrode to which the cations (the positively charged species) migrate.
Anode oxidation reaction: H
2
! 2H
.
þ 2e
0
(4:30Þ
Cathode reduction reaction: 2H
.
þ
1
2
O
2
þ 2e
0
! H
2
O(4:31Þ
Overall cell reaction: H
2
þ
1
2
O
2
! H
2
O(4:32Þ
The theoretical cell electromotive force, the e.m.f, can be calculated on the
basis that the free energy released due to the chemical reaction is available to do
external work, in this case electrical work. The change in the Gibbs free energy
DG represents the total electrical energy available. From the definition of the
volt, the electrical energy involved in transferring Q coulombs through E volts is
178 CERAMIC CONDUCTORS
Fig. 4.25 Ragone plot comparing the internal combustion engine with the ZEBRA battery
and fuel cells (very approximate). [The plot was introduced for comparing batteries
(standardized to a weight of 300 kg); to include the performances of engines and fuel cells
in a meaningful way their masses, together with the fuel carried, should be standardized to
300 kg.].
Internal combustion engine
cell
Specific power/W kg
1
Specific energy/W h kg
1
QE joules. If the transferred charge is carried by 1 mole of electrons, then Q ¼F ,
F being the Faraday constant (approx. 9.648610
4
C mol
71
). If each chemical
species carries a charge of |z|e, z being the oxidation number (plus or minus
depending on whether the species carries a positive or negative charge), then the
electrical work done in transferring 1 mole of the species is |z|FE. Equating the
chemical energy made available to the electrical work done leads to the equation
DG ¼jzjFE (4:33Þ
From tables the free energy change for reaction (4.32) taking place at 298 K
(25 8C) is 237 kJ so that from Eq. (4.33), with |z| ¼2, E ¼ 1.23 V.
In practice the PEM cell is likely to operate at approximately 80 8C (353 K)
when the theoretical e.m.f. is 1.18 V.
It is important to appreciate that the e.m.f values relate to open circuit
conditions, that is when the cell is delivering no current. When current is drawn
from the cell power is dissipated internally (internal resistive effects), resulting in
a terminal p.d. of approximately 0.8 V; this is the effective voltage for doing work
on an external electrical load.
A further factor that determines deliverable power (VI ) is the current (I) that
can be drawn from the cell. Current will be limited by the following factors:
1. the rate of the chemical reaction at either anode or cathode, the slowest being
rate-determining, and
FUEL CELLS AND BATTERIES 179
Fig. 4.26 The elements of the hydrogen fuel cell. Note: (i) The student is reminded that
a chemical species which loses electrons is oxidized; one that gains electrons, reduced.
(ii) The proton H_ is transported through the polymer electrolyte attached to water molecules,
(H
2
O)
n
H_ which causes a ‘water management’ problem. Current research is aimed at
developing proton conductors able to operate in the region of 200 8C when the proton
migrates unattached to water molecules.
H
.
2. the internal resistance of the cell determined in part by
(a) the intrinsic electrical properties of the cell components, especially the
electrolyte and
(b) electrolyte geometry.
[The student should recall G (conductance) ¼sA/l, (see Section 2.6.1)]
The optimised cell therefore comprises the following:
1. anode and cathode designed to maximize the rates of the oxidation and
reduction reactions, and to make good electronic contact with the external
circuit, and
2. an electrolyte having large area and small thickness consisting of a material
having a high ionic and zero electronic conductance; any electronic
contribution to conductance will tend to internally short-circuit the cell,
lowering the terminal p.d. and contributing to power wastage.
To maximize the rate of a reaction occurring at a gas/solid interface requires
the total reactive surface be maximized, and be catalytic. Therefore the anode
and cathode must have a high specific surface area (total internal area per unit
mass or volume) with the proviso that the flow of reactant molecules to the
reaction sites must not be impeded. As stated above, both anode and cathode
should be good electronic conductors.
It is inappropriate to pursue here optimization of the electrolyte design for a
PEM cell. The essentials of the cell are a thin polymer membrane coated on each
surface with carbon mixed with platinum particles acting as the catalyst.
Having optimized the design of a single cell there arises the challenge of
interconnecting many such cells in series to form a stack to increase total voltage
(or in parallel to increase current flow and to remove the risk of total failure
should a single cell fail). This challenge is met by introducing a bipolar plate (also
termed an ‘interconnect’).
The interconnect normally links the anode of one cell to the cathode of the
next. It must, of course, be an electronic conductor and also a gas barrier
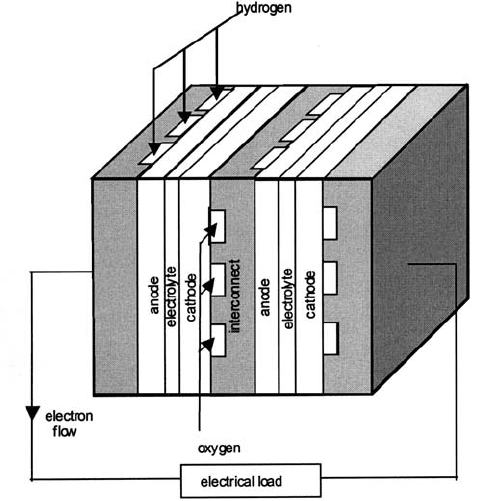
preventing the direct meeting of fuel and oxidant gases. Fig. 4.27 illustrates how
the interconnection is achieved in the case of the so-called ‘planar’ fuel cell stack.
In the later discussion of the ceramics-based cells a tubular configuration is
described, but the principles are the same.
In the case of the high temperature SOFC discussed below the principles
outlined above equally apply. The technical differences are that the cell runs
typically on hydrocarbon fuels (e.g. ‘natural’ or coal-gas) and that the electrolyte
is an oxygen ion conductor rather than a proton conductor. The complex fuel
molecules, in the presence of the water molecule and at the high operating
180 CERAMIC CONDUCTORS
temperatures (600–1000 8C), are ‘reformed’ into H
2
and CO. Oxygen supplied at
the cathode reacts with electrons flowing in the external circuit to form oxygen
ions (O
2
) which diffuse through the electrolyte to the anode where they react
with the fuel to form H
2
O and CO
2
. The open circuit voltage (e.m.f.) for a SOFC
depends, of course, on the nature of the fuel and temperature but is typically
approximately 1.1 V.
Storage batteries exploiting b’’-alumina
Overview The battery is simply an electrochemical cell just as the fuel cell. The
difference is that in the case of the fuel cell fuel is continuously supplied and
electrical power extracted, whereas in the case of the storage battery, as the name
implies, chemical energy is stored and electrical energy extracted on demand.
As for fuel cell technology there is an extensive ‘battery’ literature and various
competing types, including lead/acid, Ni/metal hydride, Na/S, Na/NiCl and Li-
ion, and each has advantages and disadvantages. The subject is dealt with
comprehensively in [7].
The familiar lead/acid battery dates back to 1860, and by the early part of the
20th century there were many electric vehicles in operation throughout the
world, mainly as taxicabs, personal cars, delivery vans, trams and buses.
FUEL CELLS AND BATTERIES 181
Fig. 4.27 Illustrating the connecting of two fuel cells via bipolar plates (‘interconnects’).
