Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.

Single-crystal single-domain BaTiO
3
has a relative permittivity at 20 8C of 230
in the polar direction and 4770 in the perpendicular directions. The random
orientation of axes in a ceramic would lead, on the basis of Lichtenecker’s
relation (Eq. (2.131)), to a permittivity of 1740. In practice the low-field relative
permittivity of the ceramic form lies in the range 2000–4500 and varies with the
method of preparation. The higher values than expected are ascribed to small
oscillations of domain walls. A large fraction of the dissipation factor can also be
accounted for by domain wall motion. Thus an understanding of the domain
structure of ceramics greatly assists the control of dielectric properties.
The principal effects determining properties are now discussed in turn.
AO/BO
2
ratio
The AO/BO
2
ratio is the ratio of the total number of ions on Ba sites to the
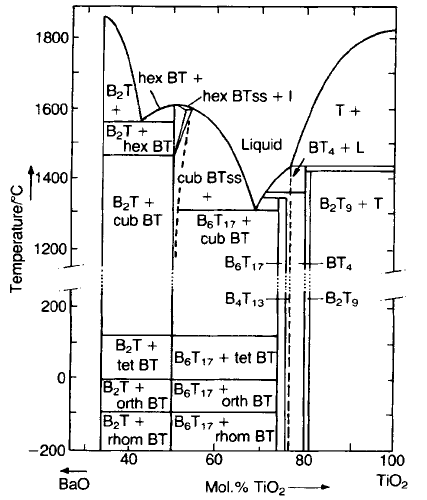
number on Ti sites. The partial phase diagram for the BaO–TiO
2
system
(Fig. 5.41) shows that there is only very slight solubility for excesses of either
BaO or TiO
2
in BaTiO
3
. Excess TiO
2
(AO/BO
2
51) results in the formation of a
separate phase of Ba
6
Ti
17
O
40
, and this forms a eutectic with BaTiO
3
that melts at
about 1320 8C so that liquid phase sintering can take place at temperatures below
1350 8C. A wide range of grain sizes (5–50 mm) results.
312 DIELECTRICS AND INSULATORS
Fig. 5.41 Phase diagram of the BaO–TiO
2
system (434 mol.% TiO
2
).
L
L
L
L
TEAMFLY
Team-Fly
®
An excess of BaO results in the formation of Ba
2
TiO
4
which forms a eutectic with
BaTiO
3
that melts at about 1563 8C. As is often the case with solid insoluble
phases, Ba
2
TiO
4
inhibits the grain growth of BaTiO
3
sintered at temperatures up
to 1450 8C, giving rise to grain sizes in the 1–5 mm range. Excess BaO also lowers
the cubic–hexagonal transition from 1570 8C to about 1470 8C in pure BaTiO
3
.
Hexagonal material seldom occurs in sintered ceramics of technical purity
because many common substituents, such as strontium for barium, stabilize the
cubic form.
The effect of the AO/BO
2
ratio varies with different substituents and additives
as discussed in the next section.
Substituents
Uniformly distributed isovalent substituents do not greatly affect the shape of the
e
r
–T curve and other characteristics. Their main effect is to alter the Curie point
and the lower transitions of BaTiO
3
.
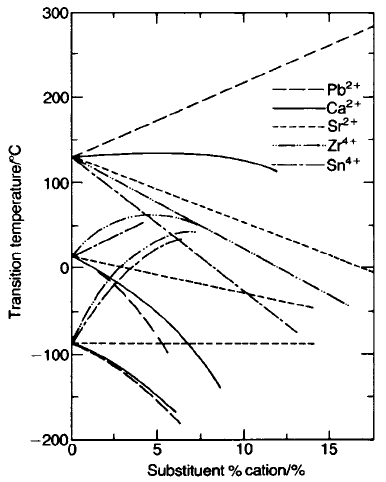
Barium can be replaced by isovalent ions with r
12
radii between 130 and
160 pm. As can be seen in Fig. 5.42 the effect on T
c
varies considerably among
lead, strontium and calcium which enables the transitions to be shifted to suit
HIGH-PERMITTIVITY CERAMICS 313
Fig. 5.42 Transition temperature versus concentration of isovalent substituents in BaTiO
3
:
---
,Pb
2+
;
-
,Ca
2+
; ----, Sr
2+
; --- ---, Zr
4+
; - - ---- - - , Sn
4+
. (After B. Jaffe et al. (1971)
Piezoelectric Ceramics, Academic Press, London.)
particular requirements. Ba, Pb and Sr ions can be mixed in any proportions to
produce a single-phase perovskite, while the solubility of CaTiO
3
is limited to
about 20 mol.%. Ti
4+
can be replaced by isovalent ions with r
6
radii between 60
and 75 pm. Zirconium, hafnium and tin have similar effects on the three
transitions, although the solubility of tin may be limited to about 10 mol.%.
They reduce T
c
but raise the temperature of the other two transitions to such an
extent that in the range 10–16 mol.% they almost coincide in the neighbourhood
of 50 8C. Particularly high values of permittivity are found for such composi-
tions. The more highly charged B-site ions diffuse less rapidly than the A-site ions
so that the effects of inhomogeneities are more often seen for B-site substituents.
Aliovalent ions (see Section 2.6.2) are usually limited in their solubility which
may depend on the AO/BO
2
ratio. K
+
can replace Ba
2+
to which it is very
similar in radius. The charge balance can be restored by the simultaneous
replacement of O
27
by F
7
. The effect on dielectric properties is minimal.
A number of trivalent ions with r
12
radii between 110 and 133 pm, e.g. Bi and
La, can substitute on the A site. La
3+
confers a low resistivity at low
concentrations (50.5 mo.%), and the electrical behaviour is discussed in Section
2.6.2, and Section 4.4.2. It has been studied more widely as a substituent for lead
in PbTiO
3
-based compositions.
Dysprosium with r
6
90 pm and r
12
115 pm is a rather large ion to be found
on a B site and rather small for an A site. At the 0.8 mol.% level it gives optimum
properties (e
r
at 25 8C, 3000; resistivity, 7610
10
O m). The resistivity falls to a low
value at higher AO/BO
2
ratios. The grain size after firing at 1450 8C in oxygen is
1 mm and few domain walls are visible. As a result e
r
is not greatly affected by
applied fields up to 2 MV m
71
. Unlike the slightly larger Sm
3+
ion, it has only a
small effect on the Curie point.
Higher-valency ions on B sites with r
6
radii between 58 and 70 pm have
similar effects to La on the A site at both high and low concentrations. Nb
5+
at the 5 mol.% level has been found to improve resistance to degradation. In
sufficient concentration these higher-charge substituents both suppress oxygen
vacancies and promote the formation of cation vacancies that act as acceptors.
The resulting dielectrics have a high resistivity and are resistant to
degradation.
Ti
4+
can be replaced by a number of trivalent ions with r
6
in the range 60–
70 pm (Cr, Ga, Mn, Fe and Co) up to about 2 mol.%. They lower T
c
and the
second transition to varying extents (23 K (mol.%)
71
for T
c
for Fe and 10 K
(mol.%)
71
for Ga). Their effects are complicated by the presence of oxidation
states other than 3+ (e.g. Co
2+
,Mn
4+
and Cr
4+
). They can also dissolve in the
intergranular phase so that their concentration in the BaTiO
3
phase is difficult to
determine. Their main effect is to form acceptors and so to compensate the
lowering of resistivity by donors. However, the charge balance may be
maintained by the formation of oxygen vacancies which lead to higher ageing
rates and to degradation under d.c. fields at lower temperatures.
314 DIELECTRICS AND INSULATORS
BaTiO
3
sintered with about 3 mol.% Fe
2
O
3
at 1300 8C gives a remarkably flat
e
r
/T relation with an average e
r
of about 2500. This may be because most of the
Fe is present in an intergranular phase that keeps the grain size down to about
1 mm and there is a non-uniform distribution of the Fe within the grains, thus
giving regions of differing Curie point that combine to give the flat e
r
–T
characteristic. The same composition sintered at 1360 8C gives a normal e
r
–T
peak at 60 8C, indicating that the Fe has diffused to a uniform concentration
within the grains following further grain growth.
About 0.5 mol.% MnO
2
is frequently added to all classes of dielectric and results
in a reduction in the dissipation factor. This may be due to its presence as Mn
4+
in
the sintered bodies with the possibility of trapping carriers by the reactions
Mn
4þ
þ e
0
! Mn
3þ
and Mn
3þ
þ e
0
! Mn
2þ
ð5:40Þ
Mn
3þ
Ti
and Mn
2þ
Ti
also act as acceptors.
Ti
4þ
can also be replaced by about 2 mol.% of divalent ions with r
6
in the
range 60–70 pm such as Ni
2þ
and Zn
2þ
, with similar results to substitution by
trivalent ions. Larger divalent ions such as Mn
2+
(r
6
¼82 pm) may be soluble to
a lesser extent. Mg
2+
(r
6
¼72 pm) is only soluble in BaTiO
3
when AO/BO
2
is
greater than unity; otherwise it forms a separate phase of MgTiO
3
. There is
evidence that Ca
2+
(r
6
¼100 pm) may occupy B sites to a limited extent when an
excess of Ba is present. All these ions can fulfil an acceptor function and, to
varying extents, can prevent BaTiO
3
-based compositions from becoming
conductive when fired in atmospheres low in oxygen (see Section 5.7.3).
Effect of crystal size
The grain size of a ferroelectric ceramic has a marked effect on the permittivity
for the size range 1–50 mm (see Fig. 2.48). Below about 1 mm the permittivity falls
with decreasing grain size. An important factor leading to this behaviour is the
variation in the stress to which a grain is subjected as it cools through the Curie
point.
As a single-crystal grain cools through the Curie point it attempts to expand in
the c direction and contract in the a directions, as can be seen in Fig. 2.40(b). It
will be constrained from doing so by the surrounding isotropic ceramic. The
resulting stresses within the grain can be reduced by formation of an appropriate
arrangement of 908 domains and, in large grains, most of the stresses can be
relieved by this mechanism. As the grain size decreases the domains become
smaller, with the domain width being roughly proportional to the square root of
the grain size. The number of domains per grain therefore decreases as the square
root of the grain size, and so the smaller the grain the larger is the unrelieved
stress. It can be shown using Devonshire’s phenomenological theory (cf. Section
2.7.1) that an increase in stress is accompanied by an increase in permittivity,
HIGH-PERMITTIVITY CERAMICS 315

irrespective of any possible contribution from the domain walls per se. As the
grain size approaches 0.5–0.1 mm, the unrelieved stresses reach values at which
they suppress the tetragonality and the permittivity falls to approximately 1000.
In addition to the direct effect of stress described above, a reduction in 908
domain width can enhance permittivity because the domain wall area per unit
volume of ceramic increases. The argument outlined below follows that
developed by Arlt et al. [19].
The decrease in mechanical strain energy resulting from the formation of
domains is counterbalanced by the increase in wall energy as the domains
develop. At equilibrium the total energy will be a minimum. The strain energy E
s
per unit of volume occupied by 908 domains has been calculated to be
E
s
¼
dYx
2
128pg
ð5:41Þ
where g is the average grain size, d is the domain width, Y is the Young modulus
and x ð¼ c=a 1) is the tetragonality. The domain wall energy E
d
per unit
volume is given by
E
d
¼
g
d
ð5:42Þ
where g is the surface energy associated with a 908 domain wall. Therefore the
total energy E
t
is
E
t
¼E
s
þE
d
ð5:43Þ
Substituting from Eqs (5.41) and (5.42) into Eq. (5.43), differentiating Eq.
(5.43) with respect to d and setting @E
t
=@d ¼ 0 yields the condition for minimum
total energy:
d ¼
128pgg
Yx
2
1=2
ð5:44Þ
Substituting the values for barium titanate (g 3mJm
2
, Y 1.7 10
11
Pa and
x 10
2
Þ gives
d 2 10
4
g
1=2
ð5:45Þ
Eq. (5.45) agrees quite well with experimental data for grain sizes between 1
and 10 mm. Above 10 mm more complex domain walls form and the domain
width is limited to about 0.8 mm. Below 1 mm the stresses are large enough to
reduce the tetragonality and this simple model is no longer valid.
It is easily shown from Eq. (5.45) that, while the domain wall area per grain is
proportional to g
5/2
, the domain wall area per unit volume of ceramic is
approximately 5000g
71/2
. The part of the permittivity due to domain wall motion
will be proportional to the domain wall area and so increases as the grain size
diminishes from 10 mmto1mm.
316 DIELECTRICS AND INSULATORS
Both the direct effect of stress and the changes in the concentration of domain
walls appear to contribute to the observed changes due to grain size. The
behaviour of domains is markedly affected by dopants and, in consequence, the
grain size at which walls become scarce has been reported as varying over a range
of values. The importance of grain size is in no doubt, but its optimum value for
each composition has to be determined empirically.
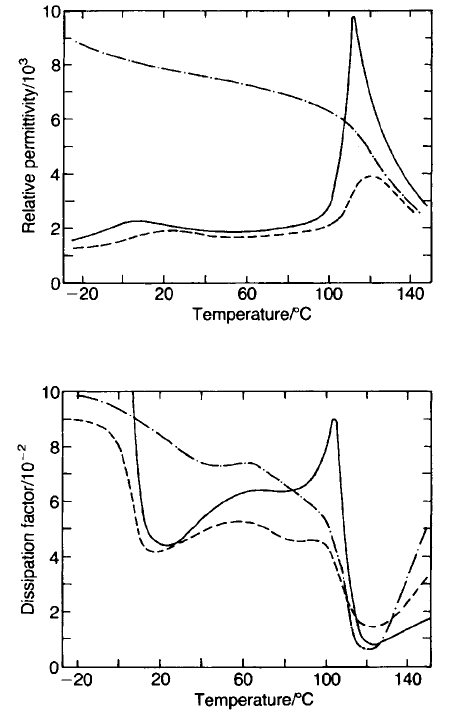
The effect on permittivity of applied electric ¢eld
The magnitude of the applied electric field has a very marked effect on dielectric
properties as can be seen in Fig. 5.43. The effects can be rationalized in very
HIGH-PERMITTIVITY CERAMICS 317
Fig. 5.43 Dielectric properties of technical grade BaTiO
3
ceramic under various field
conditions:
-
,15kVm
71
peak and 1 kHz;
---
,15kVm
71
peak and 1 kHz+
1.43 MV m
71
d.c.;
-
-
, 1.6 MV m
71
peak and 50 Hz.
general terms by considering the contributions to permittivity and energy
dissipation made by domain movement. Reference should also be made to
hysteresis effects discussed in Section 2.7.3.
Ageing of capacitors
The mechanism of ageing is discussed in Section 6.3.1, under piezoelectric
ceramics. In capacitors it is a minor nuisance and is usually in the 2–5% decade
range. However, on automated production lines a capacitance test may take
place not many minutes after silvering and soldering, and so may indicate a
considerably higher value than that measured a month or so later when the unit
is inserted in a circuit. However, once the ageing characteristic of a composition
has been determined (the logarithmic law, Eq. (2.124), is only approximate and
significant deviations may occur in the early stages), an allowance can be made to
relate the value on the production line to that found at a later date. One
advantage of automation is that the timing of operations can be very consistent.
Ageing is reversed to a large extent whenever a unit is heated above the Curie
point, although not completely unless heating is prolonged or the temperature is
increased above 500 8C. High a.c. or d.c. fields also reverse the ageing to a limited
extent, depending on the field strength and time of application. Unexpected
changes in capacitance may therefore take place under such conditions.
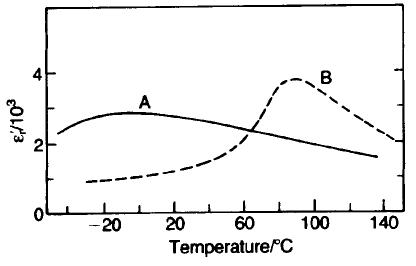
Heterogeneous dielectrics
A possible instance of heterogeneity occurs in iron-doped BaTiO
3
mentioned
above. It has often been noticed that the e
r
–T peaks obtained with mixed and
calcined constituent oxides and carbonates are sharper than those obtained with
318 DIELECTRICS AND INSULATORS
Fig. 5.44 The effect of compositional heterogeneity on dielectric properties: curve A, 43cat.%
Ba–7cat.%Bi–49cat.%Ti(BaTiO
3
and Bi
4
Ti
3
O
12
calcined separately); curve B, 46cat.%Ba–
5.5cat.%Bi–48cat.%Ti (starting materials calcined together). Both are sintered at 1300 8C for
1h.
mixed and sintered preformed compound oxides. Figure 5.44 shows the effects of
adding bismuth as Bi
4
Ti
3
O
12
to BaTiO
3
rather than as Bi
2
O
3
to a mixture of
BaCO
3
with TiO
2
. The flatter characteristics obtained with precalcined
compounds are most easily explained as due to the failure of the ions to
interdiffuse fully during the subsequent sintering to form a homogeneous
composition. The resulting inhomogeneity comprises regions with different Curie
points, and the net effect is the flattened characteristic.
In practice the use of this method of controlling e
r
–T relationships depends on
the interdiffusion of constituent ions being slow enough to be controlled by
practicable furnace schedules. For instance, (Ba
1–x
Sr
x
)TiO
3
compositions with
different x values, calcined separately and milled to an average particle size of
10 mm, yield a single peak corresponding to their average composition when
sintered to full density in air because of the relatively rapid interdiffusion of Ba
2+
and Sr
2+
ions. More highly charged ions such as Zr
4+
diffuse more slowly and
can give rise to heterogeneous compositions under sintering conditions that yield
satisfactory densities.
The effect of heterogeneity has been observed directly in the case of submicron
BaTiO
3
powder sintered with a 0.03 molar fraction of CdBi
2
Nb
2
O
9
in air at
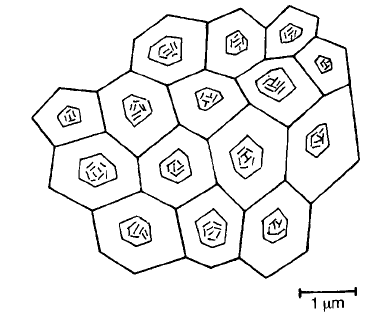
1130 8C for 4 h. A schematic diagram of the ceramic (Fig. 5.45) shows grains with
a duplex structure. The centre region of each grain exhibits a ferroelectric
domain structure which analysis shows to be low in substituent ions. The outer
region is high in substituents and appears to have a T
c
of 780 8C. The ceramic as
a whole has the relatively flat X7R characteristic. Many combinations of
additives and BaTiO
3
show similar effects, even when the constituents are mixed
as simple oxides and carbonates. This type of heterogeneity needs to be
distinguished from intergranular phases with widely differing compositions and
crystal structures from the main phase. In the present case there is a gradation
from a heavily doped to a lightly doped composition with a corresponding
HIGH-PERMITTIVITY CERAMICS 319
Fig. 5.45 Duplex microstructure of an X7R dielectric ceramic.
gradual change in lattice parameters and dielectric behaviour. It is possible that
the additives are initially uniformly distributed in the BaTiO
3
lattice but diffuse
out as a more perfect crystal is developed during grain growth. Much remains to
be understood about the processes involved.
5.7.2 Relaxor ferroelectrics
The dielectric properties of most compositions based on BaTiO
3
do not vary
greatly with frequency above about 500 Hz until the GHz range is reached.
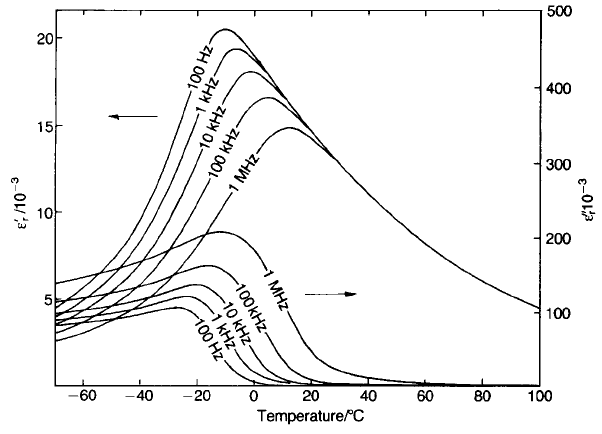
In contrast, in the case of the relaxor class of dielectric the real part of the
permittivity shows a broad peak as a function of temperature with a strong
frequency dispersion (see Fig. 5.46). The class comprises a number of perovskites
of the type Pb(B’ B’’)O
3
for example Pb(Mg
1/3
Nb
2/3
)O
3
(PMN), Pb(Zn
1/3
Nb
2/3
)O
3
(PZN) and Pb(Sc
1/2
Ta
1/2
)O
3
(PST), some solid solutions, for example
Pb
173x/2
La
x
(Zr
17y
Ti
y
)O
3
(PLZT) and some tungsten bronze structures (see
Section 6.4.4), for example Sr
57x
Ba
x
Nb
10
O
30
(SBN). The tpyical relaxor
characteristics shown for PMN in Fig. 5.46 are evidence of a gradual transition
from a macroscopic paraelectric to a ferroelectric phase below the peak
permittivity Curie temperature.
G.A. Smolenskii [20] was a pioneering researcher and at the time relaxors were
being intensively studied in Russia. Since then they have attracted very
considerable interest because of the following attributes. Firstly the low sintering
temperatures and high peak permittivity values which are attractive for the
320 DIELECTRICS AND INSULATORS
Fig. 5.46 Dielectric properties of Pb(Mg
1/2
Nb
1/2
)O
3
(PMN).
production of MLCCs of low cost and high volumetric efficiencies, and secondly
because of their high electrostriction coefficients which make them attractive for
actuators (see Section 6.4.3).
It is agreed that B-site crystalline disorder is resonsible for relaxor behaviour
although there are uncertainties regarding its precise nature, and it may be that
details differ from relaxor to relaxor. In the case of PMN one model [21]
proposes the existence of domains of nanometre order size within which the
composition is {Pb(Mg
1/2
Nb
1/2
)O
3
}
70.5e
, that is the Mg:Nb ratio is unity. The
domains are in a matrix of compositionally disordered material such that
the overall composition is stoichiometric, namely Mg/Nb ¼ 0.5. In this model the
negative charge the domains carry increases as the domain size increases; it is
the associated electrostatic energy which accounts for the observed stability of
the domain structure with high temperature annealing.
An alternative model [22] favours a uniform B-site disorder extending
throughout the volume and consisting of local clusters of ferroelectric and
antiferroelectric ordering with the highly polarizable Pb ion almost certainly
playing an important role.
Close parallels can be drawn between the dielectric response of a relaxor and
that of a random assembly of electric dipoles – a ‘dipolar glass’. The matter is
clearly one of very considerable complexity depending as it must upon the sizes,
charges and polarizabilities of the ions, and thermal history.
Closely related structural changes occur during the annealing of the
microwave dielectric BZT (see Section 5.6.5).
A number of low sintering compositions have been based on
PbFe
0.55
W
0.1
Nb
0.35
O
3
. A typical e
r
–T characteristic is shown in Fig. 5.47. e
r
is
greater than 10
4
between 78 and +45 8C but is greatly reduced when d.c. fields
are applied. Since in many applications the d.c. field is less than 0.2 MV m
71
its
effect is not of great importance.
One of the difficulties with most compositions containing lead and niobium is
a tendency to form pyrochlore-type rather than perovskite-type structures which
results in lower e
r
values. This is particularly the case when Zn ions are present.
Pyrochlore is a mineral with a composition approximating RNb
2
O
6
, where R is a
mixture of divalent ions. The pyrochlore-type phase found in lead magnesium
niobate has the composition Pb
1.83
Nb
1.71
Mg
0.29
O
6.39
. It has a room temperature
relative permittivity of 130 and is paraelectric. The structure contains corner-
sharing MO
6
octahedra but they have several different orientations. The effect on
the permittivity depends on the amount and distribution of the low-permittivity
phase. Small amounts occurring as discrete particles have rather less effect than a
corresponding volume of porosity, but they may considerably reduce the
permittivity if present as an intergranular phase interposing low-permittivity
regions between crystals of the high-permittivity phase. Larger amounts will also
result in a significant change in composition, and therefore in properties, of the
perovskite-type phase. It is found that the pyrochlore structure forms
HIGH-PERMITTIVITY CERAMICS 321
