Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.


(assuming constant E ), and
p
g
¼ p þ E
@e
@T
ð7:3Þ
where p ¼ @P
s
=@T is the true pyroelectric coefficient and p
g
is sometimes referred
to as a generalized pyroelectric coefficient. Since a temperature change DT
produces a change in the polarization vector, the pyroelectric coefficient has
three components defined by
DP
i
¼ p
i
DTi¼ 1, 2, 3 ð7:4Þ
Therefore the pyroelectric coefficient is a vector but, because in practical
applications the electrodes that collect the pyrocharges are positioned normal to
the polar axis, the quantities are usually treated as scalars, and this is done in the
following discussion.
The contribution Eð@e=@TÞ (Eq. (7.3)) can be made by all dielectrics, whether
polar or not, but since the temperature coefficients of permittivity of ferroelectric
materials are high, in their case the effect can be comparable in magnitude with
the true pyroelectric effect. This is also the case above the Curie point and where,
because of the absence of domains, the dielectric losses of ferroelectrics are
reduced, which is important in some applications. However, the provision of a
very stable biasing field is not always convenient.
Since pyroelectric materials are polar, they are also piezoelectric, and the strain
resulting from thermal expansion will result in the development of a surface
charge. However, this is a small effect that seldom exceeds 10% of the primary
pyroelectric effect.
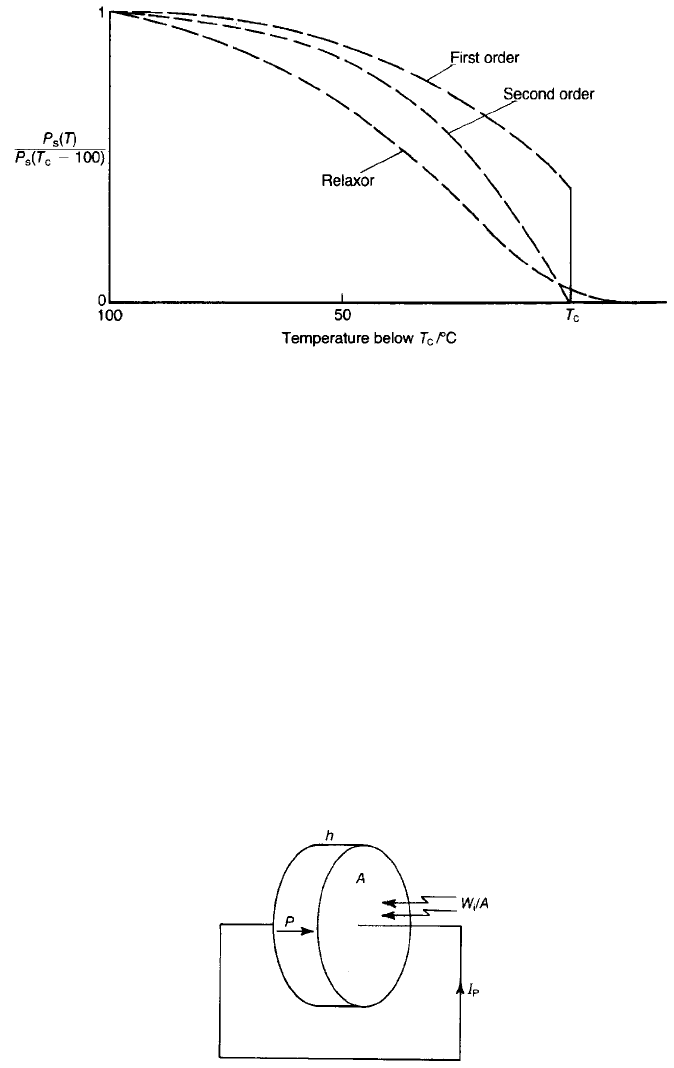
Because P
s
falls to zero at the Curie point, ferroelectric materials are likely to
exhibit high pyroelectric coefficients just below their transition temperatures. The
various ways in which P
s
falls as the Curie point is approached from below are
shown in Fig. 7.1. High pyroelectric coefficients are observed for ferroelectrics
that exhibit second-order transitions, such as triglycine sulphate with a transition
temperature of 49 8C and a pyroelectric coefficient of at least 280 mCm
2
K
1
at
20 8C. There are difficulties in exploiting materials which exhibit first-order
transitions, first because they exhibit hysteresis – the transition occurs at a higher
temperature when the temperature is rising than when it is falling – and second
because it would be difficult in most applications to keep the pyroelectrics in a
sufficiently constant temperature environment. A number of materials are used
at temperatures well below their Curie points where, although the pyroelectric
coefficients are smaller, they vary less with the ambient temperature.
For practical purposes, the very small signals generated by pyroelectric
elements must be amplified. The most widely used first stage consists of a field
effect transistor (FET) which responds to electric potential rather than to charge.
In this case, it is advantageous for the material to have a low permittivity to match
the low input capacitance of the FET. Therefore the compositions with high
412 PYROELECTRIC MATERIALS
TEAMFLY
Team-Fly
®
permittivities which are exploited as dielectrics and piezoelectrics are unsuitable,
and special materials have been developed for pyroelectric applications.
7.2 Infrared Detection
Pyroelectric materials are used mainly for the detection of infrared radiation.
The elements for the detectors are typically thin slices of material (e.g.
1.061.060.1 mm) coated with conductive electrodes, one of which is a good
absorber of the radiation.
Figure 7.2 illustrates a detector at a temperature T above its surroundings. If
radiation at a power density W
i
/A is incident on the face for a time dt, the energy
absorbed is ZW
i
dt. The emissivity Z is for the particular surface, wavelength and
INFRARED DETECTION 413
Fig. 7.1 Form of P
s
(T) for various classes of ferroelectric.
Fig. 7.2 A pyroelectric detector element.

temperature conditions and, because of Kirchhoff’s law, it is also a measure of
the fraction of incident energy absorbed.
If it is assumed that all the power absorbed in time dt is rapidly distributed
through the volume of the element, its temperature will rise by dT where
ZW
i
dt ¼ H dT ð7:5Þ
In this equation H ¼ rcAh is the heat capacity of the element where c is the
specific heat of the pyroelectric material and, in this context, r is its density. In
what follows the product rc, the volume specific heat, is given the symbol c
0
.
If part of the absorbed power is lost to the surroundings by reradiation,
conduction or convection at a rate G per unit temperature excess of the element
over its surroundings, Eq. (7.5) is modified to
ZW
i
dt GT dt ¼ HdT
or
H
_
TT þ GT ¼ ZW
i
ð7:6Þ
in which the dot notation signifies a time derivative. If the incident power is shut
off at t ¼ t
0
,
H
_
TT þ GT ¼ 0 ð7:7Þ
and
T ¼ T
0
exp
t
t
T
ð7:8Þ
where T
0
is the temperature excess at t ¼ t
0
and t
T
¼ H=G is the thermal time
constant.
To obtain a continuous response from a pyroelectric material the incident
radiation is pulsed, and this situation is analysed by assuming that the energy
varies sinusoidally with frequency o and amplitude W
0
. Equation (7.6) then
becomes
H
_
TT þ GT ¼ ZW
0
expðjotÞ
or
_
TT þ
1
t
T
T ¼
Z
Gt
T
W
0
expðjotÞð7:9Þ
Using the integrating factor expðt=t
T
Þ and integrating gives
T ¼
Gt
T
1
t
T
þ jo
1
ZW
i
ð7:10Þ
The pyrocurrent I
p
collected from the electrodes is given by
414 PYROELECTRIC MATERIALS
I
p
¼
_
QQ ¼
_
TT
dQ
dT
ð7:11Þ
where Q is the total instantaneous charge. If it is assumed that the element is
operating in a polar state
dQ ¼ A dP
s
¼ ApdT
and
I
p
¼ pA
_
TT ð7:12Þ
which, after substituting
_
TT from Eq. (7.10), becomes
I
p
¼ jo
ðGt
T
Þ
1
t
T
þ jo
1
pAZW
i
ð7:13Þ
The ‘current responsivity’ r
I
is defined as the modulus of I
p
/W
i
, so that
r
I
¼
I
p
W
i
¼
jopAZ
(
Gt
T
1
t
T
þ jo
!)
1
which, after algebraic manipulation, becomes
r
I
¼
pAZo
G
ð1 þ o
2
t
2
T
Þ
1=2
ð7:14Þ
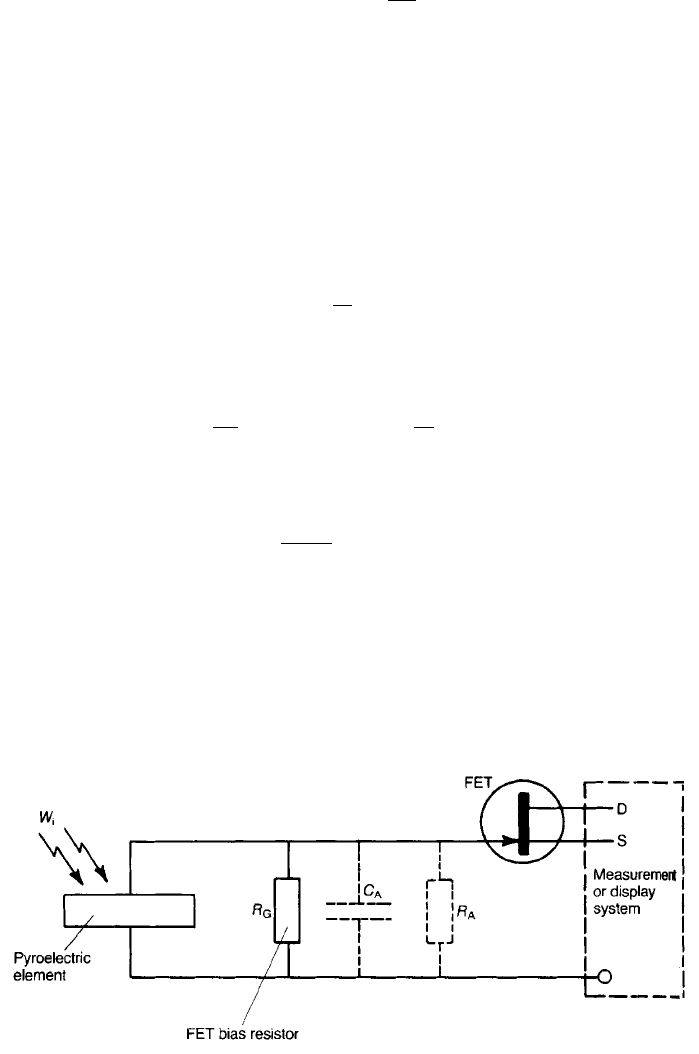
A common arrangement for a detecting system where the voltage u from the
pyroelectric element is fed to the gate of an FET with a high input impedance is
shown in Fig. 7.3. The resistor R
G
correctly biases the FET, and C
A
and R
A
are
respectively the input capacitance and resistance of the amplifying and associated
system. The voltage output is I
p
/Y where the admittance Y is given by
INFRARED DETECTION 415
Fig. 7.3 A common pyroelectric detecting system.
Y ¼
1
R
G
þ
1
R
A
þ joðC
E
þ C
A
Þð7:15Þ
and C
E
is the capacitance of the element. Usually, although not always,
R
A
R
G
and C
A
C
E
, so that
Y R
1
G
þ joC
E
and
jYj¼R
1
G
ð1 þ o
2
t
2
E
Þ
1=2
ð7:16Þ
where t
E
¼ R
G
C
E
is the electrical time constant of the circuit. Therefore the
voltage responsivity r
V
is given by
r
V
¼
u
W
i
¼
I
p
YW
i
¼
r
I
jYj
ð7:17Þ
or
r
V
¼
R
G
pAoZ
Gð1 þ o
2
t
2
T
Þ
1=2
ð1 þ o
2
t
2
E
Þ
1=2
ð7:18Þ
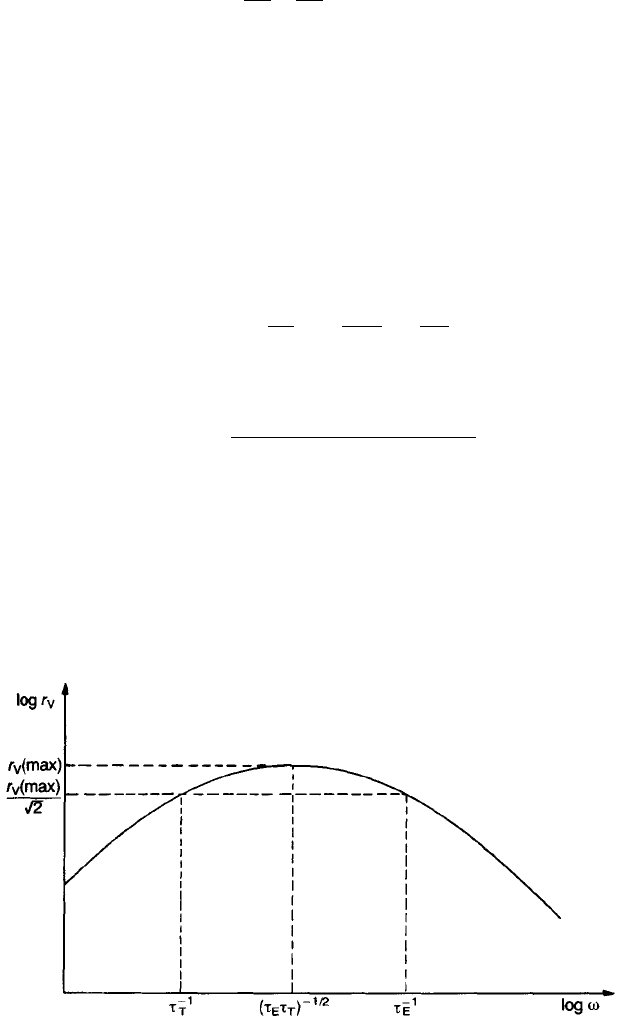
Eq. (7.18) shows that, for maximum sensitivity at low frequency, G should be
minimized by isolating the element to reduce the loss of heat. t
T
can be
minimized by reducing the thickness h of the detecting element so as to reduce its
thermal capacity. r
V
is shown as a function of frequency in Fig. 7.4. There is a
maximum at o
2
¼ 1=t
E
t
T
with only a small variation with frequency between
1/t
T
and 1/t
E
. t
E
and t
T
usually lie in the range 0.1–10 s for high sensitivity. The
maximum value of r
V
is
416 PYROELECTRIC MATERIALS
Fig. 7.4 Variation in voltage responsivity r
V
with frequency. Note that t
1
r
etc. denote o
values.

r
V
ðmaxÞ¼
pAZR
G
Gðt
E
þ t
T
Þ
ð7:19Þ
At high frequencies, when o
2
t
2
T
1, o
2
t
2
E
1 and C
E
C
A
, Eq. (7.18) reduces
to
r
V
¼
pZ
c
0
Aeo
ð7:20Þ
in which e is the permittivity of the material.
Eq. (7.20) suggests a figure of merit,
F
V
¼
p
c
0
e
ð7:21Þ
which describes, in terms of material properties only, the effectiveness of a
pyroelectric element used under defined conditions. Figures of merit only apply
under given defined operational conditions and care has to be exercised in
making use of them. As shown later, the appropriate figure of merit when the
intrinsic ‘noise’ arising from dielectric loss is a dominating influence is
F
D
¼
p
c
0
e
1=2
tan
1=2
d
ð7:22Þ
7.3 E¡ects of Circuit Noise
All signal detectors are required to detect the signal against a background of
‘noise’. Therefore, the signal-to-noise ratio must be optimized or, put another
way, for maximum sensitivity the noise has to be minimized. The sensitivity of
any detector is determined by the noise level in the amplified output signal. In the
case of a pyroelectric detector and its associated circuitry, the principal sources
of noise are Johnson noise, amplifier noise and thermal fluctuations.
The noise level can be expressed in terms of the power incident on the detector
necessary to give a signal equivalent to the noise. If the noise voltage is DV
N
then
the ‘noise equivalent power’ (NEP) is defined by
NEP ¼
DV
N
r
V
ð7:23Þ
Because it is preferable to have a quantity that increases in value as the
performance of the system is improved, the ‘detectivity’ D is defined as
D ¼
1
NEP
ð7:24Þ
EFFECTS OF CIRCUIT NOISE 417

Johnson noise and thermal fluctuations are briefly discussed below, Johnson
noise because it is usually the dominant noise and thermal fluctuations because
they set a lower limit on the achievable noise level.
7.3.1 Johnson noise
Johnson noise arises because the random thermal motion of electrons in an
isolated resistor produces random fluctuations in voltage between its ends,
covering a broad frequency band. It can be shown that
D
I
2
J
¼ 4kTgDf ð7:25Þ
where D
I
2
J
is the mean square Johnson current covering a bandwidth Df, k is
Boltzmann’s constant, T is the absolute temperature and g is the conductance of the
noise generator. In the case of the pyroelectric detector system discussed above,
g ¼
1
R
G
þ oC
E
tan d ð7:26Þ
where oC
E
tan d is the AC conductance of the pyroelectric element.
The root mean square (r.m.s.) noise voltage DV
J
for unit bandwidth is DI
J
=jYj,
where DI
J
is the r.m.s. noise current for unit bandwidth and Y is the admittance.
Therefore
DV
J
¼
ð4kTgÞ
1=2
jYj
ð7:27Þ
At high frequencies, when oC
E
tan d 1=R
G
,
DV
J
4kT
oC
E
!
1=2
tan
1=2
d ð7:28Þ
Therefore D ¼ 1=NEP ¼ r
V
=DV
J
which, on substitution from Eqs (7.20) and
(7.28), gives
D ¼
Z
ðAhÞ
1=2
ð4kToÞ
1=2
p
c
0
ðe tan dÞ
1=2
ð7:29Þ
Under the conditions defined the material parameter to be maximized is p, with
c
0
, e and tan d minimized as required by F
D
(Eq. (7.22)).
7.3.2 Thermal £uctuations
Thermal fluctuations arise even when a body is in thermal equilibrium with its
surroundings through radiation exchange only. Calculation of the mean-square
418 PYROELECTRIC MATERIALS

value of the power fluctuations can be accomplished by the methods of either
classical statistical mechanics or, when the radiation is considered to be
quantized into photons, quantum mechanics.
Both approaches give
DW
2
T
¼ 16ksZT
5
ADf ð7:30Þ
for the mean square power fluctuations covering a bandwidth Df for a body of
surface area A and emissitivity Z at equilibrium at temperature T; s is the Stefan
constant. If the body is assumed to be ‘black’ (Z ¼ 1), for unit area and unit
bandwidth
DW
2
T
¼ 16ksT
5
ð7:31Þ
The r.m.s. value at 300 K is 5.5610
79
m
71
Hz
71/2
W, placing an upper limit of
1.8610
8
mHz
1/2
W
71
on the detectivity D. The highest detectivities achieved in
practice are between one and two orders of magnitude below this.
In ferroelectrics the major contributor to tan d is domain wall movement
which diminishes as the amplitude of the applied field diminishes; the value
applicable to pyroelectric detectors will be that for very small fields. The
permittivity is also very sensitive to bias field strength, as is its temperature
coefficient. The properties of some ferroelectrics – the ‘relaxors’ – are also
frequency dependent. It is important, therefore, to ensure that when assessing the
suitability of a ferroelectric for a particular application on the basis of measured
properties that the measurements have been made using values of the parameters
(frequency, field strength etc.) appropriate to the application. This is not always
done.
7.4 Materials
The properties and figures of merit of a range of pyroelectric materials (at 20 8C
except where indicated) are given in Table 7.1. The table omits a number of
secondary characteristics that determine suitability in particular applications.
Triglycine sulphate (TGS) has high figures of merit but is a rather fragile water-
soluble single-crystal material. It can be modified to withstand temperatures in
excess of its Curie point without depoling, but it cannot be heated in a vacuum to
the temperatures necessary for outgassing without decomposing. It is difficult to
handle and cannot be used in devices where it would be subjected to either a hard
vacuum or high humidity. In contrast, polyvinylidene fluoride (PVDF) has poor
figures of merit but is readily available in large areas of thin film. It is
considerably more stable to heat, vacuum and moisture than TGS. It is
mechanically robust and, as indicated by Eq. (7.18), can have its voltage
sensitivity enhanced by the use of a large area. It also has low heat conductivity
MATERIALS 419

420 PYROELECTRIC MATERIALS
Table 7.1
Properties of some pyroelectric materials. (Data should be regarded
as approximate)
Material and form
p/mCm
2
K
1
e
0
r
tand
c
0
/MJ m
3
K
1
T
c
/8CF
V
/m
2
C
1
F
D
/TPa
1/2
*
TGS, (single crystal; 35
8C)
280
38 0.01
2.3
49 0.36
66
DTGS (single crystal; 40
8C)
550
43 0.02
2.6
61 0.53
83
LiTaO
3
(single crystal)
230
47 0.001
3.2
665 0.17 110
(SrBa)Nb
2
O
6
(single crystal)
550
400 0.001
2.3
121 0.07
70
modified PZ (polycrystalline**)
400
290 0.003
2.5
230 0.06
58
300
{
0.014
{
0.06
26
modified PT (polycrystalline)
350
220 0.01
2.5
250 0.07
32
220
{
0.03
{
0.07
18
PVDF (film)
27
12 0.01
2.4
80 0.1
10
P(70VDF/30TrFE)*** (film)
33
7.4 0.017
2.3
121 0.22
13.6
0.25PZ 0.75PT (sol-gel thin film)
220
350
{
0.008
{
2.7
– 2.5
17
(sputtered)
450
300 0.01
2.7
– 2.6
14
‘BST (a) (0.67Ba 0.33Sr)TiO
3
(ceramic; 0.6 V
mm
1
)
7000 8800 0.004
–
25 –
124
‘PST (b) Pb(Sc
1/2
Ta
1/2
)O
3
(sputtered on Si; 10 V
mm
1
;508C)
850
600 0.0025
–
10 –
80
‘PST’ (c)
(ceramic; 4 V
mm
1
;408C)
3500 2000 0.005
2.7
25 0.06 110
*F
D
is also expressed in the equivalent units, 10
6
Pa
1=2
: TGS
¼triglycine sulphate: DTGS
¼deuterated TGS: **‘PZFNTU’ see text: *** co-polymer
of vinylidene
fluoride and trifluoroethylene.
{
Measured at 33 Hz; all other dielectric data measured at
1 kHz.
Properties measured at 25
8C, unless indicated in parentheses. Where appropriate
bias electric fields in parentheses.
(a) From [2].
(b) After P. P. Donohue, M.A. Todd
et al. (2001)
Integrated Ferroelectrics
, 41, 25–34.
(c) After N. M. Shorrocks
et al.
(1990) Ferroelectrics
,
106, 387–92.
and low permittivity so that both thermal and electrical coupling between
neighbouring elements on the same piece of material are minimized. Its high
tan d is a disadvantage.
Lithium tantalate is a single-crystal material that is produced in quantity by
the Czochralski method (see Section 3.11) for piezoelectric applications and is
therefore readily available. It is stable in a hard vacuum to temperatures that
allow outgassing procedures. It is insensitive to humidity. It is widely used where
precise measurements are to be made.
Strontium barium niobate is a single-crystal material with the tungsten bronze
type of structure which is made by the Czochralski method but has yet to find a
major use. It has relaxor characteristics of the type shown in Fig. 7.1 which give
it a high pyroelectric coefficient and detectivity, but its high permittivity lowers
the figure of merit F
V
.
The ceramic based on lead zirconate (PZ) has intermediate figure of
merit values. Its final form as a thin plate is obtained by sawing up a hot-
pressed block and lapping and polishing to the required dimensions. Similar
methods must be applied to single crystals and so the costs of manufacture are
not very different.
The adaptability of ceramics is well illustrated by the development of PZ for
pyroelectric devices. It was found initially that the addition of about 10 mol%
lead iron niobate (PbFe
1/2
Nb
1/2
O
3
) to lead zirconate converted it from an
antiferroelectric to a ferroelectric material with a relatively low permittivity and a
high pyroelectric coefficient. However, this material was found to undergo a
transition between two ferroelectric rhombohedral forms at 30–40 8C which
rendered its pyroelectric output unstable. The replacement of about 5% of the B-
site atoms by Ti shifted this transition to around 100 8C without significantly
increasing the permittivity. For some devices a somewhat lower resistivity is
advantageous since it eliminates the need for a high-value, and expensive,
external resistor. It was found that replacement of 0.4–0.8% of the B-site atoms
by uranium controlled the resistivity in the 10
8
–10
11
O m range, as shown in
Fig. 7.5; at the same time the permittivity and loss were reduced. The final
composition Pb
1.02
(Zr
0.58
Fe
0.20
Nb
0.20
Ti
0.02
)
0.994
U
0.006
O
3
(PZFNTU), although
complex, can be manufactured reproducibly.
A pyroelectric response is shown by any dielectric to which an electric
field is applied provided there is a variation in permittivity with temperature
(see Eq. 7.3). The permittivity of ferroelectrics is strongly temperature-
dependent close to the Curie point and high pyroelectric figures of merit can
be obtained by operating them in the region of T
c
and with a biasing
electric field. This is because of a combination of high p and low tan d.A
little below T
c
a sufficiently strong biasing field can sweep the domains from
the material therefore removing the contribution to losses from domain wall
movements. Table 7.1 includes data for ‘BST’ and ‘PST’ operated in this
mode.
MATERIALS 421
