Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.


hexagonal crystals the anisotropy energy E
Kh
depends only upon the angle y
between the c axis and the magnetization vector, and is almost independent of
direction in the basal plane. It can be expressed approximately as
E
Kh
¼ K
1h
sin
2
y þ K
2h
sin
4
y ð9:29Þ
The energy minimum occurs for y ¼ 0, making the c axis the ‘easy’ direction.
The concept of an anisotropy magnetic field H
A
, which is referred to simply as
the ‘anisotropy field’, is also introduced to describe magnetic anisotropy. The
anisotropy energy is considered to be that of the saturation magnetization
moment M
s
in an induction m
0
H
A
, i.e. E
K
¼m
0
H
A
M
s
cos y, where y is the angle
between H
A
and M
s
.
9.1.8 Magnetostriction
Because of the spin–orbit lattice coupling referred to in the previous section,
changes in the spin directions result in changes in the orientation of the orbits
which, because they are restrained by the lattice, have the effect of slightly
altering the lattice dimensions. This effect is known as magnetostriction.
The magnetostriction constant l
m
is defined as the strain induced by a saturating
field; it is given a positive sign if the field causes an increase in dimensions in the
field direction. For single crystals l
m
varies with the crystallographic direction, and
so for the ceramic form it is an average of the single-crystal values. l
m
values for
some polycrystalline ferrites are given in Table 9.2.
9.1.9 Weiss domains
The fact that spontaneous magnetization exists in, for example, a piece of iron,
and yet the overall magnetization can be zero, is explained by the existence of
482 MAGNETIC CERAMICS
Table 9.1 Room temperature anisotropy constants
of some important ferrites
Ferrite K
1
/kJ m
73
Fe
3
O
4
711
Mn
0.98
Fe
1.86
O
4
72.8
Co
0.8
Fe
2.2
O
4
+290
NiFe
2
O
4
76.2
CuFe
2
O
4
76.0
Mn
0.45
Zn
0.55
Fe
2
O
4
70.38
Ni
0.5
Zn
0.5
Fe
2
O
4
73
BaFe
12
O
19
+330
TEAMFLY
Team-Fly
®

domains. Below its Curie temperature a ferromagnetic or ferrimagnetic body
consists of a large number of small domains, each spontaneously magnetized to
saturation. Each grain or crystallite in a polycrystalline magnetic ceramic may
contain a number of domains, each differing from its neighbour only in the
direction of magnetization.
A single crystal with uniform magnetization, i.e. a single-domain single crystal,
has magnetostatic energy due to the external magnetic field which it generates. If
the crystal is divided into oppositely oriented parallel domains, the energy will be
greatly reduced since the flux can now pass from one to another of the closely
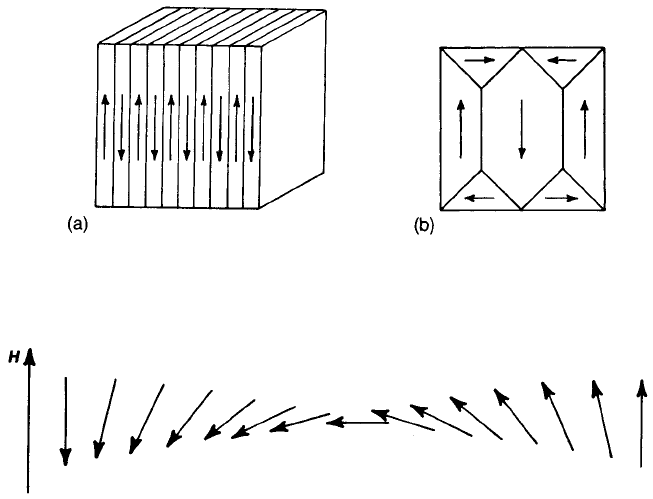
adjacent domains (Fig. 9.8(a)). In cubic materials, such as spinels and garnets,
zero magnetostatic energies are possible through the formation of closure
domains (Fig. 9.8(b)) since the external flux is now close to zero. Even so, the
system has magnetoelastic energy because magnetostriction leads to straining
between the long and the triangular domains.
The boundary between two adjacent domains is known as a domain wall or
Bloch wall – the latter term after F. Bloch who proved in 1932 that
magnetization cannot change discontinuously at a domain boundary. A Bloch
wall is the region between two domains in which the elementary spin moments
change smoothly from one orientation to another. For example, in the case of
the antiparallel domains the change in direction of the vectors in moving from
one domain to an adjacent one would be as shown diagrammatically in Fig. 9.9.
The walls have widths in the range 10–100 nm and an associated energy in the
range (1–10)610
74
Jm
72
.
An important property of a Bloch wall is its mobility. It can be seen from
Fig. 9.9 that the application of H in the sense shown will cause the wall to move
to the left by a series of minor rotations of the vectors. A more detailed
consideration of wall displacements shows them to be of two types, reversible or
irreversible; which one occurs depends upon the size of the displacement and the
MAGNETIC CERAMICS: BASIC CONCEPTS 483
Table 9.2 Saturation magnetostriction constants
for some polycrystalline ferrites
Composition l
m
/10
76
Fe
3
O
4
+40
MnFe
2
O
4
75
CoFe
2
O
4
7110
NiFe
2
O
4
726
Ni
0.56
Fe
2þ
0:44
Fe
2
O
4
0
Ni
0.5
Zn
0.5
Fe
2
O
4
711
MgFe
2
O
4
76
TbFe
2
a ‘giant’ magnetostrictor 410
3
*
*Included for comparison (H. Uchida et al. (2002) Progress in thin
films of giant magnetostrictive alloys, J. Mag. Mag. Mater., 239,
540–5).
disposition of wall energy minima, which in turn depend upon crystal lattice
defects and inhomogeneities of various types.
9.1.10 Magnetization in a multidomain crystal
The most characteristic feature of ferromagnetic or ferrimagnetic materials is the
relationship between B and H (or M and H) – the hysteresis loop (Fig. 9.10) –
which can now be considered in more detail in terms of the magnetization
processes in a multidomain single crystal. The line deOba – the ‘virgin curve’ –
represents the relationship determined experimentally when the specimen is
demagnetized before each measurement of the induction for a given field. The
change in B, very near to the origin, represents magnetization by reversible Bloch
wall displacements, and the tangent OC to this initial magnetization curve is
called the initial permeability m
i
. The steep rise in B represents magnetization by
irreversible Bloch wall displacements as the walls break away from their pinning
points, and the region ba represents magnetization by reversible and irreversible
domain rotations from one easy direction to another more favourably aligned
with the applied field. The latter process requires high field strengths because the
magnetization within a domain is rotated against the anisotropy field.
484 MAGNETIC CERAMICS
Fig. 9.8 Idealized magnetic domain configurations: (a) antiparallel domains; (b) flux closure
domains.
Fig. 9.9 The change in spin orientation across the width of a Bloch wall.
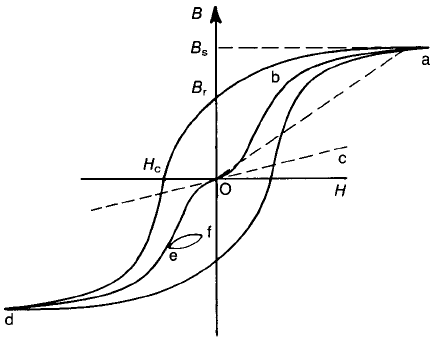
The slope of Oa, from the origin to the tip of the loop, gives the amplitude
permeability m
a
which has a maximum value when the peak field corresponds to
the point b on the virgin curve. If a relatively small alternating field is
superimposed on a static field, a minor loop such as ef is obtained, and the
amplitude permeability of this is known as the incremental permeability m
D
.As
far as applications are concerned, m
i
is important for inductors where only small
alternating fields are encountered, m
a
is important for power transformers when
large alternating fields are involved and m
D
is important for transductors to
which both alternating and static fields are applied.
If, after the material has been magnetically saturated to the value B
s
, the field
is reduced to zero, the magnetization vectors rotate out of line with the field
towards the nearest preferred direction which is determined in part by
magnetocrystalline anisotropy. The magnetization is thus prevented from
complete relaxation to the ‘virgin’ curve and hence, for zero field, there is a
remanent induction B
r
. In order to reduce the induction to zero a reverse field H
c
has to be applied. The coercive field or ‘coercivity’ H
c
depends in part on
crystalline anisotropy, as might be expected.
Because of hysteresis, energy is dissipated as heat in a magnetic material as it is
taken round a complete B –H loop, and the hysteresis energy loss W
h
per unit
volume of material is
W
h
¼
þ
BdH ð9:30Þ
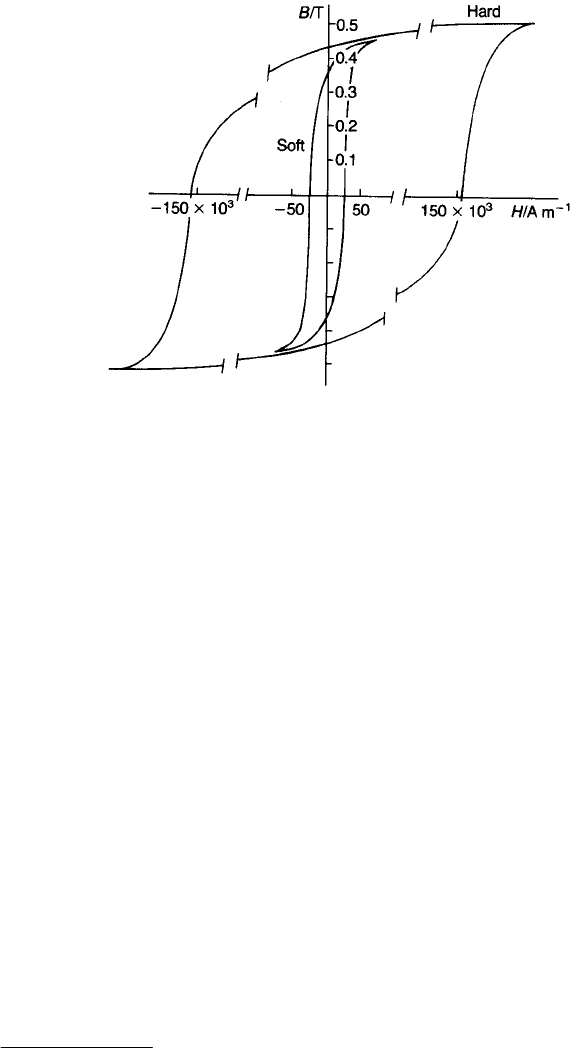
Magnetic materials are usually characterized as ‘hard’ or ‘soft’, depending on the
magnitude of their coercivity and Figure 9.11 shows typical loops.
MAGNETIC CERAMICS: BASIC CONCEPTS 485
Fig. 9.10 Magnetic B–H hysteresis loop.
9.2 Model Ferrites
9.2.1 Spinel ferrites: model NiOFe
2
O
3
Magnetite (Fe
3
O
4
), a naturally occurring ferrite, is the earliest known magnetic
material and was exploited as lodestone
{
many hundreds of years ago. Its
composition can be written FeOFe
2
O
3
, when the structural relationship to the
mineral spinel (MgOAl
2
O
3
) is apparent. There are many other possible
compositions with the general formula MeOFe
2
O
3
, in which Me represents a
divalent ion such as Mn
2+
,Fe
2+
,Co
2+
,Ni
2+
,Cu
2+
or Zn
2+
, or a combination
of divalent ions with an average valence of 2. The substitutional possibilities are
considerable, which has led to the extensive technological exploitation of ferrites.
In the spinel crystal structure the oxygen ions form a cubic close-packed array
in which, as discussed in Chapter 2 (Fig. 2.1), two types of interstice occur, one
coordinated tetrahedrally and the other octahedrally with oxygen ions. The cubic
unit cell is large, comprising eight formula units and containing 64 tetrahedral
and 32 octahedral sites, customarily designated A and B sites respectively; eight
of the A sites and 16 of the B sites are occupied. The unit cell shown in Fig. 9.12
is seen to be made up of octants, four containing one type of structure (shaded)
and four containing another (unshaded). In this representation some of the
A-site cations lie at the corners and face-centre positions of the large cube; a
486 MAGNETIC CERAMICS
Fig. 9.11 Hysteresis loops illustrating the distinction between magnetically ‘soft’ and ‘hard’
materials.
{ The attractive power of magnetite was known to the Greeks (c. 100 BC); by the early 17th century – and
probably much earlier – it was used by the Chinese to magnetize compass needles. Hence, ‘lode’ in
‘lodestone’ is synonomous with ‘lode’ in ‘lodestar’ (the Pole star).
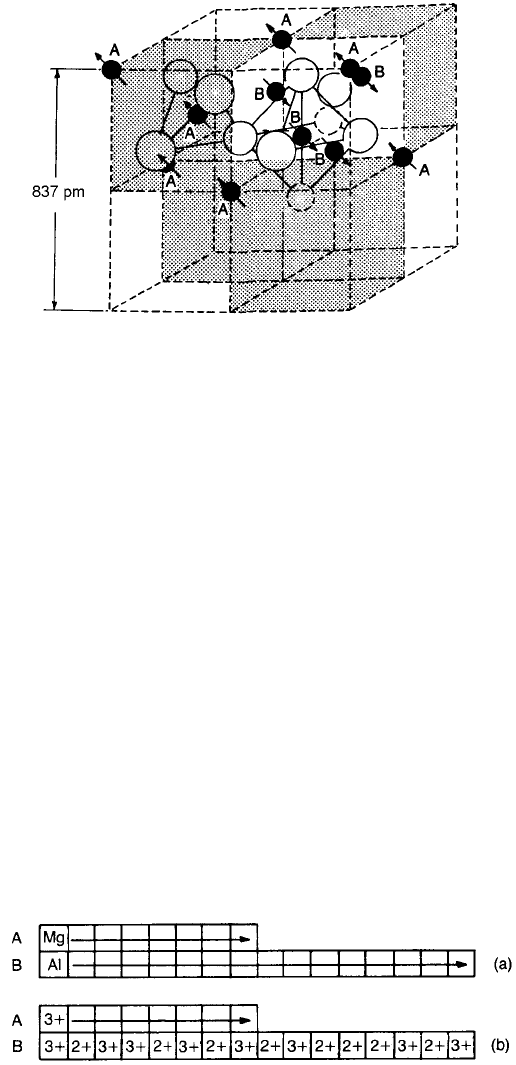
tetrahedral and an octahedral site are shown. The close-packed layers of the
oxygen ion lattice lie at right angles to the body diagonals of the cube. The
arrows on the ions, representing directions of magnetic moments, indicate that
the B-site ions have their moments directed antiparallel to those of A-site ions,
illustrating the antiferromagnetic coupling.
The occupancy of sites in the spinel-type minerals is conveniently represented
with the help of the diagrams in Fig. 9.13. In the case of the mineral spinel the
divalent ions (Mg) occupy the A sites and the trivalent ions (Al) the B sites
(Fig. 9.13(a)). This is known as the normal spinel structure. In the spinel ferrites
(MeFe
2
O
4
) different ions exhibit different site preferences. In nickel ferrite, for
instance, the Ni
2+
ions occupy B sites along with an equal number of randomly
distributed Fe
3+
ions, whilst the remaining Fe
3+
ions occupy A sites. This is
termed an inverse spinel structure (Fig. 9.13(b)). In practice, the structures of the
spinel-type magnetic ceramics are a blend of the normal and inverse types, with
the oxidation states and distribution of cations amongst the lattice sites
depending upon thermal history, i.e. on firing schedules and kiln atmosphere.
Attention is now given to the ordering of the magnetic moments. The spin-
ordering forces are of the ‘superexchange’ kind, so called because they act
between the resultant spin on the cations through the agency of an intervening
oxygen ion. Superexchange forces acting between A ions, between B ions and
MODEL FERRITES 487
Fig. 9.12 The unit cell of a magnetic inverse spinel.
Fig. 9.13 Diagrammatic representation of site occupancy in (a) normal and (b) inverse spinels.

between A and B ions are referred to as AA, BB or AB interactions, and in each
case the forces tend to hold the spins of nearest-neighbour cations antiparallel.
The AB interaction is the strongest and in consequence moments on the A ions
are directed antiparallel to those of the B ions. Ne
´
el introduced and developed
this theory in 1948.
Before proceeding further with the analysis, the magnitude of the magnetic
moments associated with the various cations must be known and, because of
orbital moment quenching, only the spin moments need be considered when
calculating the moment of the ions. For example, as a first step in calculating the
magnetic moment of NiOFe
2
O
3
it is readily shown that the resultant spin
quantum numbers for Ni
2+
and Fe
3+
ions are 1 and 5/2 respectively. Because the
spinning electron produces twice the magnetic moment expected from its angular
momentum, the corresponding magnetic moments are 2m
B
and 5m
B
. Also,
because all the A moments are directed antiparallel to the B moments, it is
evident from Fig. 9.13(b) that there is a resultant moment of 16m
B
per unit cell
which is due to the eight Ni
2+
ions. The saturation magnetization can now be
calculated from the cell dimensions:
M
s
¼
16m
B
cell volume
¼
16 9.27 10
24
ð8.37 10
10
Þ
3
2:5 10
5
Am
1
This compares well with the experimentally determined value (approximately
3610
5
Am
71
). The discrepancy is probably due partly to the assumption that
NiOFe
2
O
3
has the ideal inverse spinel structure and partly to the incomplete
quenching of the orbital moment.
An elegant confirmation of the essential correctness of the theory lies in the
explanation of why the addition of a non-magnetic ion such as Zn to a spinel
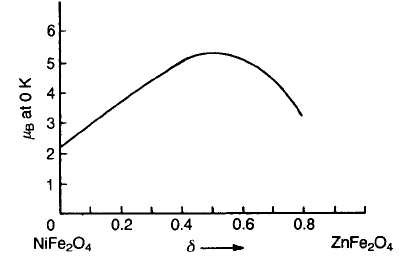
ferrite should lead to an increase in saturation magnetization. The magnetic
moment per formula unit MeFe
2
O
4
, where Me represents Ni
2+
or Zn
2+
, is shown
in Fig. 9.14 as a function of zinc substitution. Zinc ferrite is a normal spinel,
indicating that Zn ions have a preference for the A sites, so that on substituting
zinc for nickel the occupancy becomes (Fe
3þ
1d
Zn
2þ
d
)(Fe
3þ
1þd
Ni
2þ
1d
)O
4
, in which the
first and second brackets indicate occupancy of the A and B sublattices
respectively. Thus the antiparallel coupling between moments on A and B sites is
reduced because the occupancy of A sites by magnetic ions is reduced, and as a
consequence the Curie point is lowered (see Fig. 9.18 below). However, the
excess of moments on octahedral sites over those on tetrahedral sites is increased
so that the magnetization is increased. The data plotted in Fig. 9.14 confirm this
model up to d 0.4. The fall in magnetization for higher values of d is due to the
reduced antiparallel coupling between the A and B sites, and it becomes zero
when d ¼ 1, i.e. (Zn
2+
)(Fe
3þ
2
)O
4
.
Generally speaking, the spinel ferrites have low magnetic anisotropies and are
magnetically ‘soft’; exceptions are those containing Co
2+
which is itself strongly
488 MAGNETIC CERAMICS
magnetically anisotropic. Cobalt spinel ferrites can have coercivities approaching
10
5
Am
71
, placing them firmly in the ‘hard’ category.
9.2.2 Hexaferrites: model BaFe
12
O
19
Barium hexaferrite (BaFe
12
O
19
) is the model for a family of ‘M-type ferrites’, so-
called because they are based upon the hexagonal magnetoplumbite, or M-
structure. Its crystal structure, though related to that of the spinels, is very much
more complex. The large unit cell (c ¼ 2.32 nm; a ¼0.588 nm) contains two
formula units, i.e. a total of 64 ions. The Ba
2+
and O
27
ions together form a
close-packed structure with some of the layers cubic close-packed and others
hexagonal close-packed. The origins of the magnetic properties are basically the
same as those already discussed and can be summarized as follows: of the 12
Fe
3+
ions in a formula unit, nine are on octahedral sites two on tetrahedral sites
and one on a five-coordinated site; seven of the ions on octahedral sites and the
one on a five-coordinated site have their spins in one sense, and the remainder
are oppositely directed. Thus there are four more ions with spins in the one
sense than there are with spins in the other and, since there are five electrons
with parallel spins in each Fe
3+
ion, there are 20 unpaired spins per formula
unit, leading to a saturation magnetization of 20 m
B
per cell volume. BaFe
12
O
19
has a high magnetic anisotropy (Table 9.1) with its ‘prefered direction of
magnetization’ (‘easy’ direction) along the c-axis. Various substitutions are
made to tailor intrinsic magnetic properties, for example Sr for Ba, and partial
substitution of Al for Fe to increase coercivity [8].
9.2.3 Garnets: models Y
3
Fe
5
O
12
(YIG)
‘Garnet’ is the name of a group of isostructural minerals with the general
composition 3R
0
O R@
2
O
3
3SiO
2
. Examples are 3CaO Al
2
O
3
3SiO
2
MODEL FERRITES 489
Fig. 9.14 Saturation magnetization per ‘formula unit’ for the ferrite (Fe
3þ
1d
Zn
2þ
d
)
(Fe
3þ
1þd
Ni
2þ
1d
)O
4
as a function of d.
(grossularite), 3CaO Fe
2
O
3
3SiO
2
(andradite) and 3MnO Al
2
O
3
3SiO
2
(spes-
sarite). Yttrium iron garnet (YIG) is the best known of a family of ferrimagnetic
garnets because of its importance as a microwave material. It has a large cubic
unit cell (a 1200 pm) containing 160 atoms.
The general formula for the ferrimagnetic garnets is written R
3
Fe
5
O
12
, where
R stands for yttrium in the case of YIG; the yttrium can be totally or partially
replaced by one of the lanthanides such as lanthanum, cerium, neodymium,
gadolinium etc. Therefore the structure contains two types of magnetic ion, iron
and one of the rare earth group. Whilst the contribution to the magnetization
from the orbital motion of the electrons in elements of the first transition series is
close to zero (quenching) because of the orbital–lattice coupling, that of the
electrons in the lanthanide ions has a significant effect. The unpaired electrons of
the first series elements are in the outermost 3d group and therefore are not
shielded from the crystal field which is responsible for quenching. In the
lanthanide ions the unpaired electrons in the 4f group are shielded by the 5s5p
electrons and there is therefore an orbital contribution in addition to that of the
unpaired spins. As a consequence the contribution of the lanthanide ions to the
magnetization is somewhat greater than would be estimated from the simple
rules governing the elements of the first transition series. A further consequence
of this shielding is that the coupling of lanthanide ions to other magnetic ions is
weaker than that between the ions of the first transition series.
The lattice site occupancy is conventionally represented by the formula
{R
3
}
c
[Fe
2
]
a
(Fe
3
)
d
O
12
, where [ ]
a
indicates ions on octahedral sites, ( )
d
indicates
ions on tetrahedral sites and { }
c
indicates ions on 12-coordinated sites. There is
strong coupling with antiparallel spins between ions on the a and d sites, and
thus, since all are Fe
3+
, the net contribution is 5m
B
; the rare earth ions on the c
sites have their unpaired spins coupled antiparallel to the Fe
3+
on d sites and so
contribute 3m
R
, where m
R
is the strength of the moment of the R ion measured
in Bohr magnetons. The resultant magnetization per formula unit, measured in
Bohr magnetons, is therefore
M ¼ 5 3m
R
in which m
R
is greater than 7 for gadolinium, terbium and dysprosium, and falls
off to 3.5 for thulium, 2.7 for ytterbium, and zero for lutetium and yttrium.
When m
R
45/3, M is negative and its value at 0 K is dominated by the
contribution from the rare earth ions. At higher temperatures the rare earth
contribution decreases because of the weak coupling between (Fe
3+
)
d
and {R}
c
,
and thus the magnetization first falls to zero and then increases again. The zero
magnetization point is known as the compensation point. Since at high
temperatures magnetization in those rare earth garnet ferrites containing no
substitute for Fe
3+
depends mainly on the (Fe
3+
)
d
–[Fe
3+
]
a
coupling, they all
have approximately the same Curie points.
490 MAGNETIC CERAMICS

The types of saturation magnetization–temperature characteristic for the
various garnet ferrites are summarized in Fig. 9.15. Figure 9.16 illustrates for a
series of the yttrium gadolinium iron garnets an important feature associated
with a compensation point which is exploited in certain applications: the
magnetization can be arranged to be almost independent of temperature over a
chosen temperature range. For example, for the composition corresponding to
x¼0.6, i.e. Y
1.2
Gd
1.8
Fe
5
O
12
, the saturation magnetization is relatively stable over
a wide temperature range centred around 50 8C.
A further important feature of the garnets is their high electrical resistivity
resulting in a high Q suitable for certain microwave devices. This is because they
MODEL FERRITES 491
Fig. 9.15 Variation of saturation magnetization with temperature for various garnets.
Fig. 9.16 Variation of saturation magnetization with temperature for Y
3(17x)
Gd
3x
Fe
5
O
12
for
various x values.
