Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

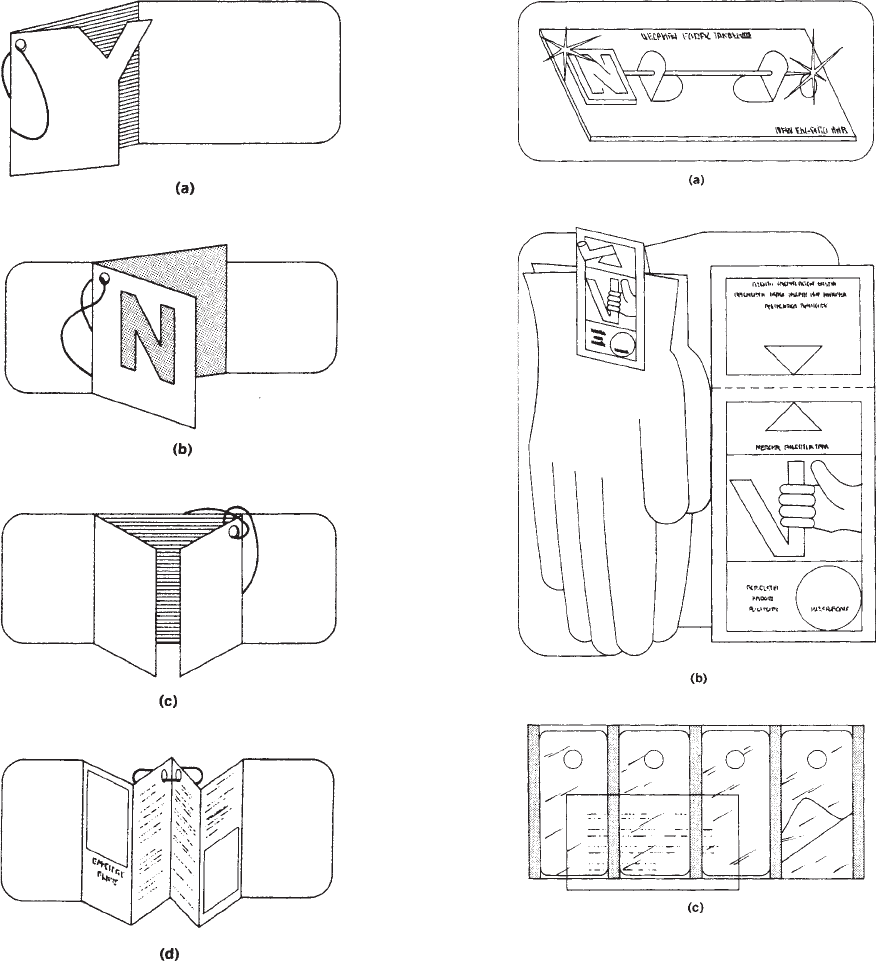
shows a hang tag of clear plastic partially coated with a
pressure-sensitive adhesive. The coated portion of the tag
is applied to the merchandise, and the die-cut hole is used
to hang the merchandise on the rack.
Tags used for price marking are generally produced as
continuous tags. These tags satisfy the special needs of
large retail organizations by affording them the opportu-
nity to ship preprinted price tags along with stock ship-
ments to their various distribution locations. The tags
come in a variety of sizes and shapes are generally affixed
with plastic barbs by tagging guns to prevent price switch-
ing. The accelerated use of computers in the retail indus-
try has had a dramatic effect on the appearance and use of
tags. Price-marking tags are now being produced with
magnetic stripes (Figure 7a), which can contain informa-
tion such as style, lot number, and manufacturer. In
addition, bar code and OCR (optical character recognition)
technologies have gained acceptance in the retail tag
market.
Tags can also aid in theft prevention, which is one of
the biggest concerns in the retail industry. Hard plastic
tags such as those shown in Figure 7b are affixed to
articles of clothing in many large retail stores. The tags
must be removed at the time of sale to prevent triggering
an alarm as the garment passes through a special gate
that contains a scanner that is sensitive to the tag.
Because of their bulk, these tags are used primarily for
clothing. Recent innovations in miniaturization have
Figure 5. (a) Die-cut outline. (b) Three-dimensional die-cut tag.
(c) Gatefold tag. (d) Accordion-fold tags.
Figure 6. (a) Jewelry display tag. (b) Display-rack tag. (c)
Transparent hang tag.
1188 TAGS
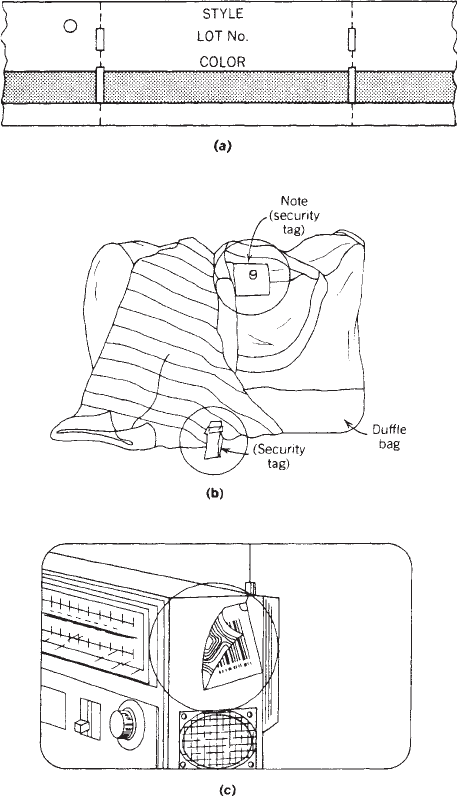
produced tags with what appears to be a strip of alumi-
num foil (Figure 7c), but which is actually a built-in
security device. If not deactivated by another strip of foil
affixed at the time of sale, these circuits interrupt radio-
waves being transmitted from the exit gate and set off an
alarm. These tagging systems are currently marketed by
companies that sell retail-security systems.
Industry indicators predict that the future of the tag
industry in retail operations will not be adversely affected
by advances in computer technology. As long as there are
products to be identified, inventoried, and priced, there
will be a market for tags of all kinds in the retail industry.
BIBLIOGRAPHY
Linda L. Butler, ‘‘Tags’’ in M. Bakker, ed., The Wiley Encyclopedia
of Packaging Technology, 1st edition, John Wiley & Sons,
New York, 1986, pp. 624–628; 2nd ed., R. J. Brody and K. S.
Marsh, eds., 1997, pp. 875–879.
TAMPER-EVIDENT PACKAGING
JACK L. ROSETTE
Forensic Packaging Concepts,
Inc., Jefferson City, Tennessee
HISTORY OF THE PROBLEM
Since 1982 the entire industry, including the consumer,
has become aware of tamper-evident packaging. This
awareness has benefited the consumer by a reduction in
loss of life due to consumption to adulterated products
from tampering. Never before has an industry reacted so
swiftly to resolve a problem that could be the ruination of
most manufacturers.
In 1982, seven people in the Chicago area died from
consuming cyanide-laced Tylenol capsules. The incident
resulted in a total product recall, massive negative pub-
licity for the product, new requirements for safe packa-
ging, and a federal statute making product tampering a
crime. Since that time, the packaging industry has become
visible to most consumers.
HISTORY OF INCIDENTS
Although there were incidents prior to 1982, product tam-
pering became highly visible after the Chicago incidents.
The exact number of incidents per year is unknown because
of various methods of reporting. Beginning with the 1982
deaths, there have been deaths from product tampering
that are widely known such as those in 1982, 1986, and
early 1991. All deaths due to targeted tampering are not
included, because we are discussing random tampering, not
tampering with a specific person in mind. It is probably a
coincidence on the timing as the various incidents are
unrelated, and in some instances the violator in earlier
cases was incarcerated at the time of the ensuing incidents .
According to government figures, the problem peaked
in the United States in 1986, when there were almost 1800
claims of possible product tampering. The number has
decreased to around 500 per year now. Is the decrease due
to better packaging or less interest on the part of potential
violators, due to the penalties for violating product-tam-
pering laws? Considering the experience in assisting law-
enforcement agencies, the decrease appears to be caused
by a change in the way claims are recorded as well as a
reluctance on the part of manufacturers to report all
claims of possible product tampering.
If the experience of other developed nations is an
indication of the future, it is expected that the problem
of product tampering will escalate in this country in the
future. Every developed nation has experienced product-
tampering incidents. The main difference between domes-
tic incidents and what is happening elsewhere is the
motive of the tamperers: extortion prior to injury, with
money appearing to be the primary motive, versus appar-
ently random tampering without prior threat in this
country. Most developed nations are either implementing
or modifying their rules on the use of tamper-evident
Figure 7. (a) Marking tag with magnetic stripe. (b) Plastic
electronic security tags. (c) Security tag with miniaturized radio
circuit.
TAMPER-EVIDENT PACKAGING 1189
packaging. Some features as they are used in the United
States would have to be modified, or the use of a secondary
feature would be required to meet the standards of various
countries.
The problem of state-sponsored terrorism is so serious
that a series of meetings were held in the late 1970s were
the problem was addressed in great detail. Participating
in the meetings were representatives of the FBI, Com-
merce Department, Defense Department, State Depart-
ment, and CIA. One of the chief concerns at the time was
the threat of retail-product tampering by a state-spon-
sored organization. The term ‘‘state-sponsored terrorism’’
refers to any group of terrorists that is supported finan-
cially, logistically, or in intelligence by a government body
of any country. It has been documented that training in
ways to tamper with retail products and how to use such
acts to further their agenda is being conducted in certain
countries that sponsor terrorist groups. Picture, if you
will, the potential for disaster that exists if a potential
tamperer has the financial and human resources available
to build a complete packaging line and can print duplicate
labels. It sounds far-fetched, but a similar incident oc-
curred in South America when a drug organization bought
a beverage plant to smuggle cocaine into the United
States. In that case at least one bottle in a specially
marked case contained the drug in a liquid form. When
the contents of the bottle was distilled in the United
States, it would yield a powder that could then be cut in
strength and distributed to the dealers. Unfortunately,
one bottle was overlooked and sold to a consumer who died
from a massive cocaine overdose.
While most incidents of retail product tampering in the
United States have been perpetrated by individuals, the
potential exists for even greater harm from the same acts
committed by a state sponsored terrorist group.
THE FDA RULE
In 1982 the FDA passed a rule (21 CFR 211.132) requiring
the use of tamper-evident packaging on all over-the-
counter (OTC) drugs and some cosmetics, while ignoring
other products they regulate. The fact that your product is
not required to use tamper-evident (TE) packaging does
not protect you in the event of a claim of product
tampering.
According to attorneys specializing in packaging and
product liability, product tampering is a foreseeable pos-
sibility, and manufacturers have a responsibility to protect
consumers against such possible acts. If your product in
an adulterated form could harm a consumer, you have a
responsibility to protect the product and consumer against
such acts. This means that the use of tamper-evident
packaging transcends FDA regulations.
The Rule specifies which packaging features are accep-
table for use in providing resistance to tampering, and the
list was modified in 1988. At the current time there is a
proposal to modify the Rule again and to make the
acceptability of a feature based on performance.
What if the feature you want to use is not on the
approved list of the FDA? The FDA has a procedure by
which methods of providing protection that are not on the
approved list may obtain approval on a case-specific basis.
To obtain approval, samples of the complete package must
be submitted to the FDA along with a written request for a
waiver. Contact the local FDA compliance officer to find
out the current procedure to obtain approval.
The inclusion of a specific form of protection does not
warrant that the feature will deter violation, nor does it
prevent legal action in the event of a claim of injury
related to product tampering. There are variances in
designs, and even tooling of the same design that affect
the effectiveness of each feature. Any evaluation of a
package relates to the exact components used in the tests,
and material from a different manufacturer will usually
result in a different level of effectiveness, much the same
as using a different resin or closure liner will affect a
stability study.
CONSUMER PREFERENCES
Studies into consumer preferences for tamper-evident
packaging have consistently revealed that the consumer
prefers products that are resistant to tampering and
prefer the features to be shelf-visible. The same studies
indicated a willingness to pay slightly more than a com-
peting brand that is not TE. The exact amount is incon-
sistent among the studies, but the consistent replies of
concern indicate consumer awareness of packaging. This
awareness increases with publicity of tampering inci-
dents, and it decreases with time until the next publicized
incident.
COST OF CLAIM OF PRODUCT TAMPERING
The cost of responding to a claim of possible product
tampering would boggle the mind of the average person.
The cost begins when the complaint is received by com-
pany personnel, and it includes the following:
. Meetings that Will Be Held to Determine How to
Respond to the Claim. A complaint of possible pro-
duct tampering requires participation of top manage-
ment in all phases of meeting the problem.
. Expenses Related to High-Level Managers Visiting
the Locale Affected. Top management or their repre-
sentative will usually visit the location involved to
get a better feel for the situation.
. Cost of Picking Up All Stock and Possible Restocking
of Retail Displays. .
If the brand is to remain viable,
fresh
supplies must replace
all product that may
have been vulnerable to violation.
. Cost of Complete Inspection of Targeted Stocks or
Destruction of Those Not Capable of Inspection.
Since it is unlikely that the entire supply of product
on the shelves at the time of the incident was
affected, inspection of the stock may prevent destruc-
tion of the entire inventory.
. Lost Sales When Consumers Changed to Other Pro-
ducts. Many consumers will switch to other brands
until they regain confidence in the brand affected.
1190 TAMPER-EVIDENT PACKAGING
. Rewards for Information on Incident. Rewards
usually lead to information as to the identity of the
violator and result in ending further incidents.
. Fees for Packaging Consultants. Since it is impossi-
ble for every company to have employees who are
experts on every contingency, outside consultants
will be required to assist in meeting the challenge.
. Cost of Legal Counsel to Respond to Litigation. Self-
explanatory.
. Damage Awards Where Determined in Court. See
above.
The list can go on. Many of these costs will be incurred
even in false claims of possible product tampering.
When compared to the potential expense for defending
a single claim of tampering, the cost of effective tamper-
evident packaging becomes insignificant. Some in the
industry will take the moral position that we have an
obligation to protect the consumer and they are right.
Beyond the moral position is the reality that many firms
simply cannot afford the cost of responding to product
tampering claims, especially if the firm is a small, with a
very limited product line where the reputation of the
entire product line can be affected by adverse publicity
on one item in the product line. Liability insurance cannot
restore lost-customer confidence.
A recent incident, later determined to be suicide, al-
most caused a household name brand to go out of business
because of the high cost of produced recall and package
redesign, even though the product and company were
linked to the incident only by circumstance.
In considering cost, the cost associated with responding to
one claim of product tampering far exceeds the cost of
incorporating tamper-evident features in the package de-
sign. Would you want to explain to your boss how your
companycouldnotaffordthepennyortwoformakingeach
package tamper-evident, and how you can afford the 10
million dollars or more your company is spending on defend-
ing a claim of possible product tampering? Who can put a
value on human life that would justify not using effective
tamper-evident packaging? The reality is, tamper-evident
packaging should be utilized if it will provide any protection
for your product, regardless of government regulation.
SELECTING WHICH FEATURE TO USE
Everyone has a preference for one type of package over
another. This preference has been developed by either the
products we manufacture or our experience with various
types of packaging in our everyday living. Selecting which
feature to use should not be affected by these preferences, but
by objective testing during the package development stage.
During the design stage, the package engineer should
consider the function of the product and how the consumer
intends to use it. Next, each TE feature that is usable on
the package should be tested to determine which feature
will offer the greatest protection to the consumer. The test
used should be objective, consistent, and replicable. Re-
cords of the test results should be retained indefinitely. If a
feature selected for use achieves a lower value than others
that were rejected, reasons for the selection should be
recorded and retained with the test results. Remember,
cost cannot be a factor in selecting which feature to use.
You would not want to be questioned by attorneys for the
other side as to why you were willing to accept less
effectiveness to save a few pennies and to compare those
few cents to the value of injury to a consumer.
One form of testing the effectiveness of tamper-evident
packaging is the Rosette Protocol, which measures the
degree of difficulty in violating a specific package and
restoring it to a nearly original condition. The Protocol
also measures increases in effectiveness through the use of
multiple features. The value for a specific combination of
features is not equal to the sum of each feature. Some
factors cover the combination, rather than each feature
separately. For example, the knowledge factor is applied
once, regardless of the number of features in the combined
package, and only one knowledge level was required. Time
is cumulative; if it takes 20 min to violate each feature, the
time required is not the value for 20 min multiplied by the
number of features used on the package. In this example
the time factor is the value for 1 h. Only one category of
equipment may be required if all tools or equipment
required to violate the different features in the combination
are in the same class. The feature visibility values for all
used on multiple feature packages are multiplied; even the
use of multiple features that are not shelf-visible increases
the effectiveness of the package. The feature material is
added for each feature replaced or reused to determine the
feature material value. The value of the feature, used with
the specific package components, on the specific product
and form of product tested, is the sum of all the factors.
The FDA and the Non-prescription Drug Manufac-
turers Association have expressed concern that the in-
dustry would gravitate to the feature achieving the
highest score in any testing procedure. In reality,
the value for a specific feature will vary, depending on
the exact product and all other packaging components
used in a specific package. The same feature from different
manufacturers may achieve different values, because
there
are slight variations
in design and manufacture,
even though the features may appear to be identical. If a
value were to be established at 20 on a scale of 0–50 for a
single feature, the value of 20 could be attained through
the use of multiple features if necessary. This would
preclude any single feature from becoming the industry
mandated method to provide protection. The above value
of 20 is for illustration purposes only; prior testing has
shown the minimum design for FDA acceptance to be
around 11, with some minimum values for approved
features scoring much higher. A standard requiring 20
as the minimum value would require most packages to be
improved before the package would meet the standard.
The use of multiple features can result in a value higher
than 50. While the Rosette Protocol has been tested and in
use for several years, there are others that may be as
objective and in use by other companies.
Certain tamper-evident features in use today, and
approved for use by the FDA, are at best window dressing.
Such features can be violated without the use of tools of
TAMPER-EVIDENT PACKAGING 1191
any type, the feature removed and replaced after violation
of the package, and all without leaving any indication of
possible violation. Some very effective alternatives to the
features are not being used because of a slightly higher
per unit cost. Other features used to provide resistance to
violation are being used in a form that does not meet the
FDA standards and can be duplicated very easily—all to
save a penny per unit. It is time for the packaging
industry—the producers and the users—to accept their
responsibility and provide effective packaging to their
customers. The best way to do this is through the use of
an objective approval process that disregards minor costs
or inconveniences.
WHAT IS THE BEST TE FEATURE?
A lot of people ask ‘‘What is the best TE feature?’’ No
single TE feature is best for all products! There are
variations in effectiveness of similar features from differ-
ent manufacturers as well as variations in effectiveness
where the product contributes to the effectiveness. An
example is a metal can which is much more effective for a
carbonated product than for a noncarbonated product. The
product can determine which feature provides the most
protection; for instance, a product that can be adulterated
effectively by penetration would require a more rigid outer
container than one that degrades visibly on violation by
penetration. The best feature for your product is the one
that provides the greatest resistance to violation for that
product in its current form and size. All features can be
violated in some manner; effective TE features provide
greater difficulty in violating the product than do ineffec-
tive features. In some cases the packaging was violated by
being opened, just as any consumer would do, and then
the tamperer replaced the original product with a toxic
substance, not making any attempt to restore the package
to its original appearance. The package worked as in-
tended—it showed that it had been opened, but still
resulted in injury to the consumer. The violation does
not have to be exotic to harm the consumer. Tamperproof-
ness does not exist!
DOES TAMPER-EVIDENT PACKAGING WORK?
Since the implementation of the FDA rule (21 CFR
211.132) in 1982, the consumer has increased awareness
of packaging. This awareness has led to an increase in the
number of complaints of possible product tampering,
although most incidents are later dismissed as unfounded.
Tamper-evident packaging prevents tasting in stores,
prevents in-store violation, and, if the feature is intact,
ensures the consumer that the product is safe. Effective
tamper-evident packaging acts as a deterrent to most
persons who would commit such acts of violation and
makes it difficult for others to violate the package and
restore it to its original appearance. Yes, effective tamper-
evident packaging works, provided that consumers are
aware of the feature and pay attention to what they are
about to use.
Most experts agree that the consumer should be more
aware of what to look for in tamper-evident packaging.
Educating the consumer could include pictures of the
feature and product on the label and in media ads. One
recent case resulted in loss of life where the package met
this test with two indicators: The product was pictured on
the label and the actual product consumed did not resem-
ble the picture on the label in two different ways, as well
as numerous other indications of violation. The consumer
used the product anyway and died from consuming the
substituted item. If the consumer had examined the
product and package, it should have been obvious that
the product was not to be used. Better consumer aware-
ness may have prevented this incident.
The consumer today is different from those in previous
generations. You may remember as a child that if a can was
dented, it would be passed over by shoppers until it was the
last package on the shelf. Most dented cans were sold at a
discount in a special bin. Today consumers are so confident
of the quality of the product, and accustomed to manufac-
turing defects, transporation damage, and other variations
in the package, that they accept without question packages
their parents and grandparents would have rejected. This
demonstrates the confidence the consumers have in our
products, which we can be proud of, but insulates them
from taking responsibility for their own safety.
Both the products themselves and their manufacturers
have taken the entire blame for past incidents of product
tampering. Another weak link in the chain of package
security is the retailer. In prior cases, the retailer has not
been held accountable for the violation of any package, or
the sale of packages with obvious indication of prior
opening. During research, many stores were visited to
observe what products are converting to the use of tam-
per-evident packaging and to determine which features
are used on what types of products. During those visits,
many packages were observed where the tamper-evident
feature indicated possible prior opening. In every case the
package was taken to the store manager, with an explana-
tion of what was observed. Their statements have ranged
from ‘‘I will put it aside for the salesman to pick up’’ to
‘‘there’s nothing wrong with it, it happens all the time’’ to
‘‘put it back on the shelf and I will have the company pick
it up.’’ What if the package had been violated and a
consumer died from using the product? How does the
manager know that another consumer will not buy the
package before the company picks up the product? Retai-
lers need to participate more in making tamper-evident
packaging work as intended, rather than being a weak
link in the chain of package security.
PRODUCT TAMPERING AS AN INTERNATIONAL
TERRORISTS TOOL
The compalcency of the world was shattered on 9/11.
When the World Trade Center collapsed, so did the feel-
ings of sereness and security of most Americans. On 9/11
all potential acts of terrorism— biological and others—
became real-world and increased our awareness of
our
surroundings and actions
by others that may indicate
1192 TAMPER-EVIDENT PACKAGING
a potential threat to our safety and wellbeing. The
risk of retail product tampering being used by interna-
tional or domestic terrorists has existed for decades, and
9/11 did not change the potential threat for its use
as a terroristic tool. The author proposed a means for
identifying potential large—scale product tamperers to
Federal law enforcement agencies in 1991. The laws at the
time precluded their ability to implement such a domestic
threat identification program. Perhaps someday we will be
able to take a proactive position and reduce the potential
use of product tampering as a terroristic tool.
MAKING TAMPER-EVIDENT PACKAGING WORK
Making tamper-evident packaging work as it should
requires the efforts of all involved: The manufacturers of
packaging components should promote effective packa-
ging for use in providing protection, product manufac-
turers should use the most effective feature for their
product, retailers should be aware of the potential for
violated products being in their display, the consumer
should maintain an awareness of what to look for in a
secure package and refuse to buy packages that look
suspicious, law-enforcement agencies should conduct pro-
fessional investigations and prosecute all violations, gov-
ernment agencies should provide for legal relief to
companies meeting or exceeding government standards,
and the media should report only those incidents that are
verified as representing a threat to the health and welfare
of the public without causing hysteria through false
alarms. It all begins with you as packaging professionals.
Product tampering will not disappear. We as an indus-
try must remain committed to providing the consumer
with quality products in packages that provide protection
to the consumer. Since 1982, we have come a long way, but
we must continue to research and develop improvements
in packaging to ensure the availability of safe products for
the next generation.
BIBLIOGRAPHY
General References
Books
J. L. Rosette, Development of an Index for Rating the Effectiveness
of Tamper-Evident Packaging Features, Master’s thesis, Cali-
fornia Coast University, Santa Ana, CA, 1985.
J. L. Rosette, Improving Tamper-Evident Packaging, Technomic
Publishing, Lancaster, PA, 1992.
J. L. Rosette, Product Tampering Detection, Forensic Packaging
Concepts, Inc., Atlanta, 1993.
Walter Stern, Packaging Forensics; Package Failure in the Courts,
Lawyers & Judges Publishing Co. 2000.
Publications
Code of Federal Regulations, Chapter 21, Government Printing
Office, Washington, DC.
Federal Register.
Food & Drug Packaging, published monthly.
Articles and Technical Papers
D. Lowe, ‘‘Crisis Management Marketing, Tampering Strategies
for International and Domestic Marketplace Terrorism,’’ San
Francisco State Univ. J. 1 (1990).
J. L. Rosette, ‘‘Defending Against Terroristic Tampering,’’ 1991.
Incident Reports, 1986 to 2006, reports of claims of possible
product tampering from various law-enforcement agencies.
J. L. Rosette, ‘‘Product Tampering as It Affects Consumers and
Law Enforcement,’’ 1992 FBI Law Enforcement Bull. (Septem-
ber 1991).
Product Tampering in the Marketplace’’ Foundation for American
Communications, 1986.
Product Tampering Problems, Foundation for American Commu-
nications, 1993.
J. Sneden, H. Lockhart, and M. Richmond, Tamper-Resistant
Packaging, Michigan State University, 1983.
Trends in Product Tampering, 1990, FDA & FBI data on product
tampering.
TAPE, GUMMED
R. W. MC KELLAR
The Gummed Industries
Association
INTRODUCTION
Gummed paper tape and reinforced gummed paper tape
have always been very effective carton sealing materials.
Technological advancements in paper, laminates, adhe-
sives, reinforcements, and dispensing and application
equipment provide today’s user with a scientifically pro-
duced high-quality product. Gummed tapes are environ-
mentally friendly because they are made from natural raw
materials and are 100% recyclable. Depending on the
packager’s need, gummed tape can be obtained with a
wide range of tailor-made special features. Advantages of
using gummed paper tape include the following: (a) It is
tamper-proof, that is, the tape cannot be removed from a
carton without leaving traces; (b) water-activated adhe-
sives in gummed paper tape penetrate right into the liner
of the carton, creating a total bond; and (c) extreme
temperatures do not change the characteristics of
gummed tape. The closure is resistant to aging (1).
TYPES
There are two basic forms of gummed tapes: single-ply
nonreinforced ‘‘paper’’ sealing tape and double-ply fiber-
glass ‘‘reinforced’’ sealing tape. Both varieties begin with
what is commercially known as ‘‘gumming kraft,’’ which
differs from ordinary kraft paper in that it is sized to
prevent the adhesive from penetrating too deeply into the
paper. Following application of a vegetable-based remois-
tenable adhesive (see Adhesives), single-ply paper-sealing
tape is slit into roll lengths of 375–6000 ft (114–1829 m).
TAPE, GUMMED 1193
Depending on the basis weight of the paper (35#, 60#, or
90#) (see Paper), the product is categorized for light-,
medium-, and heavy-duty application.
PRODUCTION
Double-ply reinforced gummed sealing tapes are produced
by sandwiching fiberglass yarns between two sheets of
kraft paper with either a hot melt of water-based adhe-
sive. The use of recycled pulp in the paper is encouraged
(2). Fiberglass yarns generally run in three directions
(machine direction and both transverse directions), form-
ing a diamond or ‘‘three-way’’ pattern. As in single-ply
paper tapes, a water-remoistenable adhesive of vegetable
and/or animal glue formulation is applied to the bottom
sheet before or after lamination. Depending on the user’s
needs, paper basis weight, yarn spacing and denier, and
laminate- and adhesive-coating, weight can be varied to
produce products for light-, medium-, and heavy-duty
application. Finally, the parent or ‘‘jumbo’’ roll is slit into
smaller rolls ranging in size from 360 to 4500 ft(110–
1372 m).
Both paper and reinforced sealing tapes are usually
wound gummed-side-in on a cardboard core. Most manu-
facturers also supply coreless rolls. Tape widths generally
range from 1 to 3 in. (25 to 76 mm) in 1/2-in. (13-mm)
increments. Gummed tapes are available in a range of
colors, widths, and lengths. They can also be custom-printed
and can be obtained in strippable grades or with special
antitheft, tamper-evident, or inventory-control features.
USES
When properly purchased and applied, both paper and
reinforced sealing tape fully meet the requirements and
specifications for rail (UCC Rule 41), truck (NCB Rule
222), plane, parcel post, UPS, and other parcel delivery-
service shipments. General Services Administration Com-
mercial Descriptions (CIDs) A-A-1492A, A-A-1671A, and
A-A-1672A govern the purchase of gummed tapes by the
federal government.
Gummed tape sticks well to a wide range of products, but
will not adhere to plastic products . T apes are strong and can
be used for very heavy cartons . They are odorless, so they
can be used in the food industry. They are hygienic, which is
ideal for pharmaceutical products. The tapes will hold firm
and do not stretch, making them ideal for bar-code read-
ability. Gummed paper tapes’ strength and tamper -evident
qualities are important for packaging high value products ,
such as alcohol and jewelry. The tape’s printability adds to
safety by enabling batch and transport information to be
printed onto the tape at the very moment of closing the box.
Last-minute printability allows for savings because fewer
preprinted boxes are needed (1).
Gummed tape dispensing and application equipment
fall within several categories: (1) hand-operated dispen-
sers; (2) electrically operated dispensers; (3) automatic
taping machinery for fixed-size boxes, and (4) automatic
taping machinery for random-size boxes.
BIBLIOGRAPHY
1. R. W. McKellar, ‘‘Taped, Gummed’’ in M. Bakker, ed., The Wiley
Encyclopedia of Packaging Technology, 1st edition, John Wiley
& Sons, New York, 1986, pp. 631–632; 2nd ed., A. J. Brody and
K. S. Marsh, eds., 1997, p. 883.
Cited Publications
1. ‘‘Why Natural Paper Tape,’’ FIPAGO, Natural Paper Tapes,
http://www.fipago.org, accessed October 2008.
2. ASTM D5749, Standard Specification for Reinforced and Plain
Gummed Tape for Sealing and Securing, ASTM, http://
www.astm.org, 2006/Standards.
TAPE, PRESSURE SENSITIVE
R. L. SHEEHAN
3M Corporation,
Minnesota
Updated by Staff
INTRODUCTION
Pressure-sensitive tape (PSA) can be defined as a contin-
uous flexible strip of cloth, paper, metal, plastic, or foam
coated on one side or both with a permanently tacky
adhesive at room temperature which will adhere to a
variety of surfaces with light pressure (finger pressure)
with no phase change and usually available on a roll (1).
PSA is used for closing boxes, combining packages, attach-
ing packaging lists, color coding, pallet unitizing, adding
carrying handles, splicing, providing ease of package
opening, protecting labels, reinforcing critical package
components, holding documents, and a variety of other
jobs.
The first pressure-sensitive tape was developed in 1925
for paint masking; it had a paper backing and a glue–
glycerol adhesive. Today, there are hundreds of specialty
tapes available for specific applications in packaging.
TAPE TESTING
As with all package components, tape testing must be
focused on the function of that component on the package.
An engineering analysis of the package followed by pack-
age testing are excellent beginnings. The critical physical
properties of the tape can then be chosen, in conjunction
with technical input from a reputable tape supplier (2).
One of the most important tape properties is often the
strength of the backing. ASTM D3759 is the standard
laboratory method for measuring the tensile strength and
elongation of pressure-sensitive tapes. Some packages re-
quire a tape with very little stretch; others require a
relatively high elongation to achieve the desired breaking
energy. For many narrow tapes, only the machine-direction
1194 TAPE, PRESSURE SENSITIVE
strength is of importance. For wider tapes, however, the
cross-direction strength can be more critical.
Peel adhesion, ASTM D3330, is perhaps the most often
quoted adhesive measurement. It measures the force in
ounces per inch of width (N/100 mm) required to peel a
strip of tape back onto itself (1801) from a stainless-steel
surface. Test conditions and rubdown are closely con-
trolled. The relationship of this test to tape functioning
correctly on a package is often questionable; the angle of
pull for the test is not seen on most packages, and the
stainless-steel substrate does not represent package sur-
faces well. However, for a given adhesive type, the test
may be useful as a quality-assurance procedure.
A packaging tape adhesive must have good tack to
allow it to adhere to a variety of package surfaces with
only a light rubdown. The literature on pressure-sensitive
tape includes several methods of measuring the initial
tack or ‘‘wet grab’’ of an adhesive. These can be valuable
research test methods, but, because most of them use glass
or steel as a test surface, it can be difficult to correlate tack
test results with the performance of tape on a package.
Tapes are often used on packages where the tape must
hold in shear: The Force on the tape acts in parallel with
the package surface. One test that can relate to this is
ASTM D3654-A, holding power of pressure-sensitive tapes
to fiber-board. A
1
2
-in:
1
2
-in: area of tape is applied to a
fiberboard surface (NIST Standard Reference Material). A
1-kg mass is attached to an end of the tape. The test
measures the time it takes for the mass to pull the tape
from the surface.
ASTM test methods and other standards relating to
pressure-sensitive tape are listed in Table 1. Other test
methods are published by the Pressure Sensitive Tape
Council (PSTC). Tape test methods are workable in both
the inch-pound system of units and the SI metric system;
physical properties are convertible from one system to the
other. The standard widths of tape are a little different in
the two systems. The metric replacement widths are based
on uniform slitting increments, resulting in standard
widths shown in Table 2; these are based on PSTC-71,
Guide for Width and Length of Pressure Sensitive Tape.
Most packaging uses of pressure-sensitive tape will read-
ily allow these metric sizes.
PRESSURE-SENSITIVE ADHESIVES
Pressure-sensitive adhesives are unusual materials because
they are are somewhere between the viscous and rubber
states at room temperature. They show sufficient liquid-like
behavior to deform or flow into contact with a smooth
surface under light contact. Yet they must show appreciable
resistance to flow duing a separation or debonding process .
The main advantage of pressure-sensitive adhesives com-
pared with other types of adhesives is convenience of use.
There is no storage, activation, or waiting involved. Often
the bond is reversible. The disadvantages of pressure-
Table 1. ASTM Standards Relating to Pressure-Sensitive Tape
ASTM Title
D1974 Practice for Methods of Closing, Sealing and Reinforcing Fiberboard Boxes
D2860 Adherence of Pressure Sensitive Tape to Fiberboard at 901 Angle and Constant Stress
D2979 Pressure Sensitive Tack of Adhesives Using an Inverted Probe Machine
D3121 Tack of Pressure Sensitive Adhesives by Rolling Ball
D3330 Peel Adhesion of Pressure Sensitive Tapes a 1801 Angle
D3611 Accelerated Aging of Pressure Sensitive Tapes
D3652 Thickness of Pressure Sensitive Tapes
D3654 Holding Power of Pressure Sensitive Tapes
D3759 Tensile Strength and Elongation of Pressure Sensitive Tapes
D3662 Bursting Strength of Pressure Sensitive Tapes
D3715 Quality Assurance of Pressure Sensitive Tapes
D3811 Unwind Force of Pressure Sensitive Tapes
D3813 Curling and Twisting on Unwinding of Pressure Sensitive Tapes
D3815 Accelerated Aging of Pressure Sensitive Tapes by Carbon Arc Exposure Apparatus
D3816 Water Penetration of Pressure Sensitive Tapes
D3833 Water Vapor Transmission of Pressure Sensitive Tapes
D3889 Adherence to Linerboard of Pressure Sensitive Tape at Low Temperature
D5105 Performing Accelerated Outdoor Weathering of Pressure Sensitive Tapes Using Natural Sunlight
D5264 Abrasion Resistance of Printed Materials by the Sutherland Rub Tester
D5330 Specification for Pressure Sensitive Tape for Packaging, Filament Reinforced
D5375 Liner Removal at High Speeds from PSA Label Stock
D5486 Specification for Pressure Sensitive Tape for Packaging, Box Closure and Sealing
D5570 Water Resistance of Tapes and Adhesives Used as a Box Closure
Table 2. Common Widths of Pressure-Sensitive Tape
Inch-Pound System (in.) SI Metric System (mm)
1
2
12
3
4
18
124
248
372
496
TAPE, PRESSURE SENSITIVE 1195
sensitive adhesives are as follows: The adhesive strength is
low, and they are unsuitable for rough surfaces. They are
also expensive in terms of cost per unit bond area. The most
well-known type of adhesive consists of natural rubber
blended with a tackifier resins and a small amount of
antioxidant. Rigid PVC and polypropylene are used
exrtensively as backing materials for general-purpose tapes.
Plastic films are used in specialized products. Polyacrylates
are generally used in high-quality tapes for their better
technical properties . The most recent development in pres-
sure-tape manufacture is the hot-melt coating process. Al-
most all of melt adhesives are based on SIS thermoplastic
rubber, mixed with hydrocarbon tackifier resins and
oils in substantial amounts and an antioxidant in minor
amounts (3).
BOX-SEALING TAPE
The largest use of pressure-sensitive tape in packaging is
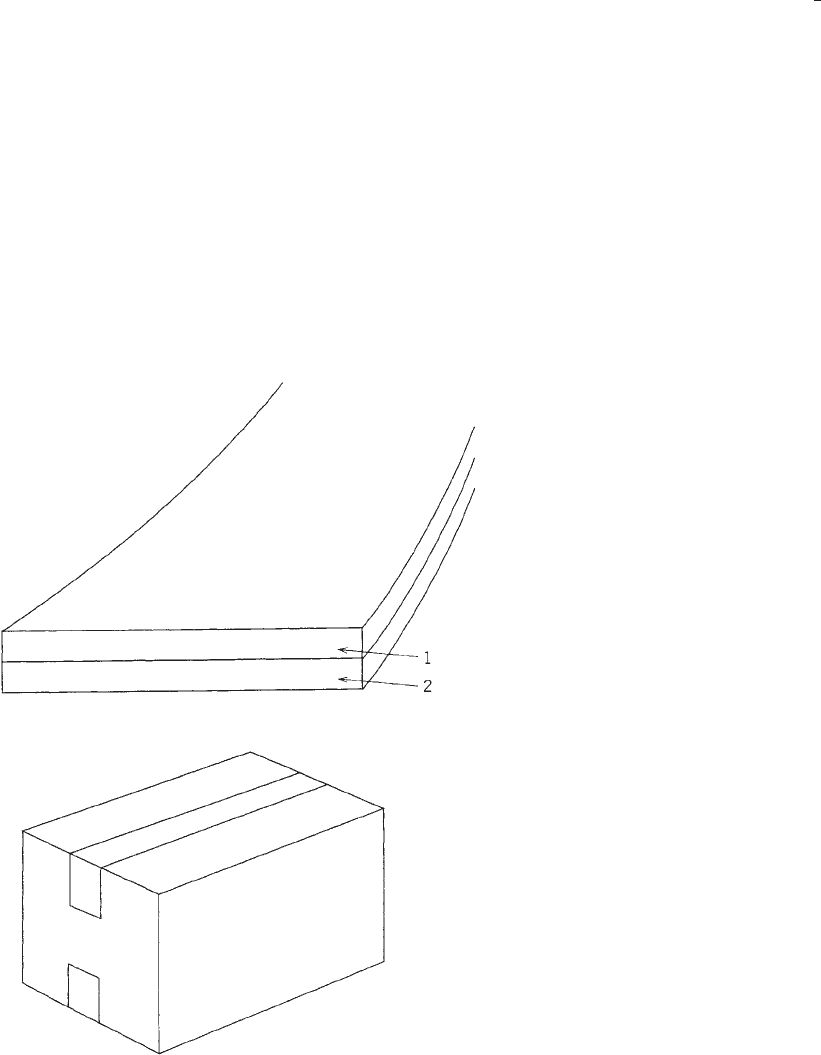
the closure of regular slotted containers. Figure 1 depicts a
typical construction of a box-sealing tape; a plastic film is
coated on one side with a pressure-sensitive adhesive. The
film may have a release treatment on one side to allow
easy removal of the tape from the roll during dispensing.
Some film backings also are treated or coated on the
adhesive side to increase the bond of the adhesive to the
backing; although for a box sealing tape, this is not a
critical factor.
The standard application of a box sealing tape is
described in ASTM D1974, Standard Practice for Closing
and Sealing Fiber Boxes. A 2-in. (48-mm)-wide tape is
applied over the center seams of a regular slotted con-
tainer (RSC) and extends about 2
1
2
in. (65 mm) onto the
end panels of the box. This seals the center seam and helps
keep dust and dirt out of the box. If a total seal is needed,
cross-strips of tape can be added at the end edges of the
box. This ‘‘six-strip seal’’ or ‘‘H-seal’’ is specified for some
military and export packages but is seldom used for
domestic shipments.
The choice of tape for box closure is very important and
affects the performance of the entire package during
storage and distribution. The most common backing ma-
terial is biaxially oriented polypropylene. Some tape back-
ing is made of a relatively ‘‘square’’ film with the machine-
direction and cross-direction properties about the same.
Most heavier-duty tapes are made of a tentered film with
the cross-direction strength higher than the machine-
direction strength; this usually results in better perfor-
mance on boxes. Polyester, unplasticized PVC, and satu-
rated papers are also used as backings.
A proper backing for a box-sealing tape is a good start,
but the tape must have an aggressive adhesive if the
backing strength is to be realized. Many box-sealing tapes
use an adhesive based on rubber and a tackifying resin,
but acrylics and other synthetic adhesive systems are also
used. Critical adhesive properties are tack (for production
efficiency) and shear-holding power (for package ware-
housing and shipping).
Tape is applied by handheld dispensers (Figure 2) or
with box-sealing equipment. A range of equipment is
available for both semiautomatic (operator feeds the boxes
into the box sealer) or fully automatic operation. Box
sealers are either adjustable to boxes of a single size or
random, able to take a mixture of boxes of varying sizes.
Most pressure-sensitive box-sealing tapes hold well in
damp and humid conditions. Water resistance (ASTM
D5570) is required by the United Nations (UN) and the
U.S. Department of Transportation (DOT) for tapes used
on boxes containing hazardous materials.
Standard Specification ASTM D5486, Pressure Sensi-
tive Tape for Packaging, Box Closure, and Sealing, re-
placed Federal Specifications PPP-T-60 and PPP-T-76.
Type I is a polyester-backed tape used in H-type closure.
Type II is a polyester-backed tape used in single-strip
closures. Type III is a polypropylene-backed tape used in
single-strip closures. Type IV is a cloth-backed tape, and
Type V is a paper-backed tape.
FILAMENT TAPES
A second broad category of packaging tape is pressure-
sensitive filament tape, sometimes known as ‘‘strap-
ping tape.’’ Figure 3 shows that this is typically made of
Figure 1. A typical box-sealing tape consists of a backing film (1)
and a layer of pressure-sensitive adhesive (2). It is used most
often as a closure for regular slotted containers.
1196 TAPE, PRESSURE SENSITIVE
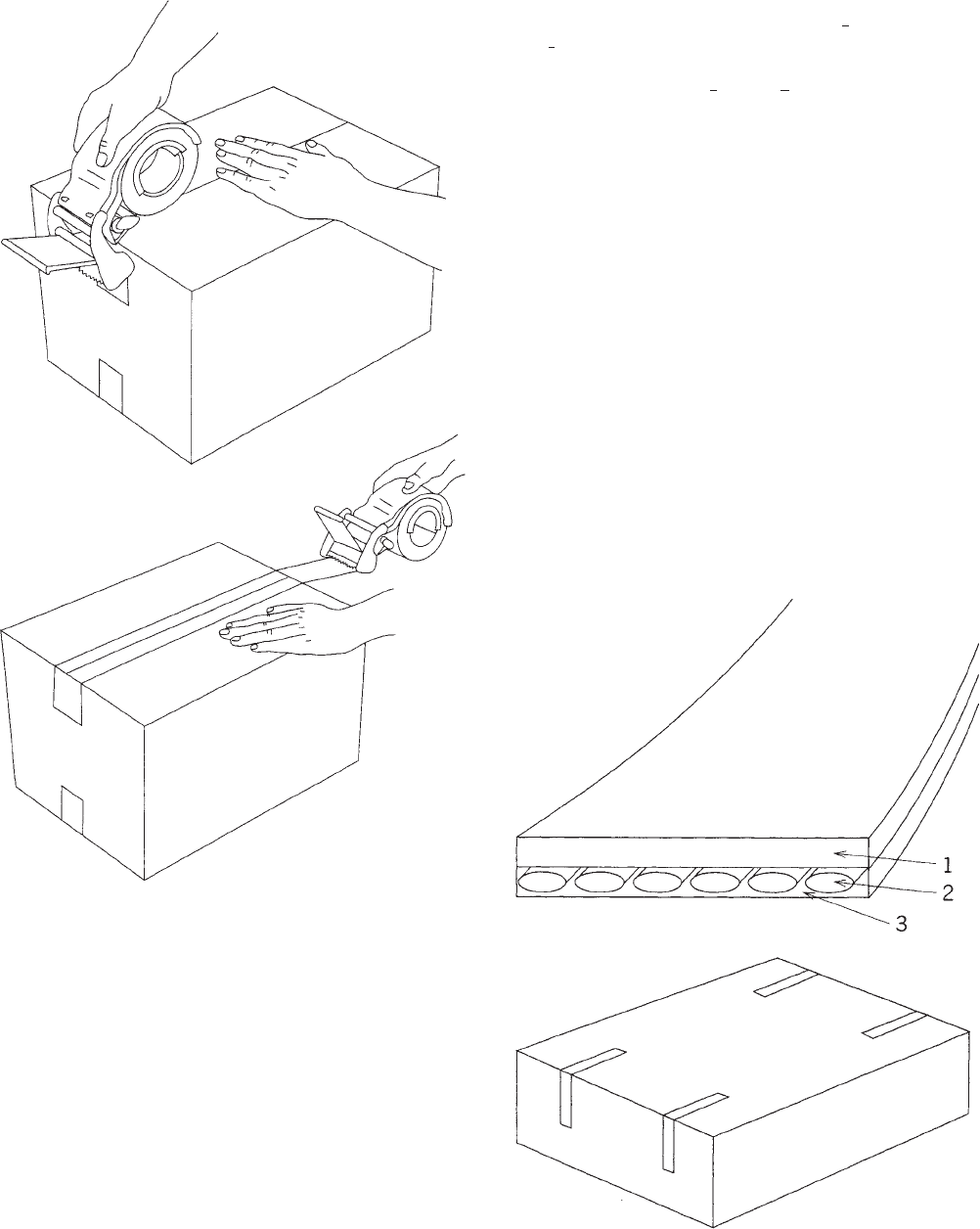
a film backing (polyester or polypropylene) with reinfor-
cing filaments embedded in the pressure-sensitive adhe-
sive. The most common filament is fiberglass, which
provides a high tensile strength with very little elonga-
tion. A few tapes have polyester or rayon filaments for
extra impact or cut resistance. Tapes are also available
with integral polymeric filaments. The adhesive require-
ments for filament tapes are at least as critical as those of
box-sealing tapes. Care should be taken to choose a tape
with a balance of tack (initial adhesion) and shear-holding
power.
Box closure can be accomplished with filament tape on
a variety of box styles. In ASTM D1974, filament tape is
recommended for use with boxes with fully overlap-
ping flaps such as a five-panel folder, full-telescope box,
or full-overlap box. A high-performance filament tape may
be applied in L-shaped strips as small as
1
2
in (12 mm) wide
by 4
1
4
in. (105 mm): two in (50 mm) on each side of the L-
clip. A tape with lower tensile and adhesive properties
may have to be used in
3
4
- in: 6
1
4
- in: (18-mm 155-mm)
L-clips for equivalent performance on a box.
Filament tape is used in dozens of other packaging
applications where high strength is required. These include
reinforcing boxes, combining boxes for shipment, bundling,
recooperage, and pallet unitizing. Special filament tapes
are available for use directly on appliance surfaces to hold
doors and drawers in place during shipment.
Standard Specification ASTM D5330, Pressure Sensi-
tive Tape for Packaging, Filament Reinforced, replaced
Federal Specification PPP-T-97. It includes four classes of
filament tape: Type I—cut-resistant, Type II—medium-
tensile-strength, Type III—high-tensile-strength, and
Type IV—high-tensile-strength, weather-resistant. Other
grades of filament tape are also available for industry,
institution, and home use.
SPECIALTY TAPES
With a full choice of films, papers, and foils for backings,
along with a wide range of adhesives available, it has been
Figure 2. Use of a handheld dispenser for applying box-sealing
tape to close RSC.
Figure 3. A typical filament tape has a backing film (1) and
filaments (2) embedded in a pressure-sensitive adhesive (3). One
of the uses is the L-clip closure of full-overlap boxes.
TAPE, PRESSURE SENSITIVE 1197
