Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

heat-resistant organism is used to determine the correct
process time. Often these processes are described in terms
of F values.
F Value. An F value is the time in minutes to heat-
inactivate a given number of certain micro-organism at a
fixed temperature (4). If the temperature is 2501F (1211C)
and the organism is Cl. botulinum, then the F value is
called F
o
. This value is the number of minutes required to
kill a given population of Cl. botulinum spores at 2501F
(1211C). Combinations of times and temperatures other
than 2501F and F
o
minutes can inactivate the same
number of spores; temperatures lower than 2501F for
longer time periods or temperatures greater than 2501F
for shorter times have an equal ability to inactivate the
spores. An F
o
= 2.45 min reduces the population of Cl.
botulinum spores by a factor of 10
12
(5). In practice, F
o
values of greater than 3 are used as a safety measure. To
prevent overprocessing (overcooking) of the food, the spore
inactivation (called lethality) is summed up during the
time the coldest spot in the can is coming up to the retort
temperature. Some viscous conduction-heated foods may
never completely reach the common retort temperature of
2501F (1211C) yet still receive the proper F
o
treatment.
The Food and Drug Administration (FDA) regulations
require that these tests and calculations be carried out
only by recognized authorities (6).
Interest in thermal processes resulted in techniques
that commercially sterilize fluid foods in continuous-flow
heat-exchange systems before packaging (7). The thermal
death calculations described above still apply to these
processes, and proper F values must be achieved (8). The
continuous-flow commercial sterilization procedures have
the advantage that products can be heated and cooled
more rapidly for shorter times with equal lethality. This
can give a higher quality product. These products must be
filled into presterilized containers (see the Aseptic packa-
ging article).
CANNING OPERATIONS
The canning process requires several unit operations that
normally take place in a set sequence (9).
Product Preparation. As soon as the raw agricultural
product is received at the canning plant, it is washed,
inspected, sorted to remove defective product, and graded.
Often, the edible portion is separated from nonedible as
in the case of peas or corn. Fruits and vegetables are
subjected to a blanching operation by exposing them to
either live steam or hot water at 190–2101F (88–991C).
Blanching serves to inactivate enzymes that would other-
wise cause discoloration or deterioration in the product. It
also softens, cleans, and degases the product. Peeling,
coring, dicing, and/or mixing operations may be carried
out next. These operations prepare the product for filling
into the can.
Container Preparation. Containers must be washed
thoroughly immediately prior to filling. Cans are washed
inverted so that any foreign objects and the excess water
can drain out. The container is now ready for filling.
Accurate and precise filling is necessary to meet minimum
labeled fill requirements yet leave sufficient headspace for
development of the proper vacuum after closure. Too large
a headspace results in an underweight container, whereas
overfilling can result in bulging or domed ends after
processing. Excessive headspace may also suggest that
large amounts of oxygen remain in the can, which accel-
erates product deterioration and can corrosion. Liquid or
semiliquid products including small pieces are filled by
automated equipment. Larger, more fragile products, such
as asparagus, are packed by hand or by semiautomated
equipment. In most products, brine, broth, or oil is added
along with the product. This liquid excludes much of the
air between the particles and provides for more efficient
heat transfer during thermal processing.
Vacuum. Proper application of the closure after filling
is one of the most critical steps in the canning operation.
The two-step seaming operation not only must produce
a sound, well-formed double seam at speeds of several
hundred cans per minute but also must produce an
interior vacuum of 10–20 in. Hg (34–68 kPa) (10). This
vacuum reduces the oxygen content, retards corrosion and
spoilage, leaves the can end in a concave shape during
storage, and prevents permanent distortion during retort-
ing. A proper internal vacuum can be achieved by several
methods. Containers that are sealed while the food is at or
near the boiling point develop a vacuum when the product
cools. This preheating or hot fill also serves to sterilize the
container when high-acid foods are packaged. Products
that are cool when filled can be heated in the container
prior to sealing with the same result as the hot fill. This is
often termed ‘‘thermal exhaust.’’ An internal vacuum may
also be achieved by mechanical means. The filled, un-
sealed container is fed into a vacuum chamber by means of
an air lock, and the closure is sealed while under vacuum.
This system has the disadvantage that flashing of the
liquid may occur if air is entrapped in the food or high
levels of dissolved air are found in the liquid. The most
common method of producing internal vacuum is by
displacing the air in the headspace with live steam prior
to and during double seaming the cover. The steam in the
headspace condenses and forms a vacuum as the container
cools.
Retorting. In conventional canning operations of low-
acid foods, the sealed containers are next thermally
processed at 2501F (1211C) in retorts. Recent regulatory
agency rule changes allow specific flexible containers to be
processed at 2751F (1341C).
Several distinct types of commercially manufactured
retorts are available for thermally processing canned food
(11). Although all, by necessity, operate at pressures above
15 psi (103 kPa), the design characteristics of each type are
considerably different. At least six design variables exist:
(a) discontinuous (batch) types versus continuous con-
tainer processing, (b) the heating medium used to transfer
heat to the container, (c) the agitation or nonagitation
of containers during processing, (d) the layout of the
188 CANNING, FOOD
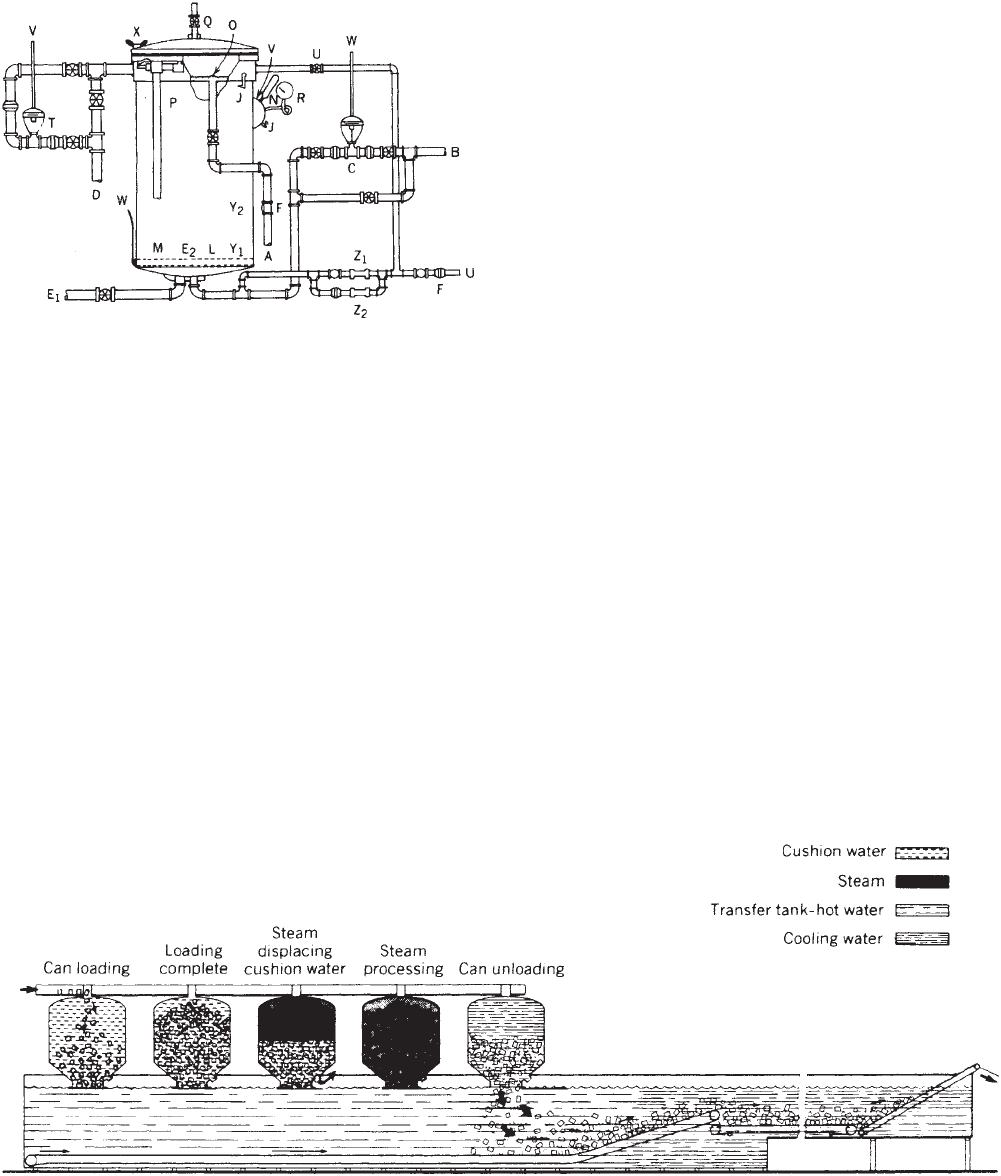
pressure vessel (vertical vs horizontal), (e) the method
used to load and unload the containers from the retort,
and (f) the cooling procedures used after thermal
processing.
A—Water line. O—Water spreader.
B—Steam line. P—Safety valve.
C—Temperature control. Q—Vent valve for steam
processing.
D—Overflow line. R—Pressure gauge.
E
1
—Drain line. S—Inlet air control.
E
2
—Screens. T—Pressure control.
F—Check valves. U—Air line.
G—Line from hot
water storage.
V—To pressure control
instrument.
H—Suction line and manifold. W—To temperature control
instrument.
I—Circulating pump. X—Wing nuts.
J—Petcocks. Y
1
—Crate support.
K—Recirculating line. Y
2
—Crate guides.
L—Steam distributor. Z—Constant flow orifice valve.
M—Temperature-controller
bulb.
Z
1
—Constant flow orifice
valve used during come-up.
N—Thermometer. Z
2
—Constant flow orifice
valve used during cook.
The simplest retorts are batch (discontinous) retorts
that use pure steam as the heating medium and do
not have provisions for mixing (agitation) of the con-
tainer contents during processing. These retorts are
termed still retorts (Figure 1). Temperature inside still
retorts is maintained by automatic control of the steam
pressure.
Loading and unloading the containers from discontin-
uous still retorts are accomplished by preloading contain-
ers into crates, baskets, cars, or trays. ‘‘Crateless’’ systems
randomly drop containers into the retort vessel, which is
filled with water to act as a cushion and prevent container
drainage (Figure 2). The water is drained prior to proces-
sing. The orientation of the retort depends on the type
of container handling system. Systems that use crates
or baskets and the crateless systems, by necessity, use
vertical vessels, whereas car handling necessitates a
horizontal orientation.
Glass, semirigid, and flexible containers must be pro-
cessed in still retorts that have been designed to accom-
modate the fragility of these containers at retort
temperatures and pressures. These retorts operate at
pressures greater than the 15 psi (103 kPa) of steam
required to reach 2501F (1211C) to counterbalance the
internal pressure developed in the container. This is
termed ‘‘processing with overpressure.’’ The pressure
buildup inside individual containers during processing
would result in the loss of seal integrity in heat-sealed
containers (see the Sealing, heat article) and could loosen
the covers of glass containers or permanently distort
semirigid plastic containers. Four design changes in still
retorts must be made to process with overpressure:
1. Either steam or air overpressure must be automati-
cally controlled. Pressures of 25–35 psi (172–241 kPa)
are typical.
2. Control of the retort temperature must be indepen-
dent of retort pressure.
3. Mixed heating media of either steam—air, water–
air, or water–steam are used in place of pure steam.
Heat transfer is less efficient in these mixed-heating
media. For this reason, some means of circulating or
Figure 1. Vertical still retort (21 CFR, Part 113).
Figure 2. FMC crateless retort system. (Courtesy of FMC Corporation, Food Processing Machinery Division.)
CANNING, FOOD 189
mixing the heating medium is necessary. For steam–
air mixtures, fans may be provided. Water–air and
water–steam systems use circulating pumps.
4. Provisions to prevent stress on the containers
caused by motion during processing are made.
These designs may be incorporated into either vertical
or horizontal retorts depending on how the containers are
handled. Glass containers are typically loaded into crates
or baskets for processing in vertical water–air or water–
steam retorts. Flexible retortable pouches are often loaded
into trays and cars (which also serve to maintain the
proper shape of the pouch) and moved into horizontal
retorts for water–steam or steam–air processing (see
Figure 3).
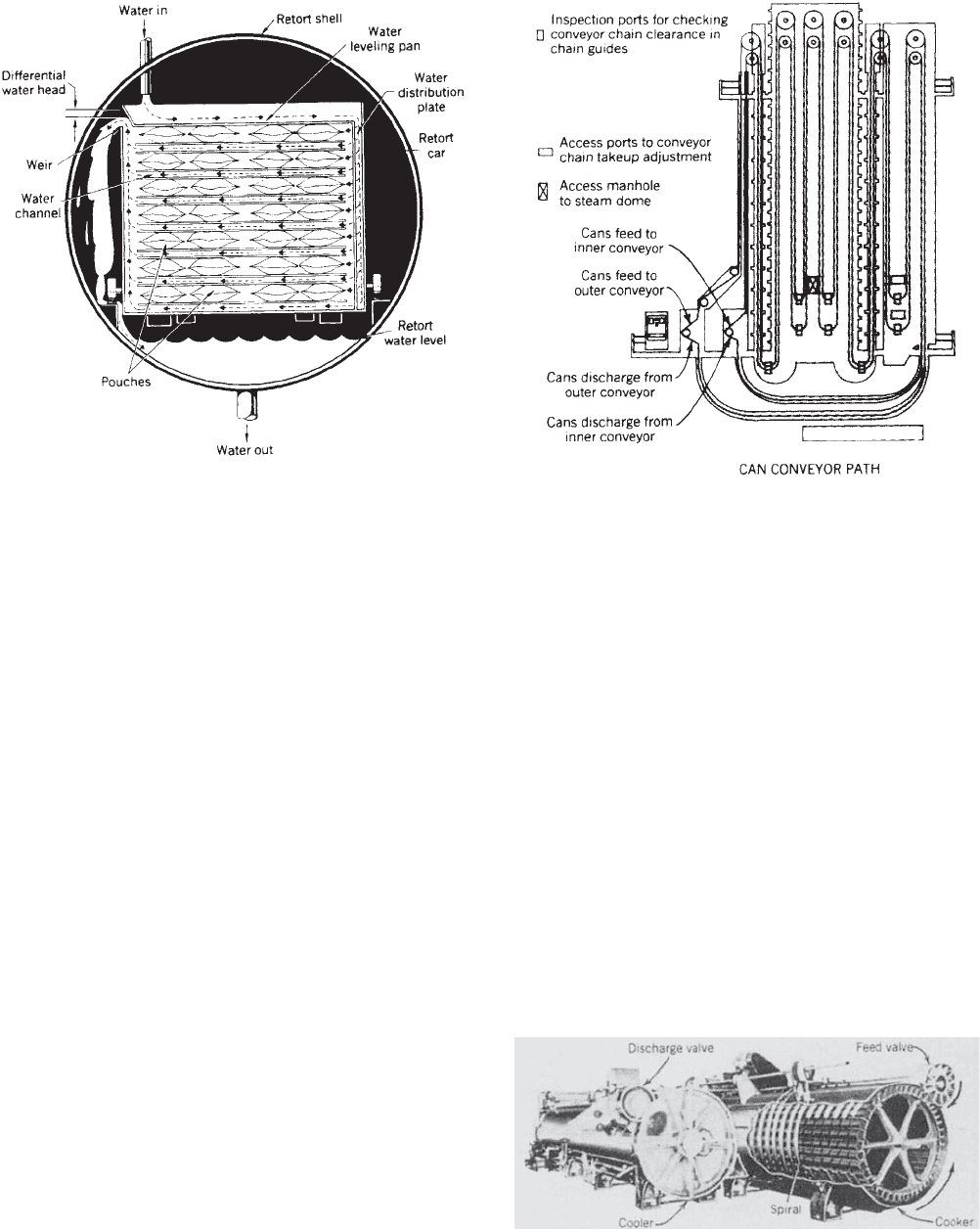
Still retorts, whether designed for metal cans or other
containers, are batch (discontinous) systems. The hydro-
static retort is technically a still retort (product is not
agitated) that continuously processes containers. The
retort operates at a constant temperature (and pressure)
as the containers are carried through the retort by a
continuously moving chain (Figure 4). The required
15 psi (103 kPa) of steam pressure inside the retort (or
steam dome) is maintained by two columns of water,
which also serve as pressure locks for incoming and out-
going containers. These columns of water (called feed and
discharge legs) must be greater than 37 ft (11.3 m) high to
maintain at least a minimum 15-psi (103-kPa) steam
pressure. Hydrostatic retorts have the highest through-
puts; are efficient in their use of floorspace, steam, and
water; can process a variety of container sizes and types
(including flexible); and are highly automated. They have
the disadvantage of high capital costs and are therefore
applicable only to high-volume operations.
The heating time necessary to ensure that the coldest
spot in the container receives the proper lethality depends
somewhat on the consistency of the product. For viscous
products such as canned pumpkin and baked beans, the
primary heat-transfer mechanism is conduction. Products
that have a thin consistency or are packed with brine
(canned peas) are heated by convection. The transfer of
heat to the center of the container can be greatly facili-
tated if internal mixing occurs in the can during retorting.
This results in a shorter processing time and higher
quality product.
Retorts that are designed to increase convection heat-
ing by container motion during processing are termed
agitating retorts. Both end-over-end and axial rotation
are used, but the latter predominates. Glass, semirigid,
and flexible containers are not agitated because of fragi-
lity. Agitating retorts may be either batch or continuous
types. Continuous retorts predominate because they have
Figure 3. FMC convenience foods sterilizer, showing water flow
and pouch restraints. (Courtesy of FMC Corporation, Food Pro-
cessing Machinery Division.
Figure 4. Flow diagram of a hydrostatic sterilizer for canned
foods. (Courtesy of FMC Coporation, Food Processing Machinery
Division.)
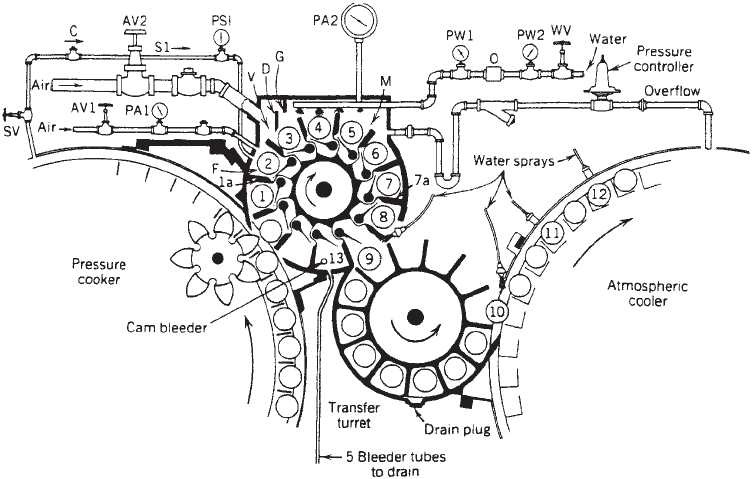
Figure 5. Cutaway view of a continuous rotary cooker–cooler’s
turning wheel and interlock. (Courtesy of FMC Corporation, Food
Processing Machinery Division.)
190 CANNING, FOOD
greater efficiency and throughout but are less easily
adapted to changes in container size.
The continuous rotary cooker–cooler (see Figure 5) has
become widely used for large-volume operations in which
convection-heated products such as vegetables in brine
are packed in metal cans. This system feeds individual
cans into and out of the pressurized vessels by means of
rotary pressure lock valves. Cans are rotated around the
inside of the vessel’s shell by means of an inner rotating
reel and a series of spiral channel guides attached to the
shell (Figure 5). This system provides for intermittent
agitation of the cans by providing rotation about the can
axis during a portion of the reel’s rotation inside the
vessel. This system has the disadvantage that container
size cannot be easily changed.
Regardless of retort design, consideration must be
given to cooling containers after processing. For glass,
flexible, and semirigid containers, cooling with overpres-
sure is necessary. These containers would fail because of
the internal pressure developed during heating should the
external pressure drop. Even metal cans may buckle and
panel if brought to atmospheric pressure while the con-
tents are at 2501F (1211C). In batch-type still retorts,
overriding air pressure with water cooling is used. Hydro-
static retorts (continuous still retorts) cool containers by
removing heat from the water in the discharge leg. If more
cooling is necessary, an additional cooling section is added
and cool water is cascaded over the containers. Contin-
uous agitating (rotary) systems cool under pressure by
transferring containers to pressurized cooling vessels by
means of rotating transfer valves (Figure 6). The second-
stage vessels are maintained at elevated air pressures
while the containers are cooled with water. A third-stage
atmospheric cooler may also be incorporated.
In addition to the pressurized heating and cooling
vessels, all retort systems require a set of precise instru-
ments and controls. Regulations require a direct reading
mercury-in-glass thermometer as well as temperature
Figure 6. Transfer valve between cooking and cooling vessels of a continuous cooker–cooler.
R Rotor containing can pockets and ejector paddles. PW1 Downstream water pressure gauge.
SV Forespace steam valve. O Orifice for flow control.
M Microcooling chamber. Overflow Release line for excess air or water.
F Forespace for pressure equilization. S1 Steam line.
D Water dam. C Check valve.
G Splash guard. PSI Pressure vacuum gauge.
Air Pressure regulated air supply. 1. Hot can.
V Vestibule. 1a. Valve-leading edge.
AV1 Forespace regulated air inlet valve. 2. The can receive some water splash.
AV2 Microcooling space air inlet valve. 3.–7. The cans are fully exposed to water flow.
PAI Forespace air pressure gauge. 7a. Valve-trailing edge.
PA2 Microcooling space air pressure gauge. 8. Can is about to leave microcooler valve.
Water Water inlet and perforated distributor. 9.–12. Cans are subject to sprays of water.
WV Water inlet regulator valve. 13. Drain pocket.
PW2 Upstream water pressure gauge.
(Courtesy of FMC Corporation, Food Processing Machinery Division.)
CANNING, FOOD 191
recording devices. A continuous temperature controller
must be installed. For retorts that use pure steam, this
may be a pressure controller; processing with overpres-
sure requires a direct temperature controller. Retorts
require reliable sources of steam, air, and water. A pres-
sure reading device is required as well as an accurate
recording timing device so that the scheduled process can
be insured and the proper records maintained. All instru-
ments must undergo periodic calibration.
REGULATION
The canning of foods is carefully regulated by the FDA, or
in the case of canned meats and poultry, the United States
Department of Agriculture (USDA). These agencies recog-
nize the serious public health implications of improperly
processed foods. The FDA has developed a complete set of
regulations commonly referred to as the Good Manufac-
turing Practices (GMPs) for canning foods. These regula-
tions govern the type of equipment used to can foods and
the procedures, the frequency of inspection of containers
and equipment, and the records that must be kept, and
they provide for the filing of individual processes prior to
production. The regulations pertaining to food canning are
contained in Title 21 of the Code of Federal Regulations
(CFR) under the following sections: (a) 21 CFR Part 108,
‘‘Emergency Permit Control’’; (b) 21 CFR Part 113, ‘‘Ther-
mally Processed Low-acid Foods Packaged in Hermetically
Sealed Containers’’; and (c) 21 CFR Part 114, ‘‘Acidified
Foods.’’
Part 108 stipulates that food-canning plants must
register their establishments and specific processes with
the FDA. This section also contains provisions for issuing
emergency permits to firms that the FDA believes do not
fully meet the regulations. Part 113 is the most extensive
section and details the equipment, procedures, process
controls, establishment of correct process, critical factors,
and necessary records for canning low-acid foods. This
section also details the procedures to be used in evaluating
the integrity of the double seams. Part 114 describes the
GMP requirements for packaging high acid foods
(pHr4.6). This section includes general provisions as
well as specific requirements for production and process
control.
The USDA’s Food Safety and Inspection Service (FSIS)
has regulatory authority over canning poultry and meat
products and has promulgated a series of regulations
under Title 9 of the CFR. Current FSIS regulations are
considerably more general than FDA regulations and
have not kept pace with changes in canning technology.
Meat and poultry canning operations are subject to con-
tinuous inspection in a manner similar to other FSIS-
regulated plants. These regulations are contained in sec-
tions 318.11 and 381.49, which deal with the cleaning of
empty containers, inspection of filled containers, coding,
use of heat-sensitive indicators (see Indicating devices),
and incubation of processed products. In 1984, FSIS
proposed a more detailed set of regulations similar to
those promulgated by the FDA for canning low-acid foods.
The sections of the CFR that deal, in part, with meat and
poultry canning, and related requirements will be 9 CFR
308, 318, 320, and 381.
TRENDS
The food-canning industry has undergone substantial
changes, most notably in the area of containers (12).
Changes are ongoing in the retort systems. Rotation,
oscillation, gentle motion, and shaka are the various
terms used by vendors to promote their particular system.
The major trends are in the uses of semirigid and flexible
pouches.
The industry is increaingly using the flexible pouch and
semirigid containers, taking advantage of the thinner
profile of these containers that can result in faster heating
and higher quality food. Also the shipping cost of the
empty can and glass jars to the retort facility is becoming
more of an issue.
Metal as well as glass cans will have increasing com-
petition from plastics and composite materials (14).
Development of thermal processes in which the food is
commercially sterilized before packaging is ongoing. This
allows food to be thermally processed in continuous-flow,
heat-exchange systems that can result in higher quality
products and allow the use of less expensive containers
based on paperboard or thin plastics. This technology is
used for juices, drinks, and milk. Recent products include
soups, stews, and vegetables (8).
BIBLIOGRAPHY
J. H. Hotchkiss, ‘‘Canning Food,’’ in The Wiley Encyclopedia of
Packaging Technology, 1st edition, Wiley, New York, 1986, pp.
86–91; 2nd edition, 1997, pp. 123–128.
1. S. R. Friedman in W. C. Simms, ed., The Packaging Encyclo-
pedia—1984, Cahners Publishing, Boston, MA, 29(4), 334
(1985).
2. A. Lopez, A Complete Course in Canning, 11th edition, Book 1,
The Canning Trade, Inc., Baltimore, MD, 1981, p. 9.
3. I. J. Pflug and W. B. Esselen in J. M. Jackson and B. M. Shinn,
eds., Fundamentals of Food Canning Technology, AVI Pub-
lishing, Westport, CT., 1979, pp. 10–94.
4. N. N. Potter, Food Science, 3rd edition, AVI Publishing,
Westport, CT, 1978, pp. 177–193.
5. Ref. 2, p. 330.
6. Establishing Scheduled Processes, Code of Federal Regula-
tions, Title 21, Part 113.83, U.S. Government Printing Office,
1983, Washington, DC, p. 112.
7. D. Wernimont, Food Eng. 55(7), 87 (1983).
8. A. A. Teixeira and J. E. Manson, Food Technol. 37(4), 128
(1983).
9. Ref. 4, pp. 550–557.
10. Ref. 2, pp. 217–219.
11. Canned Foods: Principles of Thermal Process Control, Acid-
ification and Container Closure Evaluation, 4th edition, The
Food Processors Institute, Washington, DC, 1982, p. 162.
12. B. J. McKernan, Food Technol. 37(4), 134 (1983).
192 CANNING, FOOD

13. ‘‘Welded Can Expected to Capture 3-Piece Can Market,’’ Food
Prod. Manage. 104(12), 12 (1982).
14. J. Haggin, Chem. Eng. News. 62(9), 20 (1984).
General References
A. Lopez, A Complete Course in Canning, 11th edition, Books 1
and 2, The Canning Trade, Inc., Baltimore, MD, 1981, pp. 556.
J. M. Jackson and B. M. Shin, Fundamentals of Food Canning
Technology, AVI Publishing, Westport, CT, 1979, pp. 406.
Canned Foods: Principles of Thermal Process Control, Acidifica-
tion and Container Closure Evaluation, 4th edition, The Food
Processors Institute, Washington, DC, p. 246.
CANS, ALUMINUM
TERRE REINGARDT
Ball Corporation, Westminster,
Colorado
N. F . NIEDER
Amheuser-Busch
Updated by Staff
INTRODUCTION
Over 98% of all aluminum cans are drawn-and-ironed
(D & I) cans used for beer and soft drinks (see Cans,
fabrication; Carbonated-beverage packaging). The other
1–2% is accounted for by small shallow-draw food cans. In
the United States, production of aluminum beverage cans
has reached about 100 billion/yr (1). Virtually all beer cans
are aluminum, and 100% of all soft-drink cans are alumi-
num. Until 1965, the three-piece soldered can was the only
can used for beer and beverages (see Cans, fabrication;
Cans, steel). It was generally made of 75-lb per base box
(16.8 kg/m
2
) tinplate (see Tin-mill products). Aluminum
does not have the strength of steel per unit weight and
cannot be soldered. Therefore, early in the development of
the aluminum can, attention was focused on improving
the properties of aluminum and perfecting two-piece D & I
technology. More recently, the emphasis has been on
saving metal, because the cost of metal is the single
largest component of final product cost. One of the chief
reasons for the success of aluminum cans has been their
scrap value for recycling (see Energy utilization; Recy-
cling). This article pertains to the technical developments
that have led to today’s aluminum beverage can.
HISTORICAL DEVELOPMENT
The significant developments took place in the late 1960s.
Earlier, in 1958, Kaiser Aluminum attempted to make a
7-oz (207-mL) aluminum can using a 3003 soft-temper
aluminum of the type used for aluminum-foil production,
but the effort was not a commercial success. In the
universe of aluminum alloys, the 3-series alloys contain
a small percentage of manganese as the principal alloying
element. The success of the aluminum can depended on
the development of the 3004 alloy for the can body, which
contains manganese along with a slightly lower amount of
magnesium. (The softer 5182 alloy for can ends contains a
higher amount of magnesium.) Reynolds Metals Co. began
making production quantities of 12-oz (355-mL) seamless
D & I cans in 1964 using a 3004 alloy. The walls of these
first cans were straight, with a top diameter of ‘‘211’’:
2
11
16
in. (68.26 mm). The starting gauge was 0.0195 in.
(0.495 mm); can weight was 41.5 lb/1000 (18.8 kg/1000).
Reynolds introduced the first necked-in cans (see
Figure 1) in 1966, reducing the top diameter from 211
to 209, that is, 2
9
16
in. (65.09 mm). This represented a
breakthrough in technology and container performance,
particularly as it is related to cracked flanges. Cracked
flanges were a serious problem in both double-reduced
tinplate and straight-walled aluminum cans. The intro-
duction of carbide knives at the slitter essentially elimi-
nated cracked flanges on tinplate cans. Eliminating them
on aluminum cans required a change from die flanging to
spin flanging, and necking-in before flanging, which does
not stretch the metal beyond its elastic limits.
In 1968, a new harder-temper (H19) 3004 alloy was
introduced for aluminum cans. Although the 3004–H19
combination had been available since the 1950s for other
purposes, it was not until 1968, when Alcoa and Reynolds
were in commercial production with full-hard-temper can
sheet, that it could be used to effect significant weight
reductions. Weight/1000 cans decreased from 41.5 lb
(18.8 kg) in the mid-1960s to 34 lb (15.4 kg) in the mid-
1970s and to less than 30 lb (13.6 kg) in the mid-1980s.
Both 3004–H19 and 3014-H19 are used today.
Gauge reductions have increased the point where de-
sign techniques have become critical to sustaining the
can’s ability to hold the product. With few exceptions, U.S.
brewers pasteurize beer in the can. This generates high
internal pressures, and most cans used for beer must be
designed so that they have a minimum bottom buckle
Figure 1. A necked-in can.
CANS, ALUMINUM 193

strength of 85–90 psi (586–620 kPa), depending on the
carbonation level. Brewers were asking for cans with
minimum column strength (i.e., vertical crush) of 300 lbf
(1330 N).
A revolution has taken place in bottom profiles (see
Figure 2). The original D & I bottom had a rather generous
bottom-heel radius. In order to meet the 90-psi (620 kPa)
minimum bottom buckle-strength requirement, a 211 can
with this configuration would have to be made with a
starting gauge of 0.016–0.0165 in. (406–419 mm). The next
profile development, basically the Alcoa B-53 design, is
widely used throughout the beer industry today because
it allows the starting gauge to go as low as 0.013 in.
(330 mm), in most cases without sacrificing the 90-psi (620-
kPa) minimum bottom buckle strength. The Alcoa B-80
profile allows starting-gauge reduction to B0.0126 in.
(B320 mm).
An ‘‘expandable bottom’’ design might permit use of a
0.010-in. (254 mm) starting gauge for pasteurized product.
In contrast to the current dome profiles, an expandable
bottom is essentially a flat bottom with small pods located
near the perimeter to provide stability to the pressurized
can, reduce drag, and increase mobility. A totally flat
bottom would drag too much on the filling line. The
‘‘expandable’’ bottom is designed to flex outward during
pasteurization, relieving some of the generated pressure.
Sidewalls are still being redesigned (see Figure 3). In
the 1970s, the so-called nominal thinwall (i.e., the area of
the can that has been thinned most) generally ranged
from 0.0052 to 0.0053 in. (132–135 mm). More recent
versions of the D & I can have reduced the nominal
sidewall to 0.0045 in. (114 mm). A reduction of this magni-
tude represents substantial cost reduction. It also means a
corresponding reduction in the overall column strength
(vertical crush) of the can, not below the minimum 300 lbf
(1330 N) but in terms of overall operating average. Col-
umn strength is very critical to the brewers, who ship long
distances by truck and rail. Extensive testing by the can
companies and the beer and beverage industry in general
has shown that the 300-lbf (1330-N) minimum is satisfac-
tory. Because of the reduction in body-wall thickness,
dents that were acceptable before have now become
critical owing to their influence on reducing the can’s
column strength. Can makers and brewers are monitoring
their handling systems for empty and full cans to mini-
mize denting wherever possible.
In the 1980s, can suppliers reduced costs further by
double ‘‘necking-in’’ (see Figure 4). These configurations
reduced costs primarily because of the diameter reduction
of the lid. In 1984, further activity with respect to necking-
in began to occur. Cans with three or four die necks are
now being run commercially (see Figure 4). In Japan, a
can with eight necks was tested. Metal Box (UK) intro-
duced a spin-neck can (see Figure 4), which essentially
produced the same 206 top diameter as the triple- or
quadruple-neck can, that is 2
6
16
in. (60.33 mm).
Another advantage of aluminum is the ‘‘split gauge.’’
The industry used to sell coils in 0.0005-in. (12.7-mm)
increments, for example, 0.0130 in. (330 mm), 0.0135 in.
(343 mm), etc. A new pricing structure introduced in 1983
allows can-stock buyers to order gauge stock in 0.0001-in.
(2.54-mm) increments. A can manufacturer can reduce
costs by taking advantage of these slight gauge reduc-
tions. In addition, there has been a change in the gauge
tolerance as rolled by the aluminum mills. In the 1970s,
the order gauge was subject to a 70.0005-in. (12.7 mm)
tolerance; today, it has been reduced to 0.0002 in. (5.1 mm).
This permits further gauge reduction because it allows the
can manufacturer to reduce the order gauge without
changing the minimum bottom buckle strength.
CURRENT DEVELOPMENT
Today 100% of all U.S. soft-drink cans and 95% of all beer
cans are fabricated from aluminum (1). The industry has
moved from 206-diameter necks on 211-diameter bodies to
Figure 2. Can-bottom profiles: A, original bottom profile; B,
Alcoa B-53, V-bottom profile; C, Alcoa B-80. (Courtesy of Alcoa.)
Figure 3. Design elements.
Figure 4. Double-, triple-, quadruple-, and spin-neck can.
194 CANS, ALUMINUM

204- and 202-diameter necks. These diameter changes
have reduced the metal gauge and net weight from
0.0108 in. and 7.3 lb/1000 for 206 ends to 0.0088 in. and
5.3 lb/1000 for 202 ends. The primary development is with
the 202 can ends, 200 is also being developed. Wide mouth
cans openings are now being marketed that lower metal
use by 10% (1). Resealable cans are being marketed in
France. A small amount of plastic will be used at the
opening and will not affect recycling (2).
Net can weights have also been reduced from approxi-
mately 30 lb/M at 0.0125 in. to 23 lb/M at 0.0108 in. This
has been accomplished primarily by creating new stron-
ger dome geometry, along with dome postreforming
technology to improve performance criteria, such as
dome buckle, drop resistance, and dome growth. These
new dome designs are more difficult to manufacture, but
provide improved performance and stacking at reduced
gauges. Alcoa is lightweighting by using the thinnest
aluminum possible. Finite element modeling helps with
design prototypes (3). Can body gauging down has re-
sulted in a 25–30% reduction in metal use (4).
Can sidewalls have been further reduced from 0.0045
in. down to 0.0038 in. nominal, and the number of die
necks utilized to produce finished necks has increased to
as many as 14 for the 202 diameter (4). Multidie necks
combined with spin necking are still popular for all three
neck diameters, and several canmakers are using multidie
necking with spin flow necking.
BIBLIOGRAPHY
Terre Reingardt, and N. F. Nieder, ‘‘Cans, Aluminum’’ in The
Wiley Encyclopedia of Packaging, 2nd edition, John Wiley &
Sons, New York, 1997, pp. 132–134.
Cited Publications
1. All About Cans, Can Manufacturers Institute, www.cancen-
tral.com, accessed July 2008.
2. ‘‘Coca-Cola’s Resealable Can,’’ Ball Packaging Europe,
www.ball-europe.com, March 27, 2008.
3. ‘‘Heavy Duty Light Weighting,’’ www.psc.edu/science/alcoa,
accessed July 2008.
4. Aluminum Beverage Can Innovations, Aluminum Can Council,
Washington, D.C.
CANS, COMPOSITE
M. B. EUBANKS
Sonoco Products Company
Updated by Staff
INTRODUCTION
The ‘‘composite can’’ is broadly defined as a can or container
with body and ends made of dissimilar materials. In
commercial practice the composite can has several more
focused descriptions: cylindrical or rectangular shape; rigid
paperboard (or plastic) body construction; steel, aluminum,
or plastic end closures; generally employing inside liners
and outside labels; and generally delivered with one end
attached and one end shipped separately to be attached by
the user. Today the most common form is the cylindrical
paperboard can with a liner, a label, and two metal ends.
There are other packaging forms similar to traditional
composite cans that are sometimes called composites,but
which are more closely related to folding cartons. These are
single-wrap fiber cans made from blanks and mainly
designed for the users’ in-plant production (see Cans,
Composite, Self-Manufactured).
The composite can is not a new package. Early applica-
tions included refrigerated dough and cleansers. As tech-
nology improved, motor oil and frozen juice concentrates
were converted to composites. In the last decade, snack
foods, tobacco, edible oils, shortening, powdered bev-
erages, pet foods, and many other items have been added
to the list. Figure 1 illustrates some of the products
currently available in composite cans.
Since their introduction, composite cans have generally
been marketed and used as a lower-cost packaging form
relative to metal, plastic, and glass. This emphasis has
overshadowed other positive attributes such as the fre-
quent use of recycled materials, weight advantages, noise
reduction, improved graphics, and design flexibility. Be-
cause of its early applications in cleansers and oatmeal,
the composite can once suffered from a low performance
image associated with the term ‘‘cardboard can.’’ This
term cannot begin to describe the current and potential
properties of the paperboard and other materials that go
into today’s composite cans.
By utilizing the best combination of materials, compo-
site-can construction ensures optimum presentation and
mechanical strength as well as hermetic protection. The
choice of materials and production techniques for can
components offers packaging solutions that are flexible
and cost effective for mass consumption and luxury pro-
ducts as well as for numerous industrial applications.
Composite cans are normally available in diameters of
1–7 in. (3–18 cm) and heights of 1–13 in. (3–33 cm). Dimen-
sional nomenclature for composites has been adapted from
Figure 1. Examples of composite cans in commercial use.
CANS, COMPOSITE 195
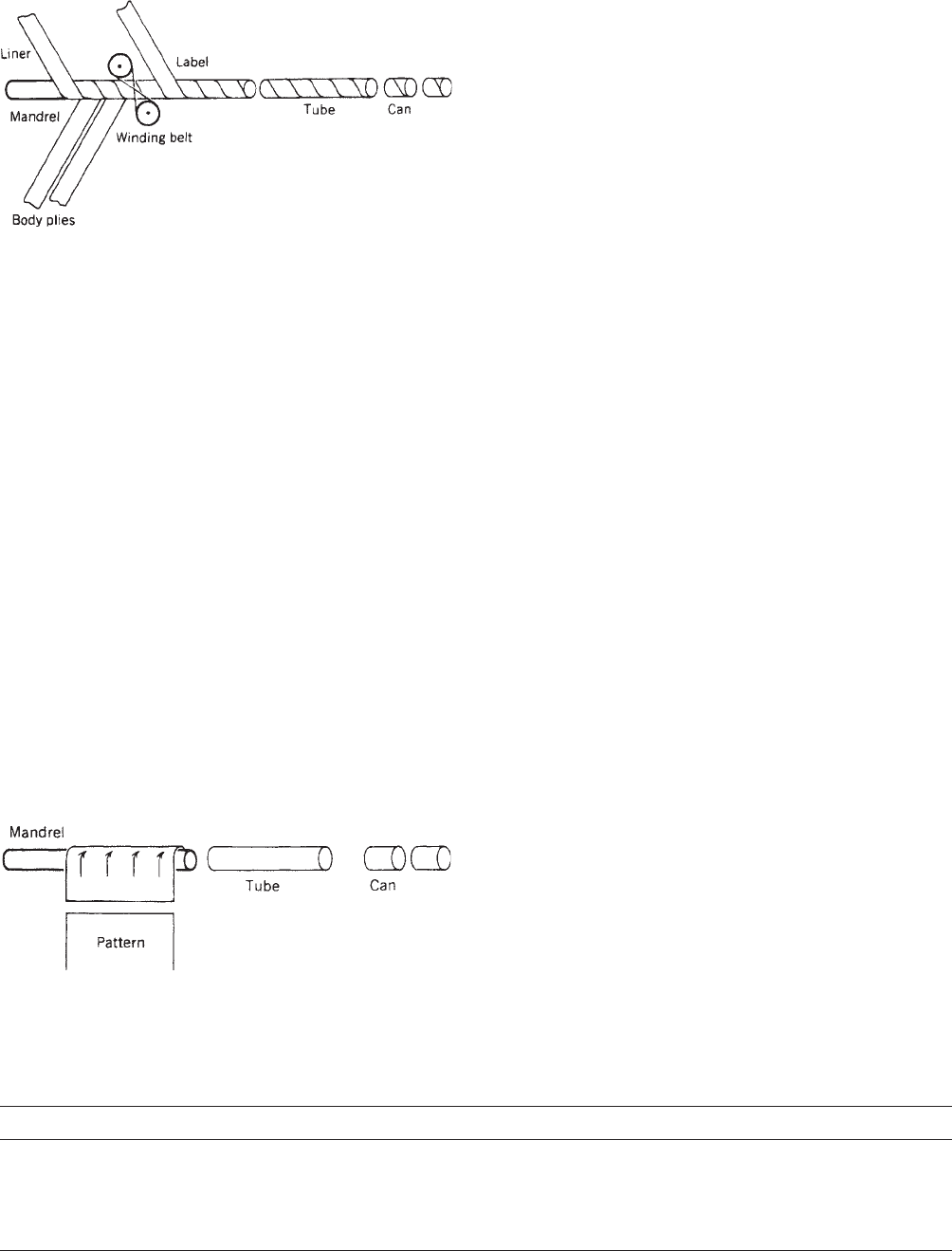
metal cans, and nominal dimensions are expressed in
inches and sixteenths of an inch (see Cans, Steel). Hence,
a 404 diameter can has a nominal diameter of 4
4
=
16
in.
(10.8 cm). Likewise, a height of 6
10
=
16
in. (16.8 cm) is
expressed as 610, and so on.
MANUFACTURING METHODS
Composite-can bodies are produced by two basic methods:
spiral winding and convolute winding. Figure 2 shows
a schematic drawing of the spiral process. Multiple
webs including a liner, body plies, and label are treated
with adhesive and wound continuously on a reciprocating
mandrel. The resulting tube is trimmed and the can
bodies are passed on to flanging and seaming stations.
Figure 3 depicts the convolute process, wherein a pattern
is coated with adhesive and entered onto a turning
mandrel in a discontinuous process. Trimming and fin-
ishing operations for the convolute and spiral systems
are virtually the same. Most composite-can manufac-
turers favor the spiral process in situations where
long production runs and few line changeovers are
involved.
BODY CONSTRUCTION
Paperboard
The primary strength of the composite can is derived from
its body construction, which is usually paperboard (see
Paperboard). Body strength in composite cans is an attri-
bute that has improved over the years and can be varied to
meet many application demands.
In the early years of composite can development, it was
common for can manufacturers to start with a readily
available body stock such as kraft linerboard or tube-
grade chip. These boards are adequate for most applica-
tions, but new boards with special qualities have also been
developed for more demanding end-use requirements.
Research and development efforts in the combination
paperboard field encompass a number of areas of exper-
tise. Examples are (a) engineering mechanics concepts
used to develop structural criteria and to develop tests
to ensure that the paperboards possess the necessary
resistance to bending, buckling, and creep; (b) surface
chemistry used to predict the resistance of the board to
penetration of adhesives, coatings, and inks and, similarly,
protection from environmental conditions such as rain,
high humidity, and freezing temperatures; and (c) process
engineering used to optimize paperboard manufacturing
and converting and to assist in quality assurance pro-
grams (see Specifications and Quality Assurance).
The scientific and engineering efforts have, in many
instances, supplemented the artisan-like judgment of
yesterday’s papermaker. However, in many cases the
new technological approaches have been blended with
the papermaker’s art to achieve the best of both worlds.
As a result, the following advances have taken place:
super-high-strength board that can be converted into
composite cans with reasonable wall thicknesses that
can resist implosion when subjected to near-perfect va-
cuum; resin-treated paperboards that retain their struc-
tural integrity when thoroughly wet; chemically treated
paper that resists penetration by water over long time
intervals; and paperboards that can be distorted, rolled,
and formed in high-speed converting equipment without
‘‘creeping’’ back to their original shape.
Adhesives
The adhesives (see Adhesives) and coatings used in the
manufacture of composite cans have also been improved to
provide better heat and water resistance plus increased
Figure 2. Spiral-wound composite-can fabrication.
Figure 3. Convolute composite-can fabrication.
Table 1. Adhesives Used in Composite-Can Manufacture
Adhesives Properties
Poly(vinyl alcohol–acetate) blends Good initial tack, good runnability, moderate to good water resistance
Dextrin Fast tack, poor water resistance
Animal glue Good tack, vulnerable to insect attack
Polyethylene Requires heat, good dry bond, moderate water resistance
Hot melts Require heat, difficult to handle, good water resistance, good water bond
196 CANS, COMPOSITE

operating efficiency. Most product applications require
precision gluing equipment to control the amount and
position of the adhesive on each web. The most commonly
used adhesives in today’s composite can production are
listed in Table 1.
Liners
Like all successful packages, the composite can must
contain and protect the products. For that reason, contin-
uous improvement is sought in liner materials. By com-
bining materials such as LDPE (see Polyethylene, Low-
Density), HDPE (see Film, High-Density Polyethylene;
Polyethylene, High-Density), PP (see Film, Nonoriented
Polypropylene; Film, Oriented Polypropylene; Polypropy-
lene), ionomer (see Ionomers), PVDC (see Vinylidene
Chloride Copolymers), metalized PET, and PE (see Film,
oriented Polyester; Polyesters, Thermoplastic) with alu-
minum foil (see Foil, Aluminum) or kraft (Paper; Paper-
board), the barrier properties of composites can be
matched with a broad range of product requirements
(see Laminating, Multilayer Flexible Packaging). The
polymers may be included as film or coatings, or both.
Tables 2, 3 illustrate the physical properties of some of the
more common liner films and complete liner constructions.
The laminates shown in Table 3 vary significantly in
cost. Depending on can size, the difference in cost per
thousand units can be substantial. The foil-based lami-
nates, which provide virtually 100% water and gas bar-
rier, are becoming quite expensive. If less than total
impermeability is acceptable, it pays to investigate coated-
or plain-film alternatives. For example, packers of frozen
juice concentrates have gradually moved away from foil
liners to laminations of PE or ionomer/PE and kraft.
Table 2. Physical Characteristics of Commonly Used Liner Films
Polypropylene
Physical
Characteristics
Oriented Oriented PVDC
Coat
Nonoriented Polyester LDPE Ionomer
Properties
50–MD
a
(0.5)
Tear strength, gf (N) 3–10
a
(0.03–
0.10)
3–10 (0.03–
0.10)
300–XD
a
(3) 12–27
a
(0.12–
0.27)
50–150
a
(0.5–
1.5)
50–150
a
(0.5–
1.5)
Burst strength, psi
(kPa)
55–80 (379–
551)
10–12 (69–83) 10–12 (69–83)
WVTR, g mil/(100 in.
2
24 h) [g mm/(m
2
d)]
0.75 [29.5] 0.3–0.5 [11.8–
19.7]
1.5 [59] 1.5 (59) 2.0–3.0 (78.7–
118.1)
2.0–3.0 (78.7–
118.1)
O
2
rate, cm
3
mil/(100
in.
2
24 h) [cm m/
m
2
d)]
160 [630] 1–3 [3.9–11.8] 160 [630] 3.0–4.0 (11.8–
15.7)
500 (1970) 250–300 (985–
1180)
elongation, % 35–475 35–475 550–1000 60–165 100–700 400–800
Product Resistance
b,c
Strong acids G G G G G G
Strong alkalines G G G G G G
Grease and oil G G G G G E
Source: Sonoco Products Company, Hartsville, SC.
a
gf/mil (0.386 N/mm) thickness.
b
G = good.
c
E = excellent.
Table 3. Physical Characteristics of Composite Can Liners
WVTR
b
at 1001F (37.8 1C) and 90% rh
Composite
a
Flat Creased O
2
Permeability
c
,cm
3
/(m
2
d)
100 ga PP/adh/100 ga AF/LDPE/25# MGNN kraft o0.001 o0.001 o0.001
100 ga PP/adh/35 ga AF/LDPE/25# MGNN kraft 0.06 o0.02
12# ionomer/35 ga AF/LDPE/30# XKL kraft 0.01 o0.02
1# PET slipcoat/35 ga AF/casein/25# MGNN kraft 0.09 0.93 o0.001
1# PET slipcoat/35 ga AF/LDPE/25# MGNN kraft 0.06 0.09 o0.001
12# HDPE/20# MGNN kraft 15.35 153
14.4# HDPE/20# MGNN kraft 12.09 126
a
key: # = lb/ream = 454 g/ream. 100 ga = 0.001 in. = mil = 25.4 mm. MGNN = Machine-grade natural Northern. XKL = extensible kraft linerboard.
b
ASTM Test Method E 96-80.
c
ASTM Test Method D 3985.
CANS, COMPOSITE 197
