Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


Light Protection by Glass and Plastics
The light protection of glass and plastic depends on mate-
rial thickness and type and concentration of pigment.
Higher pigmentation will drastically reduce the light trans-
mittance and increase the protection of the material. As
shown by Figures 2 and 3, clear glass and plastic (injection
molded polypropylene) transmit about 90% of the visible
light and, respectively, 35% and 56% of the UV light, and
they are therefore poor UV and visible light barriers. Blue
glass and plastics transmit light in the UV, blue, and the
red regions of the spectrum. As many photosensitizers
absorb light in the UV and blue regions of the spectrum,
this light is particularly harmful to many foods. Green
glass and plastics transmit light in the UV, violet/blue, and
green regions of the spectrum. In this area the porphyrins
have some small absorption maxima, and prolonged ex-
posure in this region may cause food deterioration.
Brown glass transmits light in the yellow, orange, and
red region of the spectrum, an area where chlorophylls
also have absorption maxima. However, brown glass is
usually densely pigmented, offering good light protection
for a variety of food products including milk and beer.
Figure 3 shows the light transmittance through an
SiO
2
-pigmented (white) injection-molded polypropylene
plastic. Addition of this pigment results in a relative sharp
cut off around 400 nm offering good protection against UV
radiation. However, because normal artificial light sources
used in retail normally emits only small amounts of light
in the UV region (many have only a small emission peak in
the 370-nm area), the transmittance of light by packaging
materials in this region is not a serious threat during
indoor distribution, storage and display. A limited effect is
therefore achieved by adding a UV block to a transparent
packaging material.
Light Protection by Liquid Paperboard
Different qualities of liquid paperboard coated with low-
density polyethylene are used worldwide for packaging
of fresh milk products. Figure 4 shows that despite
their modest thickness (0.5–0.6 mm) commercial liquid
9
8
7
6
5
% Light transmission
4
3
2
1
0
200 300 400 500
Wavelength (nm)
600 700 800 900
Bleached, unprinted
Bleached, pink printed
Unbleached, unprinted
Unbleached, pink printed
Figure 4. Light transmission in three different
qualities of unprinted and pink printed liquid paper-
board: bleached, unbleached, and bleached with
oxygen and light barrier as measured by a Perkin
Elmer Lambda 800 UV–vis spectrometer with an
integrating sphere (Perkin Elmer Ltd., Buckin-
ghamshire, UK).
200
0
10
20
30
40
50
% Light transmission
(Transparent film)
% Light transmission
(Metallized films)
60
70
80
90
100
300 400 500
Wavelength (nm)
600 700 800 900
0
1
2
3
4
5
Transparent film
Metallized film, blue print
Metallized film, unprinted
Metallized film, red print
Figure 5. Light transmission in unmetallized, me-
tallized, and printed, metallized polypropylene films
as measured by a Perkin Elmer Lambda 800 UV–vis
spectrometer with an integrating sphere (Perkin El-
mer Ltd., Buckinghamshire, UK).
658 LIGHT PROTECTION FROM PACKAGING
paperboard qualities offer a relatively good protection
against light. Only 4.3% of the light in the visible (400–
700 nm) region of the spectrum and 0.03% in the UV
region are transmitted through the bleached paperboard.
The unbleached board offers an even better light barrier.
Only 0.8% of the visible light and 0% of the UV light is
transmitted. Additional protection is achieved by printing.
Only 2.6% of the light is transmitted through pink printed
bleached paperboard, and most of the reduction appears in
the region of the spectrum that is most detrimental to milk
products—that is, up to 550 nm. Pink printed unbleached
paperboard offers very good light protection. Less than
0.1% of the visible light is transmitted through this
material.
If liquid paperboard is foiled with aluminum, it be-
comes virtually impermeable to light. Also the SiO
2
/car-
bon black/EVOH-based light and oxygen barrier used by
one supplier for certain products in some markets also
offers very good light protection. Only 0.3% of the visible
light is transmitted through this material (for unprinted,
bleached board).
Light Protection by Metallization of Plastic Films
Metallization may be used to improve both light barrier
and oxygen barrier of plastic films. As shown by Figure 5,
the transmission in the visible region of the spectrum is
significantly reduced from 92% for unmetallized film to
0.5% when the same film is metallized. The reduction in
light transmission caused by the metallization is rather
uniform in the visible area of the spectrum, similar to the
effect of a heavily pigmented gray (neutral) filter. The
effect of metallization is still very good in the UV region of
the spectrum, but not as good as in the visible region. As
for liquid paperboard, further reduction in light transmis-
sion is achieved by additional printing of the metallized
film. The effect of blue print results in neglectable effect on
light transmission in the blue region because blue print
transmits light in the blue region of the spectrum. Simi-
larly, the effect of red print is neglectable in the red region
of the spectrum.
BIBLIOGRAPHY
1. A. Ryer, International light, Inc. web information. Available
at http://www.cbpf.br/pdf/Light%20Measurement%20Hand-
book.pdf. Accessed June 8, 2007.
2. Wikipedia Foundation Inc, web information. Available at
http://en.wikipedia.org/wiki/Candela. Accessed June 8, 2007.
3. D. G. Bradley and D. B. Min, Crit. Rev. Food Sci. Nutr. 31(3),
211–236 (1992).
4. S. K. Pushpan, S. Venkatraman, V. G. Anand, J. Sankar, D.
Parmeswaran, S. Ganesan, and T. K. Chandrashekar, Curr.
Med. Chem. 2, 187–207 (2002).
5. D. B. Min and J. M. Boff, Compr. Rev. Food Sci. Food Safety 1,
58–72 (2002).
6. C. S. Foote, Photochem. Photobiol. 54(5), 659 (1991).
7. J. Lindsay, Oregon Medical Laser Company, web information.
Available at http://omlc.ogi.edu/spectra/PhotochemCAD/
html/index.html. Accessed June 8, 2007.
8. M. Bekbo
¨
let, J. Food Prot. 53(5), 430–440 (1990).
9. J. P. Wold, A. Veberg, A. Nilsen, V. Iani, P. Juzenas, and J.
Moan, Int. Dairy J. 15, 343–353 (2005).
10. J. P. Wold, Spectrosc. Eur. 18(5), 8–13 (2006).
11. M. B. Korycka-Dahl and T. Richardson, Crit. Rev. Food Sci.
Nutr. December, 209–241 (1978).
12. E. Choe, R. M. Huang, and D. B. Min, J. Food Sci. 70(1), R28–
R36 (2005).
13. H. Larsen, P. Lea, and M. Rødbotten, Food Qual. Pref. 16(7),
573–584 (2005).
14. L. Piergiovanni, and S. Limbo, Packag. Technol. Sci. 17(3),
155–164 (2004).
15. J. P. Wold, A. Veberg, F. Lundby, A. N. Nilsen, and J. Moan,
Int. Dairy J. 16, 1218–1226 (2006).
LIPID OXIDATION: CHEMICAL
STABILIZATION
KAREN M. SCHAICH
Department of Food Science,
Rutgers University, New
Brunswick, New Jersey
INTRODUCTION
Packaging must provide physical, microbiological, and che-
mical protection for foods . F or the most part, providing
physical protection is straightforward, as is preventing
contamination of foods after processing and packaging.
More challenging is meeting the requirement for inert
storage environments that inhibit respiration in fresh fruits
and vegetables, microbial growth in prepared foods and raw
meats, and chemical degradation in stored foods . Lipid
oxidation, or oxidative rancidity, is the chemical reaction
that most limits long-term shelf life of foods , producing
characteristic off-odors and flavors, browning, texture
changes, co-oxidation of proteins and vitamins, and even
toxic products in foods. Lipid oxidation is also a major cause
of deterioration in cosmetics and many personal care pro-
ducts, changing surface tensions, breaking emulsions, oxi-
dizing active components, and producing off-odors. Because
of these extensive effects on product quality, lipid oxidation
also forces the greatest demands on food packaging .
This chapter will describe the reactions of lipid oxida-
tion in general terms and show how packaging can be used
to inhibit and control lipid oxidation. Additional details of
lipid oxidation reactions, factors affecting lipid oxidation,
and strategies for prevention may be found in comprehen-
sive reviews listed at the end of the chapter.
CHARACTERISTICS OF LIPID OXIDATION
Lipid oxidation is a chemical process in which unsaturated
fatty acids in phospholipids and triacylglycerides react
with oxygen and degrade to a variety of volatile and
nonvolatile products. Lipids oxidize by a free radical chain
reaction process involving three stages: initiation, propa-
gation and branching, and termination. The classical
LIPID OXIDATION: CHEMICAL STABILIZATION 659

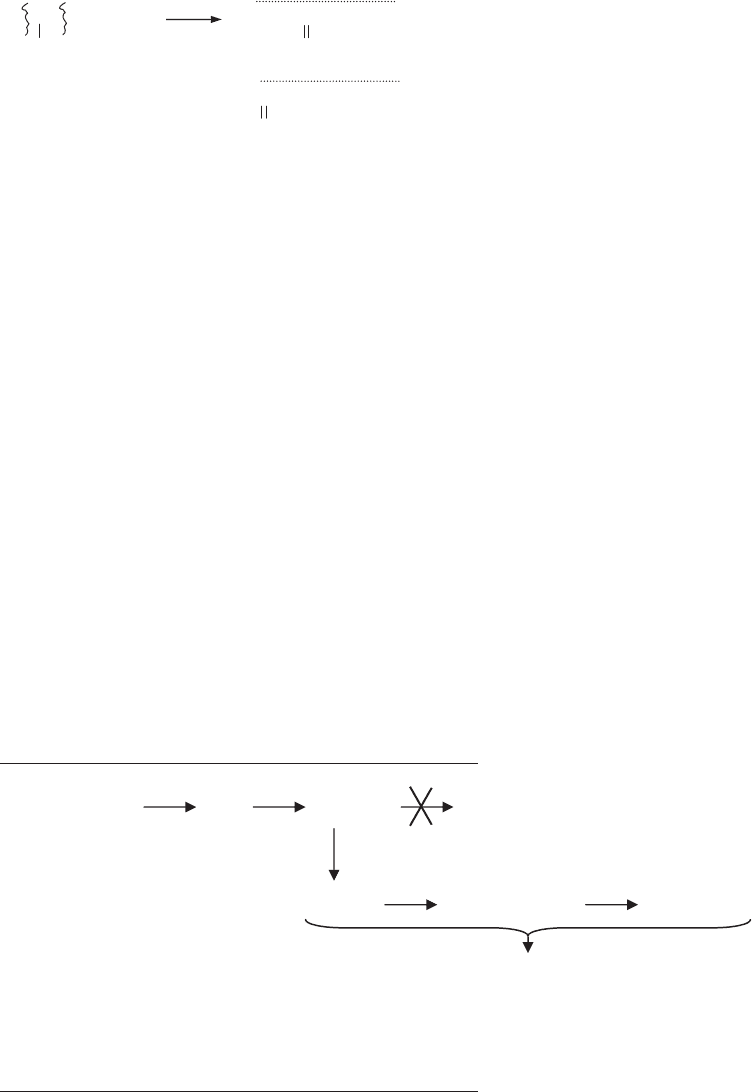
sequence describing lipid oxidation is shown in Figure 1,
along with catalysts and protectants at each stage.
The radical chain reaction has several critical conse-
quences for lipid oxidation:
. Once started, the reaction is self-propagating and
self-accelerating; that is, it keeps itself going.
. Many more than one LOOH is formed and more than
one lipid molecule is oxidized per initiating event.
. Very small amounts of pro- or antioxidants cause
large rate changes.
. Multiple intermediates and products are formed, and
they change with reaction conditions and time.
Unlike most chemical reactions, lipid oxidation is a
dynamic process with no fixed intermediates or endpoints.
That the reactions of lipid oxidation change with time and
conditions presents distinct challenges in measuring and
controlling lipid oxidation, and it explains in part why
lipid oxidation is a major problem in storage stability of
foods and other lipid-containing products.
STAGES OF LIPID OXIDATION
Initiation
LH ! L
ð1Þ
Oxygen cannot add directly to the double bonds in
unsaturated fatty acids, but it adds almost instantaneously
to L
free radicals to form peroxyl radicals that drive
radical chain reactions. Thus, lipid oxidation requires
special catalysts, or initiators, to form L
,oncarbonatoms
of lipid (L) molecules. Common initiators include ultraviolet
light, photosensitizers and visible light, radicals from other
sources, ionizing radiation, metals, heme compounds, and
heat.
Propagation
L
1
þ O
2
! L
1
OO
ð2aÞ
L
1
OO
þ L
2
H ! L
1
OOH þL
2
! L
2
OO
ð2bÞ
L
2
OO
•
+ L
3
HL
2
OOH + L
3
•
etc. L
n
OOH (2c)
Propagation reactions establish the free radical chain
and keep it going. In this stage, lipid alkyl radicals, L
,
react with oxygen almost instantaneously to form peroxyl
radicals, LOO
, that are major players in lipid oxidation.
Peroxyl radicals very selectively abstract hydrogens from
adjacent lipid molecules, forming hydroperoxides and
generating new L
radicals in the process. Each new L
radical in turn repeats the process to provide the driving
force in the chain reaction. Chain propagation continues
indefinitely until no hydrogen source is available or the
chain is intercepted.
Initiation (formation of ab initio lipid free radical)
Propagation
Free radical chain reaction established
etc.
Free radical chain branching (initiation of new chains)
++
HOH
L
1
OOH + L
n
OO
•
L
4
•
L
1
•
+ O
2
L
1
OO
•
+ L
2
H
L
2
OO
•
+ L
3
HL
2
OOH
+ L
3
•
L
1
OOH
+ L
2
•
L
1
OO
•
L
1
•
L
n
OH
L
n
OOH
L
4
H
L
1
O
•
+ L
n
OOH
L
1
OO
•
+ L
n
OOH
L
n
OO
•
HO
•
L
n
O
•
L
n
O
•
L
n
OO
•
L
n
•
L
n
O
•
L
n
OO
•
LOO
•
LO
•
L
n
•
L
n
OOH
L
n
OOH
L
n
OOH
L
n
OO
•
+ H
+
(oxidizing metals)
L
n
O
•
+ OH
–
(reducing metals)
L
n
O
•
+
•
OH (heat and UV)
L
1
OH + L
n
OO
•
Termination (formation of nonradical products)
+
Radical recombinations
polymers, nonradical monomer products
(ketones, ethers, alkanes, aldehydes, etc.)
Radical scissions
nonradical products
(aldehydes, ketones, alcohols, alkanes, etc.)
CLASSICAL FREE RADICAL CHAIN REACTION MECHANISM OF LIPID OXIDATION
Catalysts Inhibitors
Dark, filters
Refrigeration
Metal chelators
1
O
2
quenchers
Dark, filters
Refrigeration
Metal chelator
s
Water
Light
Heat
Radicals
Metals
Oxygen
Metals
Light
Heat
a
w
>~0.3
Heat
Heat
High pO
2
Water
Protic
solvents
Vacuum
Low pO
2
Radical
scavengers
Radical
scavengers
Radical
scavengers
Low pO
2
Dry systems
Neat oils
L
1
H
L
n
OOH
Figure 1. The stages and individual reactions involved in lipid oxidation as described by the classical free radical chain reaction, and the
catalysts and inhibitors associated with each stage.
660 LIPID OXIDATION: CHEMICAL STABILIZATION

Chain branching
L
n
OOH
L
n
O
•
+ OH
–
(3a)
L
n
OOH L
n
OO
•
+ H
+
more chain
reactions
(3b)
L
n
OOH L
n
O
•
+
•
OH (3c)
Branching reactions are a second stage of propagation
in which the radical chain reaction is expanded, establish-
ing new chains at faster rates. In basic propagation (Reac-
tion 2), peroxyl radicals are the only chain carriers; they
react rather slowly (k = 36–62 L mol
1
s
1
) and quite speci-
fically, and the chain continues one abstraction at a time
from the initiation point. Without forces that decompose
hydroperoxides, this process can continue indefinitely
while hydroperoxides accumulate. However, UV light,
moderate heat, metals, or heme compounds decompose
hydroperoxides to give alkoxyl and peroxyl radicals plus
ions (metals) or alkoxyl radicals and hydroxyl radicals (UV
light and heat), all of which initiate new free radical chains
that branch off of and expand the original chain. Hence,
the net effect of LOOH breakdown is a transition in
mechanism and kinetics. Lipid oxidation gathers steam,
increasing in rate and extent as LO
becomes the domi-
nant, faster chain carrier (k =10
6
–10
7
Lmol
1
s
1
) and
secondary chains dramatically amplify and broadcast lipid
oxidation. A single initiating event can thus lead to
sequential oxidation of literally hundreds of molecules in
the primary chain and in secondary branching chains.
In early stages of lipid oxidation while hydroperoxides
are just beginning to accumulate, the hydroperoxide de-
compositions occur monomolecularly (Reaction 3). As oxi-
dation progresses and hydroperoxides accumulate,
decomposition shifts to bimolecular mechanisms that
convert stable or slowly reacting species to multiple highly
reactive radicals:
2 LOOH LOOH…HOOL LO
•
+H
2
O+
•
OOL (4)
LOO
þ LOOH ! LO
þ epoxy LO
þ
OH ð5Þ
The initial effect of bimolecular decomposition is a dra-
matic acceleration of lipid oxidation, but eventually a
point of net LOOH decomposition is reached. At that
point, termination reactions such as radical recombina-
tion or alkoxyl radical scission take over, and oxidation
slows, stable secondary products form, and off-flavors and
odors become detectable.
Termination
Termination implies that a process is coming to a close.
For lipid oxidation as a process, ‘‘termination’’ is a mis-
nomer because lipid oxidation never fully stops. A specific
radical may be terminated as it forms some product, but it
leaves behind another radical so the chain reaction con-
tinues. Net oxidation slows down when radical quenching
processes exceed the rate of new chain production, and the
momentum of oxidation shifts from radical propagation
and chain expansion to generation of stable products. To
reflect this shift, ‘‘termination’’ in the discussion below
refers to stopping an individual lipid radical, not the
overall reaction. The cumulative effect on a reaction
system such as food is determined by the number of
radical chains being terminated.
Lipid free radicals terminate to form nonradical pro-
ducts primarily by three mechanisms:
(a) Radical recombinations
(b) Scission reactions when proton sources (e.g., water)
are present to stabilize products
(c) Co-oxidation of nonlipid molecules such as proteins
The mechanisms dominating in a given system are influ-
enced by the nature and concentration of the radicals, the
temperature and oxygen pressure, and the solvent.
Radical Recombinations. The limitless number of varia-
tions possible for radical recombinations is one reason for
the broad range of oxidation products detected in lipid
oxidation. Despite the possibilities, recombinations are
not random, and distinct patterns of favored recombina-
tions have been identified. Some of the most important
recombinations and their products are
Alkyl radicals ðL
1
þ L
2
Þ!alkane polymers ð6aÞ
Peroxyl radicals ðL
1
OO
þ L
2
OO
Þ
! alcohols and ketones; alkyl peroxides
ð6bÞ
Alkoxyl radicals ðR
1
O
þ R
2
Þ
! ethers; ketones; alkanes
ð6cÞ
ðR
1
O
þ R
2
O
Þ!alkyl peroxides; ketones; alcohols ð6dÞ
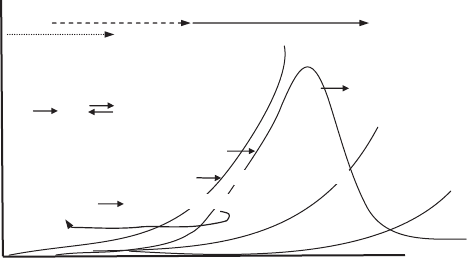
Temperature and oxygen pressure are key determi-
nants of radical recombination pathways (Figure 2). L
reactions dominate under low-oxygen (pO
2
= 1 to about
80–100 mm Hg) and high-temperature (reduced O
2
solubi-
lity) conditions. High pO
2
and lower temperatures favor
LOO
reactions. LO
contributions to the product mix
dominate in secondary stages of oxidation and at moder-
ate temperatures and oxygen pressures when LOOH or
LOO
decompositions are faster than their formation.
Radical recombinations are diffusion- and concentration-
controlled, so they are favored in neat oils; they decrease
with lipid dilution.
Formation of dimers and polymers increases viscosity
in oxidized oils. Perhaps just as importantly, alcohols,
ketones, ethers, and so on, are volatile compounds and
flavor components that augment those produced in scis-
sion reactions and provide the undertones and secondary
notes that round out flavors. Ketones and dialkyl perox-
ides, in particular, are unique to recombination reactions.
Scission Reactions of LO
. Of all the pathways active in
lipid oxidation, scission of alkoxyl radicals probably has
the greatest practical consequences for any kind of con-
sumer product because the products generated are
LIPID OXIDATION: CHEMICAL STABILIZATION 661

pO
2
(mm Hg)
ROO
•
rxs.
RO
•
rxs
1 10 100
% Termination
R
•
rxs
pO
2
(mm Hg)
ROO
•
/ R
•
1.0
20 40 60 80
45°C
35°C
25°C
Figure 2. Effects of oxygen and temperature
on termination processes in lipid oxidation.
[Redrawn from Labuza (1) and Schaich (2).
Used with permission.]
Table 1. Typical Scission Products Produced from Important Oxidized Polyunsaturated Fatty Acids Undergoing
Oxidation and Associated Off Flavors
Product Classes Carbon Chain Lengths Major Products Associated Flavors
Oleic Acid
Hydrocarbons 6–8 Nonanal Fresh, citrus
Alkanals 2–4, 5, 6, 7, 8, 9, 10, 11 Octanal Fresh, citrus
2-Alkenals 6–9, 10, 11 Undec-2-enal Sweet, fatty
Acids 1, 6, 7, 8, 9 Undecanal Fatty
Alkanols 5–8 2-decenal Tallowy
Alkylformates 2–8
Linoleic Acid
Hydrocarbons 3–5 Hexanal Green-fruity, bitter almond
Alkanals 3, 4, 5, 6, 7, 8 2,4-Decadienal Fried
Alkenals 7, 8, 9, 10 2-Octenal Woodbugs-fatty, nutty
Dienals 9, 10 2-Heptenal Fatty-putty, bitter almond
Oxo-alkanals 7–9 1-Octene-3-one Metallic moldy, mushroom
Ketones 7, 8 Pentanal Sharp, bitter almond
Alcohols 3, 4, 5, 6, 7, 8 Heptanal Oily putty, soapy, fruity
Acids 1, 5, 6, 7, 9
Esters 1, 6, 7, 8
Linolenic Acid
Hydrocarbons 1–3 2,4-Heptadienal Rotten apples, fried
Alkanals 1, 2, 3, 6 3-Hexenal Green leaves, green tomatoes
Alkenals 4, 5, 6, 7 Propanal Sharp, irritating
Dienals 7–9 2,4,7-Decatrienal Fishy
Trienals 10 2-Pentenal Sharp, painty, green
Ketones 5, 18 Octadienal
Alcohols 3, 4, 5, 6, 7, 8 Pentene-3-one Sharp, fishy
Acids 1, 5, 6, 7, 9 Octadiene-2-one Fatty-fruity
Esters 1, 6, 7, 8
Oxo-alkanals
Arachidonic Acid
Alkanal 2, 6, 7 Hexanal Green-fruity, bitter almond
Alkenal 7, 8, 9, 11 2,4-Decadienal Fried
Dienal 9, 10, 11, 12 2,4,7-Tridecatrienal
Ketones 2-Heptenal Fatty-putty, bitter almond
Alkanes 5, 6 2-Octenal Woodbugs-fatty, nutty
Aldehyde esters 4, 5 Pentanal Sharp, bitter almond
1-Octen-3-one Metallic, moldy, mushroom
4-Decenal Tallowy
3,5-Undecadien-2-one
2,6-Dodecadienal
662 LIPID OXIDATION: CHEMICAL STABILIZATION
responsible for the distinctive volatile off-odors and off-
flavors associated with rancidity. Lipid alkoxyl radicals
undergo scission of the C–C bond on either side of the LO
group to yield a mixture of carbonyl products (typically
aldehydes, alkanes, and oxo-esters from the initial alkoxyl
radicals) and free radicals:
R
1
–CH–R
2
-COOH R
1
•
+ CH–R
2
-COOH
or
R
1
CH +
•
R
2
-COOH
(7)
O
•
O
O
βα
β
α
Unsaturated radical fragments oxidize still further and
undergo secondary scissions in similar fashion to produce
carbonyls and alkanes of still shorter chain length. Con-
sequently, the mix of breakdown products that accumulate
in oxidized lipids can become quite complex, as shown in
Table 1 for the four major unsaturated fatty acids.
Scission requires a strong proton donor such as water
to stabilize the products and drive the reaction forward.
Scission is a minor process in neat lipids at room tem-
perature. However, in the presence of water (e.g., in
emulsions), acid, or other polar solvents, scission is rapid
and competitive with hydrogen abstraction, accounting for
about half of alkoxyl radical reactions, even in early
oxidation. Scission is also favored in dilute solution of
lipids where there is reduced competition from hydrogen
abstraction. However, because LO
scission has a large
activation energy and Arrhenius factor, the greatest con-
tribution of this termination (and propagation) reaction is
at elevated temperatures. Heat accelerates alkoxyl radical
scissions in all solvents, although the pattern of cleavage
may change as temperature increases.
The most notorious scission product of lipid oxidation,
malondialdehyde (MDA), arises from multiple scissions
of cyclic internal hydroperoxides formed in fatty acids
with three or more double bonds (linolenic and higher).
MDA formation is facilitated in aprotic solvents and by
low lipid concentrations, limited oxygen pressures, and
photosensitized oxidation; and it requires conditions for
endoperoxide cleavage, namely, mild heat and acid. In
autoxidizing lipids, yields of authentic MDA are usually
less than 0.1%; but in photosensitized fatty acids where
internal hydroperoxides are formed in high concentra-
tions, MDA concentrations can reach 5% or higher. Mal-
onaldehyde is probably the most common measure of lipid
oxidation in complex food systems, but its analysis is quite
often misused and misinterpreted.
Co-oxidation of Nonlipid Molecules. Hydrogen abstrac-
tions by lipid alkoxyl and peroxyl radicals are not selective
for fatty acids. Nonlipid molecules such as amino acids,
proteins, nucleic acids, antioxidants, carotenoids and
other pigments, carbohydrates, and even vitamins also
have abstractable H’s and thus can serve as substitutes for
fatty acids, intercepting the lipid free radical chain. This
process terminates lipid oxidation chains, but reroutes
radicals and oxidation to proteins and other biomolecules.
As in lipids, these radicals add oxygen to form peroxyl
radicals that may also abstract H’s from other molecules
or lead to oxidative degradation of the molecular target,
whatever class of compounds it may be. In this way, lipids
serve to ‘‘broadcast’’ oxidation damage to other molecules
which then provide footprints of lipid oxidation in foods
and biological systems.
Radical transfer to nonlipid molecules has several
critical consequences. Co-oxidation reactions alter lipid
oxidation kinetics, mechanisms, and overall effects in
foods and biological systems. Immediately, there is an
apparent antioxidant effect in terminating individual lipid
radical chains and slowing accumulation of measurable
oxidation products. However, if initiation is not concur-
rently blocked, what appears to be inhibition of lipid
oxidation is false: Detectable oxidation products are
merely shifted from lipids to other molecules, but ab initio
generation of new chains by initiators continues. If tradi-
tional lipid oxidation products such as peroxide values
and aldehydes are analyzed, oxidation appears to be
very low because radicals are transferred before lipid
products are formed. However, production of off-flavors,
browning, texture changes, nutritional losses, etc., con-
tinues, now mediated by other molecules, particularly
proteins:
In some co-oxidations, lipids add to the target mole-
cules, particularly proteins. This limits extractability of
LH L
•
LOO
•
LOOH + L
•
etc (8a)
Protein
•
Protein-OO
•
Protein-OOH (8b)
O
2
Protein
O
2
Amino acid loss
Crosslinking and fragmentation
Inactivation of enzymes
Loss or alteration in functionality
Browning
LIPID OXIDATION: CHEMICAL STABILIZATION 663
lipids for analysis and removes lipids from product
streams normally analyzed, compounding problems
in assessing the true extent of lipid oxidation. Conse-
quently, in systems where co-oxidations are active, lipid
oxidation must be followed by loss of fatty acids in addition
to oxidation products such as peroxides, malonaldehyde,
and anisidine values. Footprints of lipid oxidation in
the form of specific co-oxidation products or molecular
property changes (e.g., protein crosslinking, amino acid
loss, degradation of functionality) must also be tracked
to avoid severely underestimating both the extent and
the effects of lipid oxidation in complex systems.
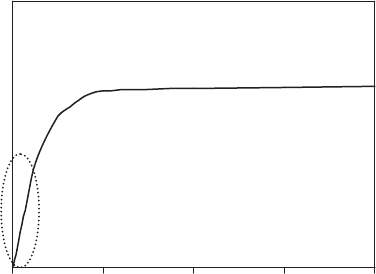
Time Course of Lipid Oxidation
Lipid oxidation is often divided into three rate periods
described in terms of hydroperoxide reactions:
1. Induction period—very low level oxidation, forma-
tion of LOOH undetectable.
2. Monomolecular rate period—initial stages of
oxidation up to B1% oxidation, LOOH accumulate
slowly and decompose as single, isolated mole-
cules when exposed to UV light, metals, or heat
(Reaction 3).
3. Bimolecular rate period—later stages of lipid oxida-
tion up to B7–15% oxidation, LOOH accumulate
rapidly, begin to decompose in pairs via LOOH
(Reaction 4) or LOO
+ LOOH (Reaction 5), and
secondary products form.
The integration and progression of individual reactions
of initiation, propagation, and termination into an overall
oxidation process with these three rate periods is shown
in Figure 3. During the induction and early monomolecu-
lar period, LOO
is the main chain carrier and propaga-
tion is relatively slow. As active oxidation develops
and LOOH decompose, both LOO
and LO
contribute to
propagation. However, LO
reactions are much faster
and more specific, so LO
becomes the dominant
chain carrier and controls directions of reactions in late
monomolecular and throughout the bimolecular rate
periods, partially accounting for increasing oxidation
rates.
FACTORS AFFECTING LIPID OXIDATION: PRO-
OXIDANTS
Many factors alter rates of lipid oxidation. Some of the
most common catalysts and inhibitors are:
(a) Nature of lipids: number of double bonds
i. Configuration of double bonds (trans versus cis
and conjugation)
ii. Level and type of phospholipids present
iii. Free fatty acids versus esters versus
triacylglycerols.
(b) Surfaces: Bulk oil – exposed surface versus emul-
sions versus dispersal on solid surface
(c) Presence of other components:
i. Pro-oxidants
. Preformed hydroperoxides
. Metals
. Porphyrins, chlorophylls, heme compounds
. Lipoxygenase and cyclooxygenase
. Amino acids
. Ascorbic acid (low concentrations)
ii. Antioxidants (endogenous and added)
. Polyphenols
. Amino acids
. Metal chelators and complexers
. Synergists
. Glutathione peroxidase
iii. Interceptors
. Proteins
. DNA
. Vitamins
. Pigments
(d) Environment and solvent system: temperature,
light, oxygen pressures, water, pH, packaging.
Of these, light, oxygen pressure, metals, water, chela-
tors, and antioxidants are amenable to control by
packaging.
Light
Light is a key catalyst of lipid oxidation in foods and
cosmetics, yet is often discounted during handling, proces-
sing, and even storage. At low wavelengths (oB254 nm),
Time of reaction
MB
Extent of reaction
I
O
2
O
2
LH L
•
LOO
•
2 LOOH LO
•
+ LOO
•
LOOH
Secondary
oxidation processes
LOO
•
LO
•
products
LOOH, epidioxide
•
LO
•
+ OH
–
/
•
OH
Figure 3. Changes in dominant lipid oxidation reactions
and products over the course of lipid oxidation. Induction
period (I), monomolecular rate period (M), and bimolecular
rate period (B). [Modified from Schaich (2). Used with
permission.]
664 LIPID OXIDATION: CHEMICAL STABILIZATION

ultraviolet light has enough energy to break bonds and
form L
radicals, although most uv damage to lipids occurs
at wavelengths o200 nm. Much more active is UV-in-
duced decomposition of hydroperoxides to generate two
reactive radicals that rapidly propagate the radical chain:
ROOH RO
•
+
•
OH ROH (9a)
HOOH HO
•
+
•
OH 2 L
2
•
+ H
2
O (9b)
LOOH LO
•
+
•
OH LOH (9c)
2L
2
H
hν(uv)
(ROOH is any organic hydroperoxide, HOOH is hydro-
gen peroxide, LOOH is a lipid hydroperoxide).
Hydrogen peroxide is actively generated in foods by
metal autoxidation, but can also be produced by UV
degradation of aqueous solutions. Light decomposition of
hydroperoxides is a major contributor to destabilization of
stored foods, particularly under fluorescent lights in la-
boratories, storage facilities, and stores.
Visible light (W400 nm) initiates lipid oxidation indir-
ectly through photosensitizers, molecules that absorb
light and transfer the excitation energy to molecular
bonds in lipids to form free radicals directly (Type 1) or
to oxygen to form singlet oxygen
1
O
2
, which then adds to
double bonds and forms hydroperoxides in unsaturated
fatty acids without intermediate radicals (Type 2). Photo-
sensitizers in foods and biological materials are usually
molecules with multiple carbonyls and/or an extended
conjugated double bond system. Most pigments and other
molecules listed below have this required structure.
Chlorophyll, in particular, is a very potent photosensitizer
that produces both free radicals and singlet oxygen, with
the dominant reactions depending on substrate and reac-
tion conditions. Food dyes are listed with a question mark
because many food colors have requisite structures that
make them good candidates for sensitization, but their
action has not yet been documented.
Sensitizers
Quenchers
Type 1: Free Radical
Chlorophyll
Pheophytin
Hemes
Protoporphyrin
Myoglobin
Hemoglobin
Flavins (especially
riboflavin)
Xanthenes
Anthracenes
Anthroquinones
Crystal violet
Food dyes (?)
Phenols, polyphenols
Tocopherols
(carotenes)
Type 2: Singlet
Oxygen
Chlorophyll
Hemes
Erythrosine
Rose bengal
Flavins
Methylene blue
Proflavine
Eosin
Food dyes (?)
Carotenes
Tocopherols
Quenchers are antoxidant compounds that can selec-
tively react with light-induced radicals and singlet
oxygen, thereby preventing their reaction with lipids
and other biomolecules. Carotenes are important
1
O
2
scavengers in foods. They are also radical scavengers,
but listed in parentheses because in high concentrations
they can become pro-oxidants.
Light catalysis is nearly always present, but can be
verified by comparison of samples incubated open versus
in the dark, by oxidation kinetics, and by presence of
specific photolysis products. Type 1 photosensitization is
qualitatively indistinguishable from autoxidation, but
much faster. It characterized by the following patterns:
Mechanism:
Kinetics:
Products:
Free radical
Induction period present
Dependent on pO
2
Rate not p number of double bonds
Relative reactivity:
18:1 B1
18:2 17
18:3 25
Conjugated dienes formed with LOOHs
in external C-9, C-13, and C-16
positions
Scission products all from external
LOOHs
Type 2 singlet oxygen photosensitized oxidation has dis-
tinctively different oxidation patterns:
Mechanism:
Kinetics:
Products:
‘‘ene’’ rx, concerted addition of O
2
to
double bonds to form LOOH without
free radicals; radicals are generated
subsequently by LOOH decomposition
No induction period
Independent of O
2
(when O
2
not limiting)
Dependent on sensitizer concentration
Rate directly p number of double bonds
Relative reactivity:
18:1 B1
18:2 2
18:3 3
Both nonconjugated and conjugated
LOOHs
LOOHs at all positions, high proportion
at internal positions (e.g., C-10 and
C-12)
High proportion of internal cyclics, en-
doperoxides, epoxides, di- and tri-hy-
droperoxides (all are precursors of
light-induced off-flavors)
Scission products from internal LOOHs,
altered scission position preferences
When these kinetic characteristics and products are
observed in products, light-mediated initiation can be
presumed, and protection from light during processing
and with packaging will be critical for stabilization.
Indeed, blanking out light (e.g., by packaging) prevents
both photosensitized generation of LOOH and UV decom-
position of LOOH and thus has huge immediate effects on
LIPID OXIDATION: CHEMICAL STABILIZATION 665
kinetics and consequences of lipid oxidation. However,
caution is required when deducing causes of oxidation or
mechanisms of a given agent from product mixes and
kinetics because many photosensitizers change mechan-
isms with solvent and concentration. For example, ribo-
flavin normally photosensitizes by free radicals but at low
concentrations it converts to the
I
O
2
mechanism. Thus,
sensitivity to light often changes with product formulation
and phase localization and distribution of photosensitizers
and quenchers. For example, cheese will behave differ-
ently than milk under light, even though the fat and
sensitizer composition is similar for each. This needs to
be kept in mind when tracking and comparing causes of
oxidation in different oils, foods, and cosmetic products
and planning protection strategies.
Oxygen Pressure
Lipid oxidation requires oxygen, so removal or limitation
of oxygen has been a traditional approach to inhibiting
lipid oxidation in packaged processed foods. Nevertheless,
effects of oxygen are evident mostly at low oxygen pres-
sures and in early stages of lipid oxidation, so use of low
oxygen to control lipid oxidation must focus on a rather
narrow reaction window.
Since oxygen does not react with lipids directly, lipid
oxidation depends critically on the concentration and
reactivity of initiators that produce reaction sites for
oxygen, either L
by conventional initiators or
1
O
2
by
photosensitization, particularly in the low pO
2
range
(1 to 80–100 mm Hg). Once reaction sites are available,
oxidation rates increase dramatically as pO
2
increases
(Figure 4), but the catalytic effect of oxygen levels off at
pO
2
B 100 mm Hg or when [O
2
]W[LH] and [L
] (exact level
depends on reaction system). Similarly, once enough radi-
cals are present to keep chain reactions going, formation
of new LOO
becomes much less important than hydrogen
abstraction to propagate chains, and the effect of in-
creased oxygen on rate reaches a steady-state equilibrium
as oxidation progresses.
Kinetic rate equations show the complex relationship of
oxygen to lipid oxidation rate. At low pO
2
, the rate of lipid
oxidation may be expressed simplistically as
Rate of oxidation ðlow pO
2
Þ
¼ k
o
ðk
i
=k
t1
Þ
1=2
½LOOH½O
2
ð10Þ
where rate constants refer to L
oxygenation, initiation,
and termination by L
recombinations, respectively. Dur-
ing early oxidation (monomolecular LOOH decomposi-
tion), the rate-limiting step is formation of L
and
peroxyl radicals, which requires oxygen. As oxidation
progresses and LOOH increases, propagation reactions
(i.e., hydrogen abstractions by LOO
and by LO
from
LOOH decomposition) become the dominant rate determi-
nants and the relative effect of oxygen diminishes.
At high pO
2
, more than enough oxygen is present to
add to every L
formed, so the rate dependence shifts from
oxygen to the concentration of available oxidizable lipids
and propagation processes:
Rate of oxidation ðnonlimiting pO
2
Þ
¼ k
p
ðk
i
=k
t3
Þ
1=2
½LOOH½LH
ð11Þ
The overall rate then is determined by the balance be-
tween initiation (k
i
) and peroxyl radical recombinations
(k
t3
), and it is proportional to the total amount and degree
of unsaturation of available lipid plus propagating LOOH
concentrations. The rate-limiting step now is abstraction
of hydrogens to form LOOHs (k
p
).
Hence, low oxygen needs to be combined with mini-
mization of initiators and catalysts to maximize inhibition
of lipid oxidation.
Metals
Redox-active metals are probably the most important
initiators and catalysts of lipid oxidation in oils, foods,
and biological systems because they are ubiquitous and
active in many forms, they exert critical influence on both
initiation and propagation, and trace quantities (nanomo-
lar) are sufficient for effective catalysis. Only metals
undergoing one-electron transfers (e.g., iron, copper,
nickel, cobalt, vanadium, titanium, cerium) are active
catalysts. Metals in their higher valence (M
n+1
) states
are primary initiators: They form L
both directly by
oxidizing double bonds in unsaturated fatty acids (Reac-
tion 12, RCHQCHR is in a lipid chain) and indirectly by
oxidizing other molecules to produce radicals that abstract
hydrogens from unsaturated lipids LH (Reaction 13, R =
lipid or any other oxidizable molecule).
RCH¼CHR þM
ðnþ1Þþ
! RC
HC
þ
HR þ M
nþ
!
LH
L
þ RH
ð12Þ
RH þM
ðnþ1Þþ
! R
þ H
þ
þ M
nþ
!
LH
L
þ RH ð13Þ
Metals in their reduced forms (M
n+
) —for example,
Fe
2+
and Cu
+
—are not good initiators themselves, but
they form reactive species by complexing with oxygen
0 200 400 600 80
0
Oxygen pressure (mm Hg)
Rate of oxidation (arbitrary units)
Target range for packaging
Figure 4. Effects of oxygen on the rate of lipid oxidation. Rate of
oxidation increases rapidly with increasing pO
2
when oxygen is
limiting but is not affected by additional oxygen when [O
2
]W[LH].
[Figure generalized from data of Bolland, (3).]
666 LIPID OXIDATION: CHEMICAL STABILIZATION
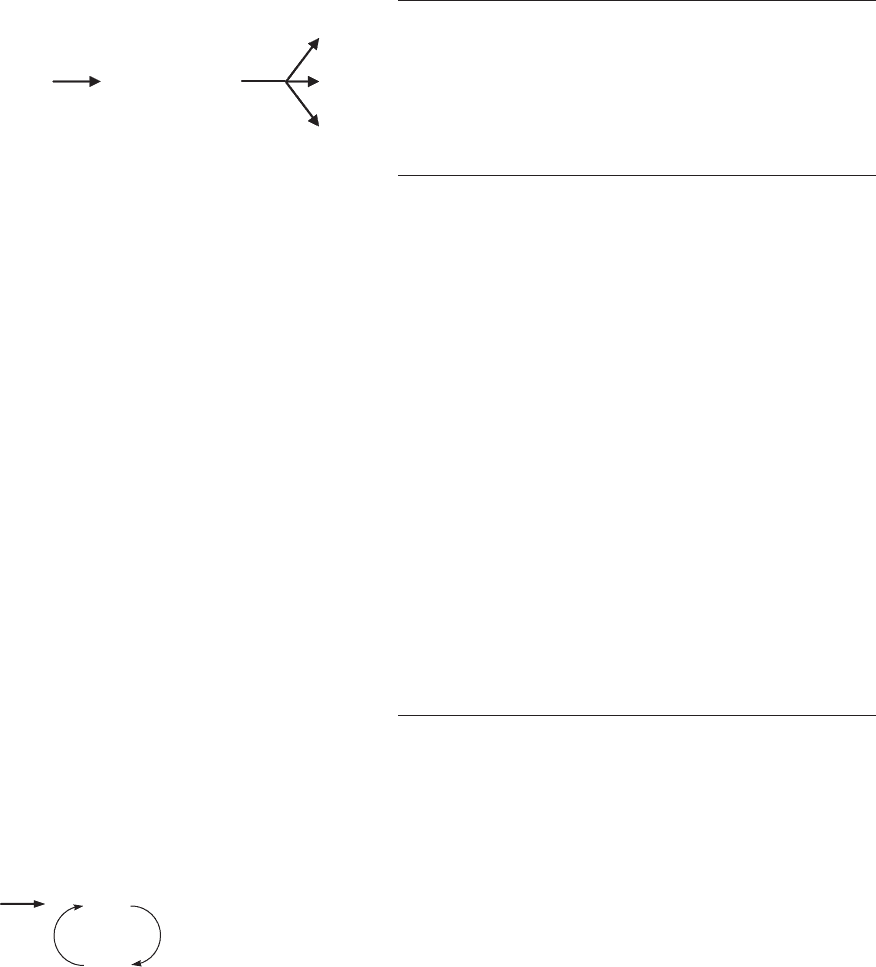
(Reactions 14a–c) and by reducing oxygen to superoxide
anion O
2
, which dismutates to hydrogen peroxide and
very reactive hydroxyl radicals (Reactions 14d–f).
M
nþ
þ O
2
! M
nþ1
þ O
2
ð14dÞ
2O
2
or O
2
=HOO
! H
2
O
2
!
M
nþ
HO
þ OH
ð14eÞ
HO
þ LH ! H
2
O þL
ð14fÞ
Metals also decompose any traces of preformed hydro-
peroxides ROOH present in foods (in lipids or any other
molecule) to alkoxyl radicals (RO
) (Reaction 15a), and
higher valence state metals oxidize hydroperoxides to
peroxyl radicals (ROO
) (Reaction 15b); both of these
radicals initiate lipid oxidation chains. Note that unlike
UV reactions, metal decomposition of hydroperoxides re-
leases only a single reactive radical that reacts with lipids
to generate L
.
Fe
2þ
þ ROOH !
fast
Fe
3þ
þ RO
þ OH
!
LH
ROH þL
ð15aÞ
Fe
3þ
þ ROOH !
extremely slow
Fe
2þ
þ ROO
þ H
þ
!
LH
ROOH þ L
ð15bÞ
When both reduced and oxidized metals are present,
catalysis of lipid oxidation is amplified tremendously by
redox cycling (Reaction 16). Redox cycles occur slowly with
metals and hydroperoxides alone, but rates can be accel-
erated several orders of magnitude in the presence of
appropriate reducing agents, particularly ascorbic acid,
that maintain the faster reacting lower valence state in
metals:
M
n+1
+ ROOH M
n+
+ ROO
•
+ H
+
(16a)
ROOH
M
n+1
+ RO
•
+OH
–
(16b)
The mere presence of metals is not sufficient to guar-
antee catalysis of lipid oxidation. In foods and other
products, metals are complexed to a wide variety of
molecules, including organic acids, carboxylic acids, vic-
inal diphenols, amino acids, and proteins. How the metals
are bound controls the rate of initiation by altering the
availability of orbitals for electron transfer, the redox
potential and oxidizing or reducing capacity, partitioning
between aqueous and lipid phases, and associations with
other molecules. In general, higher redox potentials make
metal complexes better primary initiators, and low redox
potential metal complexes markedly enhance propagation
branching reactions. Selection of appropriate chelators
can thus greatly modify metal reactivity in foods.
Heat
At frying temperatures, there is sufficient thermal energy
to break covalent C–C or C–H bonds in the acyl backbone
and form a variety of lipid alkyl radicals which then start
the radical chains of oxidation. At moderate temperatures,
heat exerts its main effects on processes that have high
activation energies, so they can benefit most from a
thermal boost. The rate constants in the list below refer
to propagation, termination, and LOOH dissociation in
sequence. As can be seen, most steps of lipid oxidation
require little or no activation; only monomolecular and
bimolecular decompositions of hydroperoxides have en-
ergy hurdles. Thus, in early stages of lipid oxidation, low
to moderate heat breaks O–O bonds in traces of ROOH or
LOOH preformed by other reactions, particularly metals,
lipoxygenase, or photosensitizers. The RO
,LO
, and
OH
thus generated abstract hydrogens from neighboring li-
pids to form L
and initiate radical chains. As oxidation
progresses and LOOH accumulate, the major effect of heat
shifts to acceleration of propagation rather than initiation.
Reaction
(L
+O
2
)
k
p
(LOO
+ LH)
k
t
(2 ROO
)
k
t
(2 R
)
k
t
(R
+ ROO
)
*k
d
(monomolecular)
*k
d
(bimolecular)
Activation Energies (E
a
)
0 kcal/mol
B5–15
B4
5
1
31
50 uncatalyzed system
Accompanying faster oxidation kinetics is a heat-in-
duced shift in dominant reaction pathways and resulting
products that markedly affect food and cosmetic quality.
The most important changes include:
. Abstraction of hydrogens from LOH and LOOH in
preference to LH, shifting dominant products from
volatile a and b scission products to W 90% dimers
and polymers.
. Shifts in types of dimers formed: b-scission of peroxyl
oxygen increases - more C–O–C and C–C dimers
and fewer C–O–O–C crosslinks.
. Altered scission patterns; for example, hexanal is
major product of L under mild oxidation conditions,
but 2,4-decadienal becomes dominant when systems
were heated.
L
•
+ [M
(n+1)+
…
–
O
2
H]
M
(n+1)+
…O
2
–
•
]
M
n+
+ O
2
L
•
+ M
n+
+ HO
2
•
(14b)
(14a)
LO
•
+ [M
(n+1)+
…
–
OH] (14c)
LH
LIPID OXIDATION: CHEMICAL STABILIZATION 667
