Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


. Increased proportion of trans isomers, which cer-
tainly has implications under current labeling re-
quirements for trans fats.
Although storage temperatures are always considered,
the changes described above are much more pronounced
at temperatures used in processing than at typical trans-
portation and storage temperatures. Even relatively small
increases in processing temperatures can dramatically
shorten shelf life, as can be seen in the decreased induc-
tion period in oils [from Kaya et al. (4)]:
Induction Period of Fresh Oils (h)
Sunflower oil
Olive oil
1001C
9.0
12.4
1101C
4.4
6.2
1201C
2.2
2.8
Thus, control of temperature effects on lipid oxidation
must be addressed at every stage of production as well as
storage.
Water
No discussion of lipid oxidation is complete without dis-
cussion of moisture effects. As can be seen in the classical
graph of moisture and water activity effects (Figure 5),
lipid oxidation has very different reactivity patterns than
other degradation reactions in foods. Unlike browning,
enzyme reactions, nutrient decomposition, and microbial
growth, which can all be stopped by sufficient removal or
binding of water, there are no conditions under which lipid
oxidation is not active. As would be expected, lipid oxida-
tion is rapid at high moistures and water activity, as are
all reactions (D), but surprisingly, lipid oxidation is just as
rapid in very dry systems (A). The region of lowest lipid
oxidation (B) corresponds to conditions in only a mono-
layer of water molecules is present on molecules in foods.
Moving between regions, water appears to have both pro-
and antioxidant effects on lipid oxidation. This biphasic
action may be explained as follows:
A Dry systems—high oxidation:
Molecular sites of oxidation are bare, pro-
viding ready access of oxygen to lipids.
A-B
B
B-C
C-D
Metals are unhydrated and reactive.
Hydroperoxides are uncomplexed and de-
composed readily.
The food matrix is open and porous, allow-
ing free flow of oxygen
Water binding to molecules provides surface
protection—decreasing oxidation:
Water hydrogen bonds to LOOH, retards
decomposition.
Water hydrates metals, inhibits
electron transfers, and shifts redox
potentials.
Water binding to surfaces of the macromo-
lecular matrix retards oxygen diffusion.
Water attaches to potentially reactive sites,
excluding oxygen.
Monolayer value—lowest oxidation:
Conceptual single water molecules on
molecular surface protects reactive sites
from oxygen and catalysts but is insuffi-
cient for mobilization.
Intermediate moisture region—increasing
oxidation:
Hydration of molecules increases molecular
mobility and reactivity.
Multilayers of water accumulate on mole-
cular surfaces, dissolve catalysts, and in-
crease their diffusion and interaction with
lipids.
Metals mobilized and activated.
Fluid water region—high but decreasing
oxidation:
Water dilutes metals and other catalysts
and reactants.
Emulsion formation separates lipid phases
from most catalysts.
High moisture promotes nonenzymatic
browning
,
products
are
antioxidants.
It is clear from the graph that the moisture content
and water activity (a
w
) of a product dictate an innate
propensity for oxidation of products with unsaturated
fatty acids, over and above effects of metals, heat, light,
0
Relative reaction rate
0.2 0.4
Water activity (a
w
)
0.6 0.8 1.0
Lipid oxidation
Monolayer
B
C
D
A
Non-enzymatic browning
Enzymatic activity
Mold growth
Bacteria growth
Yeast growth
Moisture content (%)
Figure 5. Effects of moisture content and water activity
on rates of lipid oxidation in food products. [Modified from
Labuza et al. (5). Used with permission.]
668 LIPID OXIDATION: CHEMICAL STABILIZATION
and other factors. While few foods or other products move
from one extreme to the other during storage, drying out
and moisture absorption to moderate extents are quite
common and can occur within relatively short times,
leading to rapid changing in lipid oxidation kinetics and
products. A dry high-lipid product stabilized at the mono-
layer a
w
can undergo explosive oxidation when it either
loses or gains moisture and changes a
w
. Similarly, inter-
mediate moisture foods are moderately reactive, but they
can be stabilized by losing moisture or have shelf-life
severely shortened by gaining moisture and increasing
a
w
. Thus, moisture content and degree of water binding by
system molecules (a
w
) must be carefully controlled by
formulation and packaging in order to stabilize foods
and other materials against lipid oxidation.
FACTORS AFFECTING LIPID OXIDATION—
ANTIOXIDANTS
Type 1. Agents that Prevent Free Radical Production
These are metals chelators (the most important of which is
EDTA, ethylenediamine tetraacetic acid, shown below),
metal complexers (e.g., citric acid, polyphosphates, and to
a lesser extent ascorbic acid), and singlet oxygen scaven-
gers, particularly carotenoids. The conjugated double
bonds of carotenoids provide many sites that preferen-
tially react with
1
O
2
and spare fatty acids.
EDTA
Carotenoids
1
O
2
HO O C -CH
2
HO O C -CH
2
N-CH
2
-N
CH
2
-COOH
CH
2
-COOH
MM
Initiation inhibitors are commonly overlooked as
antioxidants but serve perhaps the most critical role
because they control the radical load that must be over-
come by free radical scavengers. The radical load is
the cumulative total of radicals from all sources that
must be quenched to seriously limit progression of lipid
oxidation. The radical load determines the type, amount,
and reactivity of antioxidant necessary to stabilize a given
system. If the radical load is too high, no amount of radical
chain stopper—what is usually thought of as antioxi-
dants—can prevent lipid oxidation. At the same time,
initiation inhibitors cannot work alone. Some radicals
from various sources are always produced, and these
must be quenched to prevent establishment of cycling
chain reactions. Thus, Type 1 antioxidants are usually
combined with Type 2 antioxidants for improved all-round
efficiency.
Type 2. Free Radical Chain Stoppers
These are radical quenchers that donate hydrogens to
LOO
or LO
faster than lipid chains:
LOO
þ AH ! LOOH þ A
R
ð17aÞ
LOO
þ AH
2
! LOOH þAH
! A þRH ð17bÞ
For a compound to be an antioxidant, its radical A
must
be stable and unreactive or yield nonradical products, so it
does not propagate radical chains. The dominant antiox-
idant structures with H-transferring activity are phenolic
compounds in which the A
reactivity is reduced by
resonance delocalization of the free electron over the
aromatic ring.
O
O
O
O
The standard synthetic antioxidants BHA, BHT, and
propyl gallate are all phenolic compounds, as are natural
antioxidant tocopherols, flavonoids in fruits, and many
herb and spice components, to mention just a few. Struc-
tures of some common phenolic antioxidants are shown in
Figure 6.
Some antioxidants have more than one phenolic group
per ring. In general, antioxidant activity increases with
the number of phenolic groups on a single ring and per
molecule in complex structures. Phenols with multiple –
OH groups next to each other on the aromatic ring also
complex metals, so they have multiple mechanisms for
inhibiting lipid oxidation.
M
n
OH
OH
HO
HO
OH
OH
M
n
Type 3: Environmental Factors that Limit Oxidation
Environmental factors are not initiators or catalysts
of lipid oxidation in themselves, but they provide condi-
tions under which oxidation reactions occur, and thus they
play critical roles in controlling the rate and direction
of lipid oxidation as well as the resulting product
mix during storage. Indeed, if environmental factors are
ignored, no amount of primary antioxidant will be suffi-
cient to stabilize a food or other material against lipid
oxidation.
Environmental factors that can influence lipid oxida-
tion encompass a wide variety of conditions to which
a product is exposed in moving from formulation through
processing to storage. Formulation factors include
LIPID OXIDATION: CHEMICAL STABILIZATION 669
moisture levels and pro-oxidant enzymes such as lipoxy-
genase and lipase that require blanching to inactivate.
Processing environment factors that are important in-
clude everything from potential metal contamination
from equipment and processing water to temperature,
light, and oxygen exposure during processing. Packaging
and storage environments include similar factors: pO
2
inside the package and surrounding it, vacuum versus
inert atmosphere versus air headspace, moisture inside
the package and migrating into or out of it, temperature
and light exposures, and so on. Control of environmental
factors to limit or prevent lipid oxidation during storage
must be a major focus for packaging of foods.
WHAT ROLE DOES PACKAGING PLAY IN
CONTROLLING LIPID OXIDATION?
Successful strategies for stabilizing any product against
lipid oxidation start with product components and formu-
lations that first minimize endogenous catalysts, then
incorporate chelators, antioxidants, and
1
O
2
quenchers
to deactivate those that cannot be eliminated. Then add
processing that protects at every step (e.g., by use of
lowest feasible temperatures and light exposure), as well
as limitation of oxygen via nitrogen blankets or vacuum.
Packaging is the critical finishing step that maintains
product quality during storage. Packaging cannot cure
deficiencies of formulation and processing, but the storage
environment it provides largely determines what reac-
tions occur and how long products are stable during
storage.
Packaging barrier properties and delivery modes con-
tribute to control of lipid oxidation at all three stages:
initiation, propagation and chain branching, and termina-
tion. Different reactions dominate and different factors
affect each stage, so it is not likely that a single protective
approach will meet the demands of all stages. Here, the
concept of radical load must be highlighted. To be success-
ful, any antioxidant or protective function must deal with
the total lipid radical load generated from all sources in
formulation, processing, and packaging. If the total radi-
cal load is extremely high, no single approach can elim-
inate the oxidation. Thus, two general guidelines should
be followed to efficiently limit lipid oxidation:
1. When designing antioxidant strategies, emphasize
limitation of initiation. Radicals not generated do
not propagate chains or need to be quenched.
2. Use combinations of antioxidant approaches that
address action at each stage of lipid oxidation and
that act on multiple aspects of lipid oxidation
simultaneously.
Table 2 shows the multiple functions that packaging
provides to limit lipid oxidation during storage and the
various approaches that can be used and combined to
achieve them. Limitation of initiation is especially critical
for products requiring significantly extended shelf lives, so
most emphasis is placed on this stage. The light and
oxygen barriers of traditional packaging materials such
as glass, tin cans, and some polymers provide an essential
primary line of defense against lipid oxidation, preventing
or severely limiting levels of L
and LOO
produced during
3,5-Di-tert-butyl-4-hydroxytoluene
(BHT)
3-t-butyl-4-hydroxyanisole
(BHA)
Propyl gallate
(PG)
HO
O
CH
3
H
3
C
H
3
C
H
3
C
CH
3
CH
3
CH
3
CH
3
CH
3
α-Tocopherol
(highest biological activity)
HO
O
H
3
C
CH
3
CH
3
CH
3
CH
3
CH
3
δ-Tocopherol
(highest antioxidant activity)
OH
C(CH
3
)
3
(CH
3
)
3
C
OH
OCH
3
OH
CO
2
CH
2
CH
2
CH
3
OH
HO
OH
HO
H
3
CO
OH
O
OCH
3
Curcumin (turmeric) Gingerol (ginger)
O
HO
HO
OH
OH
OH
OH
Epigallocatechin
(
g
reen tea)
Carnesol
(rosemary)
Sesamol
(sesame seed)
Eugenol
(cloves)
HO
OH
O
HO
CH
3
H
3
C
O
C
HO
H
3
CO
OH
O
OH
O
O
OCH
3
OH
CH
2
CH=CH
2
C(CH
3
)
3
Figure 6. Structures of some common
antioxidants. The top row shows synthetic
antioxidants, whereas the other rows are
antioxidants from natural materials.
670 LIPID OXIDATION: CHEMICAL STABILIZATION

Table 2. Packaging Actions and Approaches for
Controlling Initiation, Propagation, and Termina
tion of Lipid Oxidation
Oxidation Stage and Action
Necessary Packaging Action
Control Factor
Packaging Approach
Examples
Initiation
Oxygen
Provide and maintain inert atmosphere Oxygen
Remove or replace oxygen Hermetic packaging with
vacuum,
deaeration, nitrogen headspace
Oxygen barrier, prevent LOO
formation
Impermeable materials Glass, EVOH, polya
mide, PVDC
Laminants with barrier
layers
Metal foils and oxides, hydrocarbon
films, whey protein isolates,
Starch films, hydroxy-functional
hyperbranched polyester films
Barrier coatings
Hydrocarbon films on PET
Polymer multilayers w/wo
additives
Montmorillonite clay nanoparticles in
PET, PVDC/PE
O
2
scavenger in package or film, remove
residual oxygen
Oxygen sorbent sachets,
labels, lid liners
Elemental or ferrous iron, cobalt-fatty
acid salts in olefin plastics
Oxygen sorbent packaging Fe
2+
, ascorbic acid, glucose-glucose
oxidase, Co(II) acetate,
PP in food contact layer
Light
Block light access
Light
Light-impermeable materials Colored glass, cans
Impermeable laminant
layers
Metal laminant layers
Light filter
Plastics or coatings with UV
blockers
Hydroxyaromatic compounds,
hindered amines, salicylates
Metals
Remove metals from package contents Chelators
Controlled release packaging EDTA, citric acid
, amino acids
Metal-bonding layers
Chitosan, N-carboxymethyl chitosan
Radicals
Deliver radical quenching antioxidants Antioxidant
levels
Controlled release packaging Tocopherol, sesamol, BHT
, BHA,
TBHQ
Propagation
H abstraction
Deliver radical quenching antioxidants
over time
Antioxidants
Controlled release packaging Tocopherol, ses
amol, BHT, BHA,
TBHQ
LOOH decomposition
Light barrier (UV and visible):
Light
See light in Initiation
prevent decomposition of LOOH
-
branching
(Continued
)
671

Table 2. Continued
Oxidation Stage and Action
Necessary Packaging Action
Control Factor
Packaging Approach
Examples
Moisture barrier:
Moisture
Impermeable materials Glass, oriented
PP, metallized
polyester films
Prevent dehydration and exposure of
reactive sites
Laminants with barrier
layers
Polyamide layers, zein, starch, soy
protein/wheat gluten,
Slow molecular migration
Propylene glycol midlayer between
PVA sheets
Stabilize LOOH
Impregnated or coated
papers
Polyolefins, waxes, starch acetates,
acrylic-based latex,
Limit scission reactions that release
volatiles
Polyvinyl alcohol
Maintain low oxidation rates
Moisture sorbers
Silica gel, clays, encapsulated polyvinyl
alcohol
Temperature maintenance, prevent
LOOH decomposition
Heat
Insulation
Biofoams
Regeneration of LOO
Oxygen barrier, limit L
-LOO
Oxygen
See oxygen in Initiation
Termination
Radical recombination Oxygen barrier
, limit oxydimer and
polymer formation
Oxygen
See oxygen in Initiation
Intercept radical reactions
Antioxidant levels
Controlled release packaging Tocopherol, sesamol,
BHT, BHA,
TBHQ
LO
scission
Moisture barrier: prevent moisture
sorption
Moisture
See moisture in Propagation
Limit LO
scission
Inhibit generation of volatile off-flavors
and odor compounds
Secondary oxidation
reactions
Oxygen barrier, limit oxidation to a few
major products
Oxygen
See oxygen in Initiation
672
storage. This, in turn, allows fewer oxidation chains to be
generated and fewer radicals to be propagated, greatly
reducing the amount of antioxidant needed to quench the
radicals present. Innovative packaging that can deliver
active compounds such as UV absorbers, singlet oxygen
scavengers, metal chelators, or phenolic antioxidants sup-
plements traditional packaging and offers new opportu-
nities for creative combinations of approaches that
increase overall antioxidant effectiveness.
Barrier properties of packaging also limit progression
of lipid oxidation, particularly by excluding light that
decomposes hydroperoxides and by controlling moisture
loss that destabilizes LOOH; also, moisture gain mobilizes
catalysts. Radical propagations are particularly active
during this period, so exclusion of oxygen remains very
important, and here the advent of oxygen scavenging films
and sachets have added exciting new opportunities to
remove residual oxygen that remains in packages after
processing and fuels oxidation during storage. Caution is
warranted, however, when using iron-based oxygen sca-
vengers: Any leaching of molecular or solid iron into the
package can catalyze rapid lipid oxidation. New con-
trolled-release packaging is being developed to deliver
either volatile antioxidants such as BHT (synthetic) or
sesamol (natural) or nonvolatile antioxidants such as
mixed tocopherols slowly over time as endogenous anti-
oxidants are consumed. Combining controlled release with
high barrier packaging greatly increases the effectiveness
of packaging, protecting food quality and significantly
extending shelf life.
Although usually overlooked, barrier properties of
packaging also play important roles in directing the types
of products formed during lipid oxidation. Oxygen and
moisture, in particular, favor formation of volatile break-
down products that are responsible for the off-odors and
flavors associated with rancidity; these volatile products
are immediately noticeable when the package is opened
and can lead to consumer rejection. Packaging delivery of
radical scavengers in this stage, where antioxidants natu-
rally present in the food or added in the formulation have
already been consumed, can have a major impact on
limiting secondary oxidations and polymerizations that
markedly degrade product flavor and aroma, color, and
texture, and nutrition.
Unfortunately, some questionable practices are also
developing. Because the off-odors of lipid oxidation are
one of the first signs of trouble when a package is opened,
and a bad whiff can lead a consumer to dump the product
directly, there is currently intense interest in incorporat-
ing carbonyl scavengers in packaging to bind and remove
volatile odor products of lipid oxidation. However, there
are serious legal issues with this practice because FDA
regulations prohibit any practices that cover up faulty
practices or decomposing foods. Rancid off-odors definitely
indicate product deterioration and the possible presence of
toxic products. Removing them eliminates consumers’ first
indicator of trouble and is tantamount to covering up
spoiled food. Both short-term and long-term toxicity issues
with oxidized lipids remain to be clarified, but all evidence
points to the need to avoid ingestion of oxidized lipids
through foods, nutraceuticals, or cosmetic lotions.
SUMMARY
Lipid oxidation is indeed a complicated reaction that plays
havoc with stability of any product or material containing
lipids. The multiple pathways and products can sometimes
seem overwhelming and impossible to control. Neverthe-
less, there is also logic and organization in lipid oxidation
that, with careful consideration, make it possible to moni-
tor, understand, and control in a wide range of products.
Packaging alone cannot prevent lipid oxidation, but no
product can be stabilized against lipid oxidation without
appropriate and effective packaging. By providing barriers
against oxygen, light, and moisture, packaging works in
concert with endogenous antioxidants to limit the total
radical load in foods, cosmetics, and other products so
that primary antioxidants can be more effective at lower
concentrations; these barriers also provide an environment
that greatly slows down radical reaction chains that do
become established. New active packaging that delivers
radical scavengers, metal chelators, and singlet oxygen
quenchers offer unique opportunities for tailoring packa-
ging to individual product compositions and increasing
antioxidant effectiveness. Combining barrier and delivery
properties of packaging provides flexible options for protect-
ing product quality and significantly extending shelf life.
BIBLIOGRAPHY
1. T. P. Labuza, ‘‘Kinetics of Lipid Oxidation in Foods,’’ CRC Crit.
Rev. Food Sci. Nutr. 2, 355–405 (1971).
2. K. M. Schaich, ‘‘Lipid Oxidation in Specialty Oils’’ in F.
Shahidi, ed., Nutraceutical and Specialty Oils, Marcel Dekker,
New York, 2005, pp. 401–448.
3. J. L. Bolland, Quaert. Rev. 3, 1–21 (1949).
4. Kaya et al., Lebensm. Wiss. Technol. 26 , 464–468 (1993).
5. T. P. Labuza et al., Food Technol. 24, 543–544, 546–548, 550
(1970).
Selected References for General Background on Lipid
Oxidation Processes
W. O. Lundberg, ed., Autoxidation and Antioxidants. Vol. 1 and 2,
Interscience, New York, 1961.
G. Scott, Atmospheric Oxidation and Antioxidants, Elsevier,
London, 1965.
H. W.-S. Chan, Autoxidation of Unsaturated Lipids, Academic
Press, London, 1987.
E. N. Frankel, ‘‘Volatile Lipid Oxidation Products,’’ Prog. Lipid
Res. 22, 1–33 (1982).
E. N. Frankel, ‘‘Lipid Oxidation: Mechanisms, Products, and Biolo-
gical Significance, JAm.OilChem.Soc.61, 1908–1917 (1984).
E. N. Frankel, ‘‘Chemistry of Free Radical and Singlet Oxidation
of Lipids,’’ Prog. Lipid Res. 23, 197–221 (1985).
N. A. Porter, ‘‘Mechanisms for the Autoxidation of Polyunsatu-
rated Lipids,’’ Acct. Chem. Res. 19, 262–268 (1986).
E. N. Frankel, ‘‘Secondary Products of Lipid Oxidation,’’ Chem.
Phys. Lipids 44, 73–85 (1987).
J. P. Cosgrove, D. F. Church and W. A. Pryor, ‘‘The Kinetics of the
Autoxidation of Polyunsaturated Fatty Acids,’’ Lipids 22, 299–
304 (1987).
LIPID OXIDATION: CHEMICAL STABILIZATION 673
J. C. Allen and R. J. Hamilton, Rancidity in Foods, 2nd edition,
Elsevier Applied Science, London, 1989.
H. W. Gardner, Oxygen radical chemistry of polyunsaturated
fatty acids, Free Radic. Biol. Med. 7, 65–86 (1989).
N. A. Porter, S. E. Caldwell, and K. A. Mills, ‘‘Mechanisms of Free
Radical Oxidation of Unsaturated Lipids,’’ Lipids 30, 277–290
(1995).
K. M. Schaich, ‘‘Free Radical Initiation in Proteins and Amino
Acids by Ionizing and Ultraviolet Radiations and Lipid Oxida-
tion—Part III: Free Radical Transfer from Oxidizing Lipids,’’
CRC Crit. Rev. Food Sci. Nutr. 13, 189–244 (1980).
K. M. Schaich, ‘‘Metals and Lipid Oxidation: Contemporary
Issues,’’ Lipids 27, 209–218 (1992).
K. M. Schaich, ‘‘Lipid Oxidation in Fats and Oils: An Integrated
View’’ in F. Shahidi, ed., Bailey’s Industrial Fats and Oils, 6th
edition, John Wiley & Sons, New York, 2005, pp. 2681–2767.
E. N. Frankel, Lipid Oxidation, 2nd edition, The Oily Press, PJ
Barnes & Associates, Bridgewater, UK, 2005.
References for Specialty Packaging Approaches
M. L. Rooney, Active Food Packaging; Blackie Academic & Profes-
sional, New York, 1995, p. 260.
A. L. Brody, E. R. Strupinsky, and L. R. Kline, Active Packaging for
Food Applications, Technomic Publishing: Lancaster, PA, 2001.
LIQUID-CRYSTALLINE POLYMERS,
THERMOTROPIC
ROBERT R. LUISE
Hytem Consultants, Inc.
Boothwyn, Pennsylvania
Main-chain thermotropic or melt-processible liquid-crystal-
line polymers (TLCPs) are a class of high-performance
condensation polymers represented mainly by para-ex-
tended aromatic polyesters (1). Although these polymers
were invented in U.S. industrial research laboratories in
the 1970s, development was inhibited until the 1980s by
high monomer costs and the lack of attractive markets.
With the emergence of a market niche in molded electronics
packaging and enhanced prospects for lower monomer
costs, rapid 20–25% annual growth of the TLCP market
currently estimated at 10–15 million lb is projected for the
remaining 1990s (2).
TLCP property advantages include high thermal, elec-
trical, and solvent resistance combined with exceptional flow
(low-melt viscosity) in the 3001C (5721F) range, precision
moldability, and dimensional stability; these features are
ideal for injection- molded, close-tolerance applications such
as thin-walled electrical connectors that constitute about
90% of the present market. In film form, the dense uniaxial
LC morphology affords excellent barrier properties, but
development in packaging applications has been inhibited
by high resin costs and the need for special technology to
prepare balanced film. With lower resin costs and recent
progress in balanced and coextruded film technology, most
notably using counter-rotating dies (3), medical and food-
packaging applications are expected to emerge rapidly. In
addition, the unique post-heat-strengthening capability of
these materials by solid-phase polymerization of the final
part results, for example, in high-strength fibers with the
mechanical properties of Kevlar aramid fiber (4).
FUNDAMENTALS
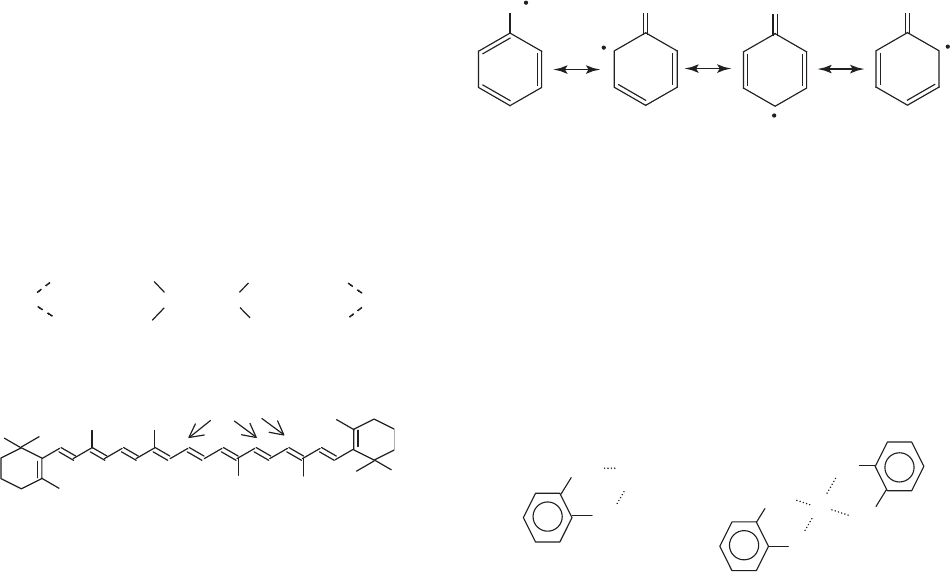
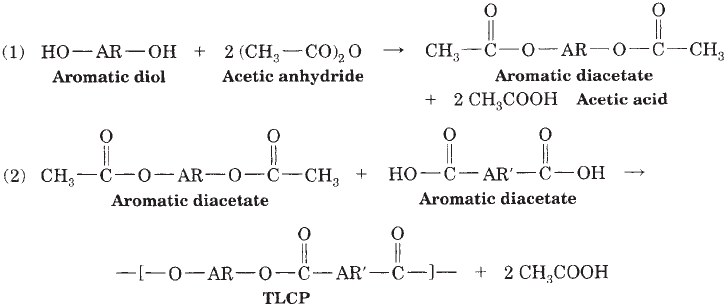
TLCP chemistry is relatively simple and involves reaction
of an acetylated aromatic diol with an aromatic dicar-
boxylic acid, as illustrated in Figure 1. The diol is usually
acetylated in situ with acetic anhydride at 150–2001C
(300–3921F) under nitrogen in the presence of the diacid,
and the melt polymerization finished under vacuum at
higher temperatures (3001C, or 5721F range) over a period
of several hours. Acetic acid is the principal reaction
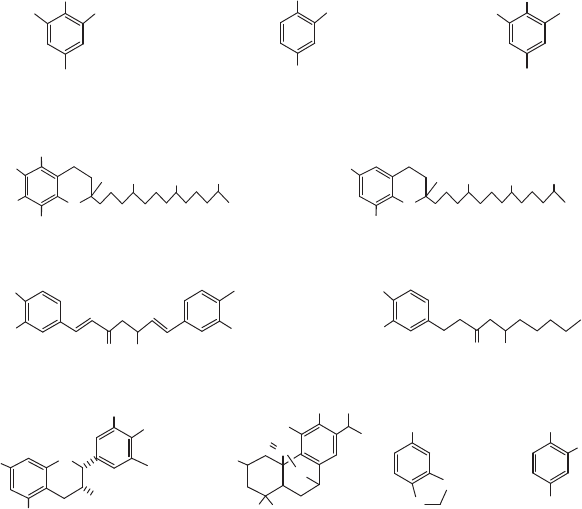
byproduct. Principal monomers used in commerical mate-
rials shown in Figure 2 are para-oriented, phenyl or
naphthyl rings preferred, but a certain amount (r40
mol%) of non-para or ‘‘soft’’ comonomers (e.g., isophthalic
acid, ethylene glycol) can be tolerated without destroying
liquid crystallinity. Other linkages or colinkages are also
feasible, e.g., ester–amide, ester–imide, carbonate, and
azomethine, but the predominant linkage is ester.
The rodlike character of the extended chain molecules
results in a preference for parallel packing in the melt,
Figure 1. Melt polymerization.
674 LIQUID-CRYSTALLINE POLYMERS, THERMOTROPIC
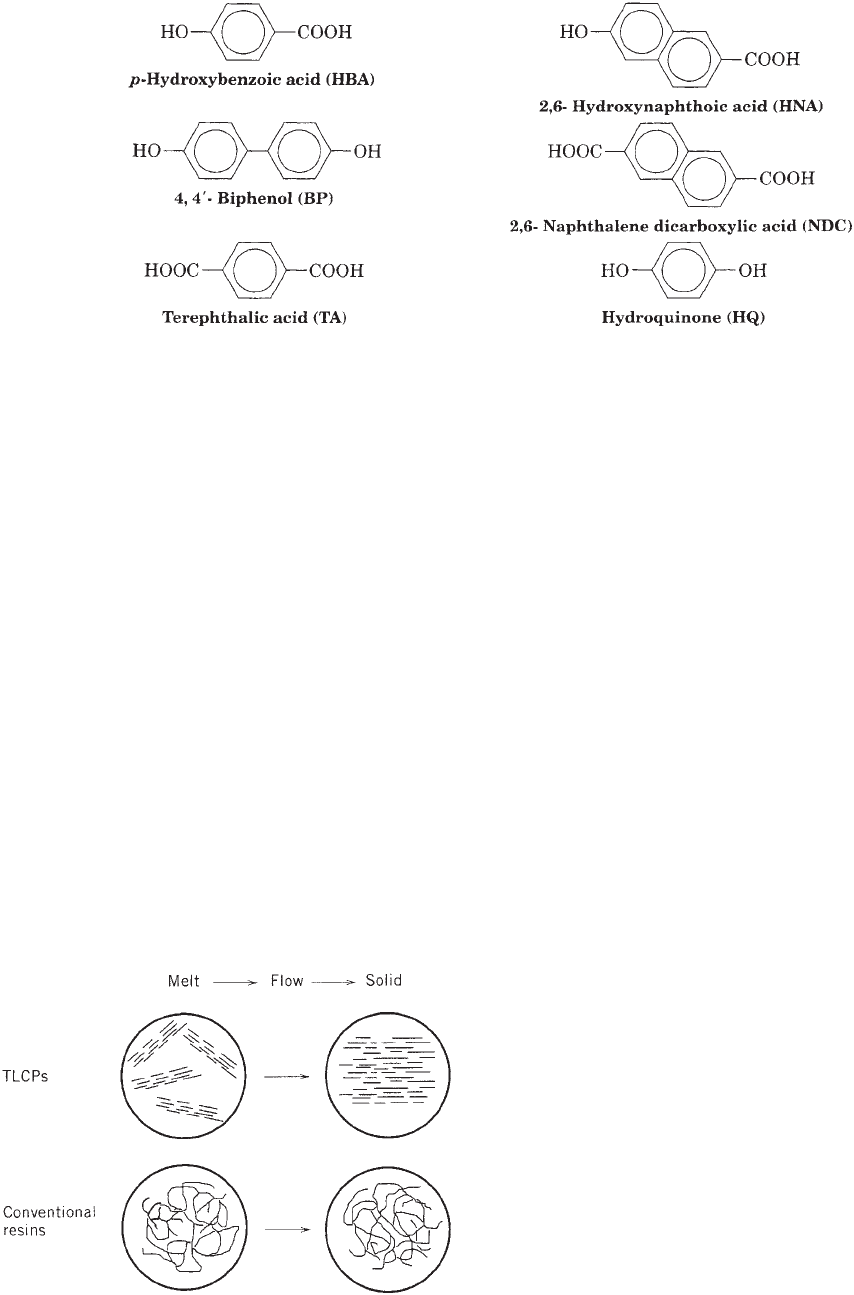
driven by entropy, much like ‘‘logs in a stream.’’ Packing
occurs in the form of optical-size birefringent domains that
contain uniaxially oriented polymer chains, which are
randomly oriented with respect to one another. At flow
conditions, the domains easily orient with respect to each
other, which frequently results in anisotropic properties in
the final part. This result can be an advantage in some
structural materials (e.g., fibers), but a disadvantage in
others that require balanced properties (e.g., films). Differ-
ences in flow behavior of liquid crystalline and conventional
or flexible chain polymers are illustrated in Figure 3. This
type of simple uniaxial ordering is referred to as a ‘‘ne-
matic’’ mesophase in LC terminology and is predominant
for TLCPs. The use of optically active comonomers can also
lead to chiral stacking of domains and the ‘‘cholesteric’’
mesophase in the melt (5).
COMMERCIAL SOURCES
Major commercial TLCPs include Vectra (Hoechst-
Celanese), Xydar (Amoco), and Zenite (DuPont). All are
high-temperature aromatic polyesters with heat-distor-
tion temperatures in the 240–2701C (464–5181F) range
and are glass-filled, geared mainly to injection-molded
applications in electronics. Vectra is a 73/27 copolymer of
p-hydroxybenzoic and hydroxynaphthoic acids, Xydar a
50/25/25 copolymer of p-hydroxybenzoic acid, 4,4u-biphe-
nol and terephthalic acid (and optionally a small amount
of isophthalic acid) developed initially by Carborundum in
the 1970s and Zenite, an all-aromatic polyester. Vectran is
a high-performance TLCP fiber based on the Vectra
composition manufactured by Kuraray in Japan under
license from Hoechst-Celanese. Worldwide capacity of
these materials is estimated at 15–20 million lb;
Hoechst-Celanese/Polyplastics and DuPont have recently
announced new plants in Japan and Tennessee, respec-
tively, onstream in 1995. Rodrun, a lower-temperature
TLCP based on PET (20–40%) and p-hydroxybenzoic acid
developed initially by Eastman in the 1970s, is also
available in limited quantities from Unitika–Japan.
TLCP cost is presently high: $5–$11/lb for glass- or
mineral-filled molded grades and $12–$22/lb for extrusion
grades; resin cost, however, is expected to approach the
$3–$5/lb range over the next several years (2), which
results lower prices projected for key monomers such as
2,6-naphthoic acid, also a monomer for PEN and now
available from Amoco at $1.50/lb, and economies-to-scale
price reductions expected for p-hydroxybenzoic acid (to
$1.00/lb range from $2.50/lb present) and 4,4u-biphenol (to
$2.00/lb from $5–$7/lb).
FILMS
Processing. TLCP ease-of-flow orientation results in
unbalanced films with poor lateral properties by conven-
tional film extrusion that are generally not poststretchable.
Kuraray has reported reasonably balanced Vectra-type
films by conventional film blowing at high shear rates (6),
but the best results have been obtained by Superex, which
uses a combination of film-blowing and counter-rotating
extrusion dies (3). In the latter case the annular die consists
of counter-rotating inner and outer mandrels that impart
an opposing biaxial or ‘‘crisscross’’ orientation to the respec-
tive inner and outer film surfaces. Stretch is controlled by
gas blown through the center and attenuation/takeup
Figure 2. TLCP monomers.
Figure 3. Flow behavior of TLCPs versus conventional resins.
LIQUID-CRYSTALLINE POLYMERS, THERMOTROPIC 675
speed. The latter technology, also applicable to tubing, is
available from Superex by special licensing. Another ap-
proach reported in patents by Nippon Oil involves conven-
tional extrusion of cholesteric TLCP film with improved
property balance through the use of chiral (optically active)
comonomers that results in a biaxial twist or chirality to the
morphology in the film plane (7).
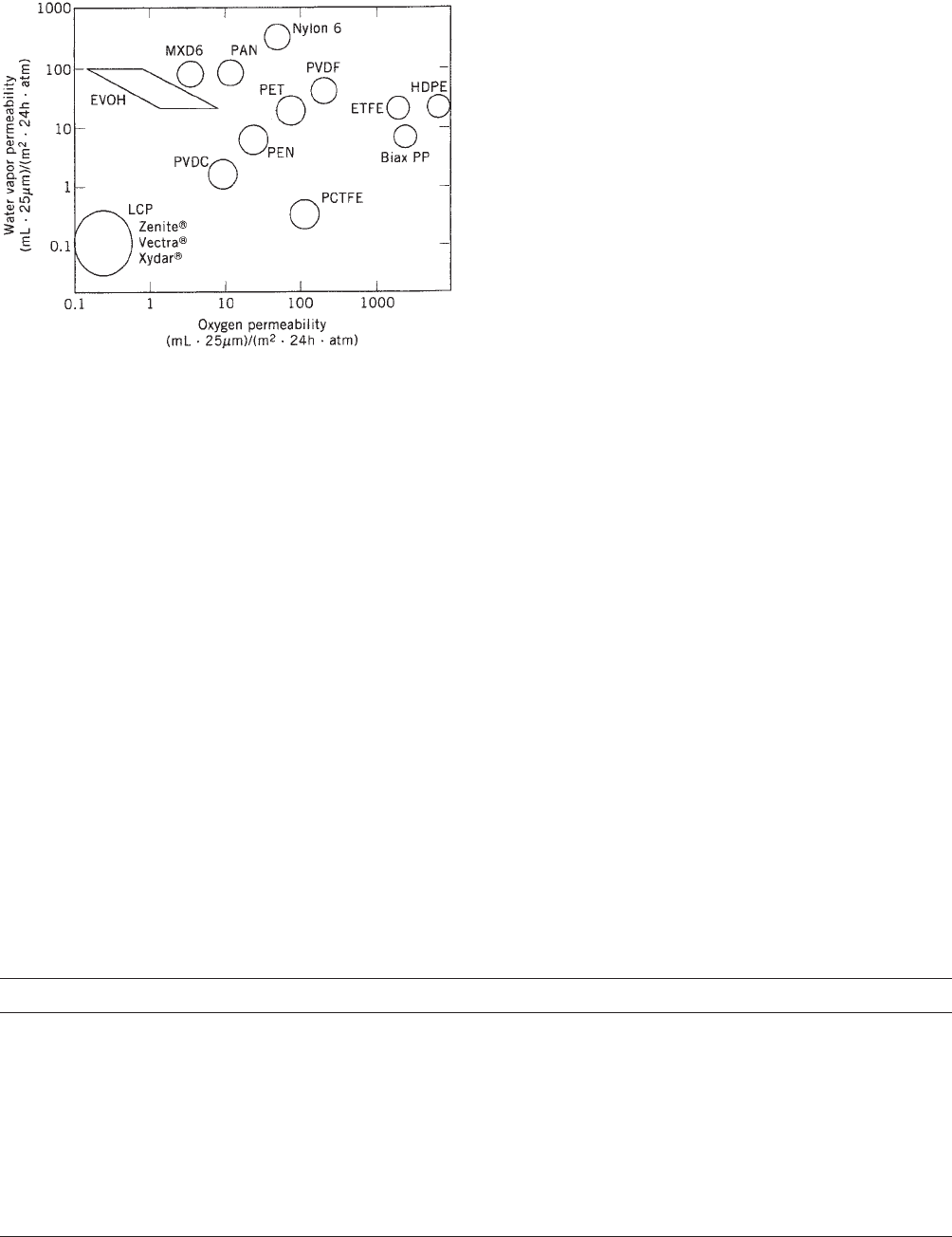
Properties. TLCP films generally possess exellent per-
meability resistance to gases (e.g., oxygen, water vapor,
and carbon dioxide), which is attributed to the highly
compact uniaxial morphology. A comparison of permeabil-
ity of various polymer films in Figure 4 illustrates the
unique barrier resistance of TLCPs to both oxygen and
water vapor, which far exceeds other packaging resins.
Film studies attribute low permeability to low solubility
indicative of a highly ordered substrate (8); moderate
reductions in oxygen permeability were observed (9)
with orientation (45%) and solid-phase polymerization
(10–20%). Larger reductions in oxygen permeability
(200–300%) have been observed by Kuraray in studies of
aliphatic—aromatic TLCPs based on PET and PEN copo-
lymers with HBA and HNA (10), whereby permeability is
found to decrease with increasing naphthyl (vs phenyl)
content, probably due to decreased solubility. These par-
tially aliphatic materials also were found to be post-
stretchable biaxially and, in some cases, transparent.
Wholly aromatic TLCP films (e.g., Vectra), in contrast,
are not poststretchable and are opaque, except in the case
of ultra-high-oriented (‘‘monodomain’’) film prepared from
uniaxial layups of TLCP fiber (11).
Applications. A comparison of the general properties of
balanced TLCP Vectra film with other high-performance
polyester (PET, PEN) and polyimide (Kapton, Upilex)
films is shown in Table 1. In contrast to polyimide,
TLCP appears to offer similar high-temperature and
electrical properties with lower moisture absorption and
lower cost, for electrical applications in flexible-film sub-
strates. Compared to polyester, TLCP offers higher ther-
mal, mechanical, and barrier properties for medical and
food-packaging applications that require sterilizing, re-
torting and long shelf life. In the latter case, even at a
resin cost of $20/lb, TLCP offers about five times better
cost-barrier performance (oxygen, water vapor) vs PET
and PEN, assuming resin costs of $1 and $4/lb for the
latter polymers (12).
Also noted is the high-use temperature capability of
TLCP film [B2001C(B3921F)] despite relatively low glass
temperature [110–1251C(230–2551F)], attributed to the
crystalline uniaxial morphology. TLCPs with glass tem-
peratures in the 2001C(3921F) range that may have future
application in packaging have recently been disclosed (13).
A TLCP drawback has been the lack of availability of
balanced film in the marketplace, but this may change
very soon. Superex has recently announced a number of
consortium activities targeted for the marketplace, which
include development of a copper-clad balanced film for
electronics multilayer circuit boards and multichip mod-
ules (14), and coextruded TLCP and PET film and tubing
for electronics, medical, and food applications that require
high temperature [2051C (4001 F)] resistance (15). The
latter features a tie layer, probably of a PET/TLCP blend,
to improve heat-sealing and formability. A one million-
pound production level is expected by 1997 and cost will be
competitive with EVOH and PVDC. A possible application
is a shelf-stable, retortable food tray. Superex also has
Figure 4. Permeability of selected polymer films. (Courtesy of
Superex Polymer, Inc.)
Table 1. Properties of Balanced Films
Vectra TLCP PET(ICI) MD/TD Kaladex PEN Kapton PI Upilex S PI
Tensile strength, MPa 345 165/228 220 90 413
Tensile modulus, GPa 8.3 4.1/4.8 6.0 2.6 8.9
T
g
/T
m
, 1C 110–125/280 72/269 120/262 385/none W500/none
Upper use temperature, 1C 180–200 130 150 200 260
CTE, ppm/C 5 to + 9 23/18 20 22 2.6
Moisture absorption, % 0.02 o0.1 2.1 1.0
Solvent resistance Excellent Good Good Good Excellent
Relative permeation, O
2
/H
2
O 1/1 100/100 25/30
Dielectric constant at 1 kHz 3.3 3.2 3.4 3.5
Dissipation factor at 1 kHz 0.003 0.005 .002 0.0013
Resin/film price, $/lb 12–22/44 1/2–6 4/10–19 –/55 –/60
Source: Courtes y of The PST Group.
676 LIQUID-CRYSTALLINE POLYMERS, THERMOTROPIC
announced consortium development of a PET/TLCP alloy
with twice the oxygen- and moisture-barrier resistance of
PET at 10% TLCP loading (16). Flexible-to-rigid mono-
layer or coextruded packaging that is both recyclable and
reusable should be feasible. Projected price is $3/lb with
up to 50% package downgauging possible. A key to the
PET alloy technology is the preservation of the TLCP
barrier phase using special compatibilizers to control ester
interchange. In addition to film packaging, Superex TLCP
biaxial tubing is now being manufactured under license by
ACT Medical for endoscopic surgical instruments (17).
Advantages include five times higher stiffness vs conven-
tional tubes and 50% cost savings over fiber-reinforced
composite tubes. Superior electrical properties and broad
sterilization capabilities are also exhibited. Superex’ biax-
ial TLCP tubing was a 1995 Award Winner selected by
R&D Magazine as one of the top 100 technologically
significant products of the year.
Neste Oy (18) has also announced development of
coextruded LDPE/TLCP barrier film based on a new low-
melting TLCP [210–2601C (410–5001F)] with heat-distor-
tion temperature of 1481C(3001F). The blend includes a
compatibilizer to improve heat sealing and processibility.
Multilayer coextruded and paper-coated constructions
with LDPE have been prepared by conventional proces-
sing. Special biaxial processing is not required, apparently
because films are less anisotropic because of the higher
flexibility of the TLCP (perhaps due to aliphatic linkages).
Three- and five-layer constructions with LDPE far exceed
oxygen-barrier cost performance of similar PET construc-
tions and are competitive with those of EVOH, with the
added advantage of recyclability. The LDPE/TLCP materi-
als are intended as EVOH and aluminum-foil replacement,
particularly in aseptic packaging.
SUMMARY
TLCPs are a class of high-performance engineering poly-
mers represented mainly by aromatic copolyesters with
exceptional processibility and high thermal, electrical, and
barrier properties. About 90% of the present TLCP market
of 10–15 million lb consists of injection-molded electronics
connectors, but rapid development of biaxial film and tubing
technology (Superex) and coextruded films and blends with
conventional packaging resins (e.g., PET, PEN and LDPE)
should expand opportunities near-term to applications in
electronics, medical, and food packaging. With resin costs
expected to drop to the $3–$5/lb range over the next several
years because of reduced monomer costs, rapid 20–25%
annual growth is forecast for the remaining 1990s to a
40–50-million-lb market by the year 2000. It is expected
that food and medical packaging will constitute a significant
share of this market driven by improved thermal stability,
longer shelf life, and ease of recycling afforded by TLCPs .
BIBLIOGRAPHY
1. W. J. Jackson, Jr., Br. Polym. J. 154–162 (1980).
2. D. J. Williams, ‘‘Applications and Markets for Thermotropic
Liquid Crystalline Polyesters (TLCPs),’’ paper presented at
Conference on High Temperature Polymers, Executive Con-
ference Management, Clearwater Beach, FL, Feb. 20–21,
1995; in Applications of High Temperature Polymers,R.R.
Luise, ed., CRC Press, Boca Raton, FL, 1996.
3. Plast. Technol., 35 (1991).
4. R. R. Luise, ‘‘Advances in Heat-Strengthened Thermotropic
Liquid Crystalline Polymer Materials,’’ in Polymeric Materi-
als Encyclopedia, J. C. Salamone, ed., CRC Press, Boca Raton,
FL, 1996.
5. E. Chiellini and G. Gallo, ‘‘Chiral Thermotropic Liquid Crys-
tal Polymers,’’ in Recent Advances in Liquid Crystalline
Polymers, L. Chapoy, ed., Elsevier, London, U.K. 1985.
6. EP Patent Appl. 346 926 (Dec. 20, 1989), T. Ishii and M. Sato
(to Kuraray).
7. U.S. Patent 4,698,397 (Oct. 6, 1987) and U.S. Patent 4,891,418
(Jan. 2, 1990) T. Toya et al. (to Nippon Oil).
8. J. S. Chiou and D. R. Paul, J. Polym. Sci., B, Polym. Phys. 25,
1699 (1987).
9. D. H. Weinkauf and D. R. Paul, J. Polym. Sci., B. Polym. Phys.
30, 817 (1992).
10. EP Patent Appl. 466 085 (Jan. 15, 1992), T. Kashimura and
M. Matsumoto (to Kuraray).
11. R. R. Luise, ‘‘Liquid Crystalline Condensation Polymers,’’ in
Integration of Fundamental Polymer Science and Technology,
Vol. 5, P. J. Lemstra and L. A. Kleintjens, eds., Elsevier,
London, U.K. 1990.
12. M. A. Kirsch and D. J. Williams, ‘‘Understanding the Thermo-
plastic Polyester Business,’’ Chemtech, 40 (1994).
13. WO Patent Appl. 94/28069 (Dec. 8, 1994), R. R. Luise (to
DuPont).
14. T. Knoll, ‘‘Controlled Thermal Expansion Printed Wiring
Boards on TLCP Dielectrics,’’ Technical Conference Proceed-
ings, IPC, Boston MA, April 1994.
15. Plast. Technol., 104 (1994).
16. Modern Plast., 25 (1995).
17. R. W. Lusignea, ‘‘Fabrication of LCP Tubing for Medical
Applications,’’ paper presented at Specialty Polyesters ’95,
Schotland Business Research, Brussels, Belgium, June
27–28, 1995.
18. T. Heiskanen and E. Suokas, ‘‘A New Low Processing Tem-
perature LCP with High Mechanical Properties and Suit-
ability for Blending,’’ paper presented at Specialty Polyesters
’95, Schotland Business Research, Brussels, Belgium, June
27–28, 1995.
LOGISTICAL/DISTRIBUTION PACKAGING
DIANA TWEDE
School of Packaging, Michigan
State University, East Lansing,
Michigan
The two key drivers of logistics management are the goals
of reducing the total cost of logistics activities and improv-
ing customer service. It aims to integrate the supply chain
to eliminate waste and optimize service.
Logistics Management is defined as ‘‘that part of supply
chain management that plans, implements, and controls
LOGISTICAL/DISTRIBUTION PACKAGING 677
