Yao N. Focused Ion Beam Systems: Basics and Applications
Подождите немного. Документ загружается.

Finally, gallium can be easily distinguished from other elements, so if
implantation occurs, the gallium ions will not interfere with the analysis of
the sample. Although other ions can be used, gallium has become the ion of
choice for the focused ion beam system.
Ultimately, the purpose of precision engineering and attention to detail in
FIB design is to produce a focused ion beam that impacts the surface at a
desired point. It is important to consider the implications of this impact. A
beam of light incident upon a surface causes local temperature increase, small
electromagnetic fluctuations, and photoelectron emission. A beam of charged
particles does all of this and more. The incident particles raise the tem-
perature of the area they impact, although generally not to a significant
degree; they change the local charge densities of the region, resulting in a
temporary charge imbalance; the addition of their kinetic energy to the
energy of the sample causes secondary electrons to be emitted, which can be
captured and used to image the surface of the sample; they produce a degree
of characteristic X-ray emission, which can be used for spectroscopy pur-
poses; and, of course, they cause damage to the surface structure at different
levels depending on the physical nature of charged particles.
The emission of secondary electrons from the surface is what gives both
beam types their imaging abilities. Adding the energy of the particles in the
beam to that of the electrons in the sample allows some of those electrons to
escape from within the material, depending on the penetration depth of the
beam and the conductivity and work function of the sample. These electrons
come from a roughly spherical region around and beneath the beam spot,
with increased numbers of electrons exiting from the sides of topographical
features that would not have escaped from a flat surface otherwise. In
addition to the secondary electrons, there is a degree of radiation produced
by the rapid deceleration of the charged particles, generally in the X-ray
spectrum. This radiation can later be used for X-ray spectroscopy. Between
this emitted radiation and the radiation from the accelerating potentials in
the sources, the chamber requires good shielding in order to preserve the
safety of the operator. All the modern electron and ion instruments have
handled this issue satisfactorily.
The ion beam is capable of efficiently and precisely depositing material.
By creating a cloud of, for example as shown in Figure 1.3, platinum atoms
above the sample, platinum can be deposited by letting the ion beam strike
these atoms, imparting some kinetic energy to them and causing them to
impact the surface, on which they remain, adsorbed onto the sample by
Van der Waals forces. This technique can be used to deposit both con-
ductive and resistive materials, since the type of atom in the suspended
Focused ion beam systems8
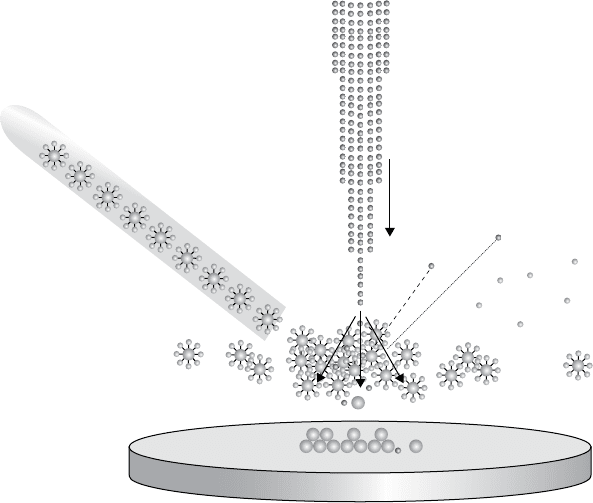
material does not matter, making the ion beam quite useful for nano-
fabrication purposes. This technique can also be used to improve electron-
beam imaging, by depositing a thin layer of conductor over the surface of a
feature to be imaged. Similar to sputter- or deposition-coating of a sample,
this method preserves more detail at a small scale by using a precisely
controlled, very thin layer of conductor to reduce charging effects and
define the surface [8].
The electron beam can be used, to some degree, to perform deposition.
However, due to the low mass of the electrons, the deposition occurs slowly,
and it is more feasible if the beam is very intense and focused precisely, to
increase the probability of electrons intersecting atoms of the deposition
material. In general, electron-beam deposition is not done with the beams
found in smaller scanning electron microscopes, instead requiring a larger
and more powerful apparatus. Until the advent of the focused ion beam, this
type of precision deposition was extremely difficult to achieve.
While the electron beam barely affects the surface, the heavy particles of
the ion beam penetrate deeper within the lattice, kicking out atoms as they go
and embedding them in the sample. Therefore, the lithographic abilities of
Cloud of platinum atoms
with organic atoms attached
Raw platinum beam
Ion beam
Reflected ion
Organic atoms
Sample
Purified platinum
deposition
Figure 1.3 A Schematic diagram of an ion beam induced platinum
deposition process.
Introduction to the focu sed ion beam system 9
the ion beam are extremely useful. It is capable at relatively low beam cur-
rents of removing atoms from a surface in a very precise and controlled
manner; it is able to make very small cuts or take large cross sections, all
without changing the chemical or structural composition of the sample.
Unlike traditional etching methods, it does not require masking or resist
stages. The ion beam can be used to etch and mill almost any material, with
little or no sample preparation. Operated at higher currents, it can achieve
very high resolution etching at rapid speeds, with high reproducibility. In
addition, the beam can be used to implant ions within the surface of the
sample in order to tune the electronic properties of the material.
Unlike from an electron beam, collisions that result from the use of a
gallium ion beam induce many secondary processes such as recoil and
sputtering of constituent atoms, defect formation, electron excitation and
emission, and photon emission. Thermal and radiation-induced diffusion
that result from these collisions contribute to various phenomena of inter-
diffusion of constituent elements, phase transformation, amorphization,
crystallization, track formation, permanent damage, and so on. Also, pro-
cesses such as ion implantation and sputtering will change the surface mor-
phology of the sample, possibly creating craters, facets, grooves, ridges,
pyramids, blistering, exfoliation, or a spongy surface.
Because of the interrelatedness of these processes, no single phenomenon
can be understood without the discussion of several others. Therefore, it is
imperative that one possesses a quantitative understanding of the experi-
mental observations as well as creativity in design so that new and more
sophisticated combinations of these versatile processes can be applied in the
field of nanotechnology. With it, we can aim at more advanced material
modification, deposition techniques, implantation, erosion, nano-fabrication,
surface analysis, and many other applications.
1.3 The ion source and electron source
In order to properly understand the focused ion beam, it is necessary to
consider the source of the beam itself, as illustrated in Figure 1.4. In almost
all focused ion beam systems, a reservoir of heavy-metal atoms (typically
gallium for the aforementioned reasons) is heated to near evaporation, after
which it flows and wets a sharp, heat-resistant tungsten needle with a tip
radius of 2–5 mm. Once heated, the Ga can remain liquid for weeks without
further heating due to its super-cooling properties. The Ga atoms then flow
to the very end of the needle, drawn there by an annular electrode concentric
with the tip of the needle and positioned close to it, called the extractor.
Focused ion beam systems10
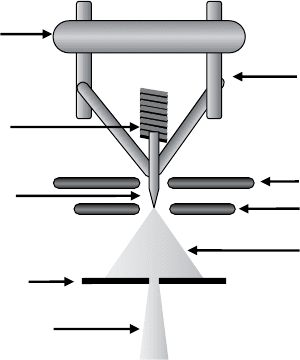
A potential difference between needle tip and aperture creates an electric field
on the order of 10
10
V/m and causes energetic ions in the region immediately
above the tip to accelerate toward the extractor. These ions exist in a region
where the balance between electrostatic and surface tension forces has drawn
the liquid-metal into a sharp ‘‘Taylor cone’’ whose apex is only about 5 nm in
diameter. The cone tip is small enough such that the extraction voltage of the
aperture can pull Ga from the needle tip and efficiently ionize it by field
evaporation of the Ga at the end of the Taylor cone, after which it is accelerated
by a potential down the ion column. The current density of ions that may be
extracted from such an LMIS system is on the order of 10
8
A/cm
2
,as
the evaporated ions are continuously replaced by a flow of liquid Ga to the
cone. The source is generally operated at low emission currents of 1–3 mAto
reduce the energy spread of the beam and to yield a stable beam. At higher
emission currents, the probability of the formation of dimers, trimers, and
droplets increases, which both increases energy spread and decreases source
lifetime.
The tip of the tungsten needle is situated just above the extractor, which is
held at a voltage on the order of 6 kV relative to the source. The resulting
intense electric field at the source tip draws the liquid Ga into a Taylor cone
and creates a tiny cusp at its end (called the ‘‘incipient jet’’). After field
evaporation causes ion emission to occur, the ions begin to accelerate down
the column. The current emitted from the tip is known as the extraction
current. It is regulated by both the suppressor and the extractor, as shown
in Figure 1.4, which roughly correspond to ‘‘fine’’ and ‘‘coarse’’ controls of
Suppress
Extract
Ion
Tungsten tip
Primary ion
Beam aperture
Electrical
feedthroughs
Insulating
mount
Coil heater
and Ga
reservoir
Figure 1.4 A cross-sectional diagram of a liquid-me tal ion source (LMIS)
found in FIB systems.
Introduction to the focu sed ion beam system 11
the emitted ion distribution. The suppressor uses an applied electric field of
up to þ2 kV to work alongside the extractor in maintaining a constant
beam current [3]. This is particularly important to control the etching rate
during milling operations. Adjusting the suppressor voltage will change the
extraction current, which means that the extraction current may be regu-
lated without changing the voltage of the extractor. This is generally the
preferred method of adjustment, as changing the extractor voltage can result
in spatial displacement of the Taylor cone and apparent beam drift on the
sample surface. This instability corresponds to the fact that LMIS have a
highly nonlinear current–voltage relationship [9]. Adjusting the suppressor is
also very useful to offset the gradual downward drift in extraction current
that occurs in LMIS as the surrounding electric field causes electrons to
collide with contaminants in the vacuum and ‘‘fix’’ them to the source.
Thus, the ability of the suppressor to maintain constant extraction voltage
without altering the source tip is an essential requirement for FIB system
stability.
Field evaporation, the process responsible for ion production in the LMIS,
is a physically complex phenomenon, and complete treatment is beyond the
scope of this chapter. Fundamentally, however, field evaporation takes place
when the potential barrier preventing evaporation has been lowered by the
presence of a field and can only be crossed by the ionization of the evapor-
ating atom on the surface of the field emitter. It can be described analytically
by first calculating the field needed to produce ions in free space, that is, in
the absence of any other field. The energy Q
0
needed to produce an ion in free
space in this case is given by Q
0
¼ H
a
þ I
n
– n, where H
a
denotes the heat of
atomic desorption, I
n
refers to the ionization energy to produce an n-fold ion,
and is the work function of the field emitter. The term n corresponds to
the released energy when n electrons return to the metal. Taking into account
the presence of a field E, which lowers the potential barrier for field ionization
by
ffiffiffiffiffiffiffiffiffiffiffiffi
2
3
e
3
E
p
, the required energy for evaporation of the liquid Ga is
Q
E
¼ Q
0
ffiffiffiffiffiffiffiffiffiffiffiffi
n
3
e
3
E
p
: ð1:1Þ
Alternatively, the atoms in the Taylor cone will ionize when
I ¼ eEx
c
; ð1:2Þ
where I is the ionization potential near a metal with work function in the
presence of an electric field E. It has been found that the critical distance x
c
from the tungsten needle tip required for ion production is approximately
0.2 nm at a field strength of 10
10
V/m.
Focused ion beam systems12

For comparison, we can consider one type of source of electrons in an
electron beam column. In the most common configuration, a tungsten filament
is heated by a large current, causing it to emit a spectrum of radiation
accompanied by a number of loose electrons that have gained sufficient energy
to overcome the work function of the metal and escape. These electrons are
then accelerated away from the tip by a set of electrostatic fields generated by
large coils, and reduced to a relatively clean beam by an aperture below the
source. The filament itself is formed into a sharp point, since this shape causes
charge to cluster at the tip, giving a greater output current from the source.
The basics of the electron gun revolve around raising an electron’s energy
above the Fermi level of the cathode material. In a standard thermal emission
gun, the energy of the electrons in the emission region is raised by the
addition of heat in the cathode and a potential from the anode, resulting in a
current density j
c
that follows Richardson’s Law:
j
c
¼ ATc
2
e
w
kT
c
; ð1:3Þ
where A is a constant related to the cathode material, T
c
is the temperature of
the emissive tip,
w
is the work function (related to the Fermi level) of the
cathode, and k is Boltzmann’s constant. This current density produces a
roughly Gaussian beam profile:
jrðÞ¼j
0
e
ð
r
r
0
Þ
2
; ð1:4Þ
where r
0
is the radius of the emissive region on the cathode tip, usually
ranging from 10 to 50 micrometers.
The tungsten filament is also usually zirconated (ZrO) to increase the
thermionic emission of electrons by means of the Schottky effect, in which an
accelerating field for electrons exists at the surface of the filament due to an
external applied electrostatic field. Zirconated tungsten exhibits a decreased
work function (2.5 eV versus 4.5 eV) and provides adequate electron emission
at only about 1600 K, compared with 2500–3000 K.
Two other classes of enhanced electron guns are worth mentioning. The
potential of rare-earth hexaboride materials, especially lanthanum hexaboride
(LaB
6
), for thermionic electron emission was first reported in 1951 by J. M.
Lafferty [10]. The work function of LaB
6
is lower than that of W (2.7 eV
compared to 4.5 eV), which, combined with its low vapor pressure at high
temperature, makes it a superior thermionic electron source for the electron
microscopes (both scanning and transmission). LaB
6
crystals can provide
considerably higher current densities, and LaB
6
offers improved coherence
and a smaller energy spread [ 11]. Extensive research in the 1970s and 1980s
Introduction to the focu sed ion beam system 13
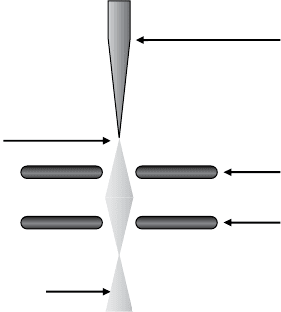
followed Lafferty’s work in an attempt to optimize the performance of this
class of materials. The drawback to this source is mainly on its relative larger
energy spread and low beam intensity for an electron probe of nanometer
size. Cold field emission guns (CFEGs) are another recent development in
SEM source technology. Unlike thermionic guns, they operate at relatively
low temperatures of about 300 K and offer superior resolution and perfor-
mance. FEGs are simpler in their operation – they use a pair of anodes below
the tungsten tip to generate intense electric fields that extract electrons by
enabling them to tunnel from the extremely sharp tip (Figure 1.5). As a result
of the small tip size and low operating temperature, the electron beam is
highly spatially coherent and experiences almost no energy spread, which
limits the deleterious effects of chromatic aberration; its current density is
also remarkably high [12]. The weakness of FEGs is that they can only be
under ultra high vacuum (< 10
10
), which requires more expensive
machinery.
Both ion and electron sources are subject to a pair of limitations. First,
the total current i n the beam leaving the extraction aperture is only a
fraction of that produced by the sources; the majority of the particl es
generated are blocked by the sides of the aperture, as their velocity vectors
are not pointed along the direction of the beam. This means that the
‘‘brightness’’ of the resulting beam depends on both aperture size and
source current. A larger aperture means a brighter beam with better ima-
ging and milling characteristics; however, the beam may not be a s uniform
or as precisely focused as one traveling through a smaller aperture. A
higher sour ce current means more charg ed p article s produced, bu t on ly a
First anode
Second anode
Field emission tip
Tungsten needle
Primary electron beam
Figure 1.5 A cross-sectional diagram of a cold field emission electron source
found in high-resolution SEM systems.
Focused ion beam systems14
fraction of those will go into the beam itself, so a subst antial increase i n
current is nee ded for a marked increase in beam brightness. The inc reased
current can cause source instability and lower the li fetim e of the source
element.
A second limitation is that of uniformity. While the electrons emitted
from the zirconated tungsten are relatively evenly distributed in energy, the
ions emitted by a liquid-metal ion source (LMIS) as described above tend
to follow a Gaussian energy distribution, sometimes asymmetrical, which
leads to chromatic aberration. The energy required to evaporate ions from
a liquid is dependent on local temperature, field strength, and surface
tensions in the area around each atom, which can vary from point to point
within the emissive region [13]. The ion emission current is strongly
dependent on the tip radius and on the tip surface condition. The sharper
the tip is, the higher the field; and the higher the field, the stronger the ion
emission. However, the inter-particle repulsion effect at high emission and
liquid flow rates with capillary characteristics need to be balanced out in
order to maintain a stable, consistent ion beam emission. Electron emission
is determined by the work function of the metal used and the field strength
at the source, with electrons escaping as soon as they have reached a well-
defined critical energy.
Both sources experience an additional degree of energy spread due to
mutual repulsion between the charges just beyond the source. The like
charges of the ions and the electrons repel each other, imparting a small but
significant random velocity that changes the overall energy profile, causing
further chromatic aberration and forming the fundamental limit for the
focusing ability of the system. Variations in emission characteristics can be
controlled to a degree, but before the limit of that control is reached, it is
overshadowed by the aberrations arising from these mutually repulsing
charged particles.
1.4 Ion optics and electron optics
After a beam of charged particles is produced, whether of ions or electrons, it
must be focused to the desired spot size by a series of lenses, usually with one
or more apertures along the beam path as well to help control aberrations. A
lens for an ion or electron beam can be thought of in the abstract as being
almost identical to a lens for light, with a similar function and similar
parameters, such as focal length and refractive index. Standard light optics
concerns thus enter the picture, including chromatic aberration, spherical
Introduction to the focu sed ion beam system 15
aberration, and astigmatism. An additional concern specific to electron and
ion beam applications is that of apparent source size, which is related to the
inter-particle repulsion mentioned above.
Electron and ion lenses function very much like light optics, but their
construction is quite different. Instead of using a material with a certain
geometry and index of refraction to bend the path of the light, electron and
ion optics use magnetic and electrostatic fields to change the paths of the
particles.
Electron beams, consisting of fast-moving, low-mass particles, are gen-
erally focused using only magnetic fields. The advantage of a magnetic field is
that it is relatively easy to produce a uniform field over a region, and that
magnetic fields do no net work on objects within them, so that the kinetic
energy of the electrons is not changed. This makes it easier to determine the
results of the electron’s impact on the surface and to process and understand
the resulting data. The magnetic lenses in an electron beam column are
generally washer-shaped coils with small central holes, so that the field is
intensified by being compressed into a smaller space. The deflection of the
beam is proportional to the distance from the axis of the lens, as in light
optics, so that the field behaves just as a glass lens would.
Charged particles, however, require stronger fields to focus higher-energy
particles because charged particles have kinetic energy vectors that must be
diverted by an applied force, while light can be focused by changing the
nature of the medium through which it propagates. The higher the kinetic
energy, the higher the force needed and the higher the lens fields must be.
Higher kinetic energies correspond to shorter wavelengths, which provide
higher resolution. In order to achieve this higher resolution, however, ever-
stronger fields must be produced in the focusing column.
Ion beams, in contrast to electron beams, use electrostatic lenses almost
exclusively. The reason for this stems from the fact that the force on a
charged particle due to an electric field E, F
e
¼qE is independent of the
particle’s velocity, whereas the force exerted by a magnetic field B, F ¼qv · B,
depends directly on the velocity. A particle accelerated by a potential drop
1V will gain a speed
ffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffi
2q1V=m
p
, where q1V is the energy and m is the
particle’s mass. Given the higher mass of ions, their velocity is 0.0028 that of
an electron accelerated by the same potential, while their momentum is 370
times higher. Magnetic optics would need to be impractically large to provide
enough focusing power for an ion beam. Electrostatic lenses, however, can be
made extremely small and are capable of producing much faster response for
Focused ion beam systems16
beam deflections, an important capability also used for beam blanking (a
toggling of the beam’s incidence on the sample by deflecting it from the
optical axis upstream from an aperture).
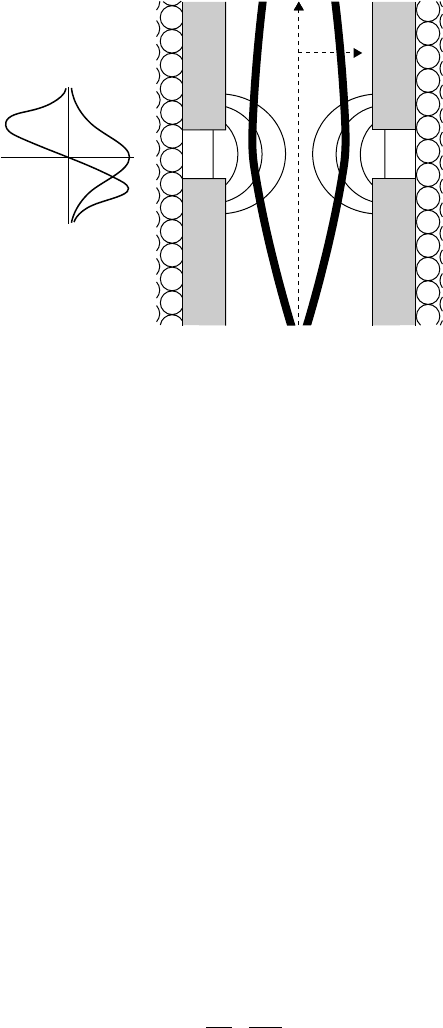
1.4.1 The lenses
Immediately after emission in an SEM, the electrons are accelerated by the
voltage V between cathode and anode to a kinetic energy of E ¼eV, where e is
the electron charge. After the beam is reduced to a relatively coaxial column
by an aperture, it enters the top of the electron optics column and passes into
the magnetic field of the first electron lens, an axial field with rotational
symmetry about the axis of the column. This field B
z
has a bell-shaped dis-
tribution along the axis where it swells out from the center of the lens coil (see
Figure 1.6). B
r
, the radial component of the magnetic field, obeys the fol-
lowing equation:
B
r
¼
r
2
@B
r
@z
: ð1:5Þ
Field
B
z
parallel to axis
B
r
perpendicular to axis
z
B
z
r
Figure 1.6 A cross section side view of the cylindrical magnetic lens within
an SEM column. Magnetic field lines in a magnetic lens are shown in thin
lines, while the thick light lines represent two possible electron paths. The
smaller inset to the left gives an approximation of the magnetic field strength
along the optical axis and perpendicular to the optical axis. The narrowing
helical rotation of electrons within the lens cannot be faithfully represented
in two dimensions but can be visualized.
Introduction to the focu sed ion beam system 17
