Masters G.M. Renewable and Efficient Electric Power Systems
Подождите немного. Документ загружается.


218 DISTRIBUTED GENERATION
So, from (4.33),
η
max
=
G
H
=
−237.2kJ/mol
−285.8kJ/mol
= 0.830 = 83.0%
This is the same answer that we found in Example 4.9 using entropy.
4.6.6 Electrical Output of an Ideal Cell
The Gibbs free energy G is the maximum possible amount of work or electricity
that a fuel cell can deliver. Since work and electricity can be converted back and
forth without loss, they are referred to as reversible forms of energy. For an ideal
hydrogen fuel cell, the maximum possible electrical output is therefore equal to
the magnitude of G. For a fuel cell producing liquid water, this makes the
maximum electrical output at STP equal to
W
e
=|G|=237.2kJpermolofH
2
(4.34)
To use (4.34) we just have to adjust the units so that the electrical output W
e
will
have the conventional electrical units of volts, amps, and watts. To do so, let us
introduce the following nomenclature along with appropriate physical constants:
q = charge on an electron = 1.602 × 10
−19
coulombs
N = Avogadro’s number = 6.022 ×10
23
molecules/mol
v = volume of 1 mole of ideal gas at STP = 22.4 liter/mol
n = rate of flow of hydrogen into the cell (mol/s)
I = current (A), where 1 A = 1 coulomb/s
V
R
= ideal (reversible) voltage across the two electrodes (volts)
P = electrical power delivered (W)
For each mole of H
2
into an ideal fuel cell, two electrons will pass through
the electrical load (see Fig. 4.26). We can therefore write that the current flowing
through the load will be
I(A) = n
mol
s
· 6.022 × 10
23
molecules H
2
mol
·
2 electrons
molecule H
2
· 1.602
× 10
−19
coulombs
electron
I(A) = 192, 945n (4.35)

FUEL CELLS 219
Using (4.34), the ideal power (watts) delivered to the load will be the 237.2 kJ/mol
of H
2
times the rate of hydrogen use:
P(W) = 237.2(kJ/mol) × n(mol/s) × 1000(J/kJ) ·
1W
J/s
= 237,200n(4.36)
And the reversible voltage produced across the terminals of this ideal fuel cell
will be
V
R
=
P(W)
I(A)
=
237,200n
192,945n
= 1.229 V(4.37)
Notice the voltage does not depend on the input rate of hydrogen. It should also
be noted that the ideal voltage drops with increasing temperature, so that at the
more realistic operating temperature of a PEM cell of about 80
◦
C, V
R
is closer
to 1.18 V.
We can now easily find the hydrogen that needs to be supplied to this ideal
fuel cell per kWh of electricity generated.
Hydrogen rate =
n(mol/s) × 2(g/mol) × 3600 s/h
237,200n(W) × 10
−3
(kW/W)
= 30.35 g H
2
/kWh (4.38)
4.6.7 Electrical Characteristics of Real Fuel Cells
Just as real heat engines don’t perform nearly as well as a perfect Carnot
engine, real fuel cells don’t deliver the full Gibbs free energy either. Acti-
vation losses result from the energy required by the catalysts to initiate the
reactions. The relatively slow speed of reactions at the cathode, where oxy-
gen combines with protons and electrons to form water, tends to limit fuel cell
power. Ohmic losses result from current passing through the internal resistance
posed by the electrolyte membrane, electrodes, and various interconnections in
the cell. Another loss, referred to as fuel crossover, results from fuel passing
through the electrolyte without releasing its electrons to the external circuit.
And finally, mass transport losses result when hydrogen and oxygen gases
have difficulty reaching the electrodes. This is especially true at the cathode
if water is allowed to build up, clogging the catalyst. For these and other rea-
sons, real fuel cells, in general, generate only about 60–70% of the theoretical
maximum.
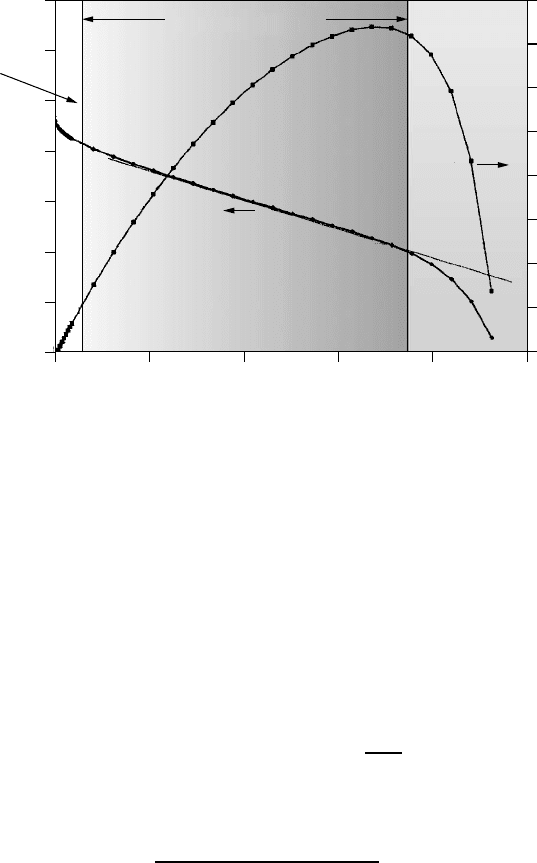
Figure 4.29 shows the relationship between current and voltage for a typical
fuel cell (photovoltaic I –V curves bear a striking resemblance to those for a
fuel cell). Notice that the voltage at zero current, called the open-circuit volt-
age, is a little less than 1 V, which is about 25% lower than the theoretical
value of 1.229 V. Also shown is the product of voltage and current, which is
power. Since power at zero current, or at zero voltage, is zero, there must be a
point somewhere in between at which power is a maximum. As shown in the
figure, that maximum corresponds to operation of the fuel cell at between 0.4
220 DISTRIBUTED GENERATION
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0
0 0.5 1.0 1.5 2.0 2.5
Cell Voltage (V)
Power (W/cm
2
)
V
P
Ohmic Loss Region
Activation
Loss
Region
Mass Transport
Loss Region
Current Density (A/cm
2
)
1.4
1.2
1.0
0.8
0.6
0.4
0.2
0
Figure 4.29 The voltage–current curve for a typical fuel cell. Also shown is the power
delivered, which is the product of voltage and current.
and 0.5 V per cell. The three regions shown on the graph point out the ranges of
currents in which activation, ohmic, and mass-transport losses are individually
most important.
Over most of the length of the fuel cell I –V graph, voltage drops linearly
as current increases. This suggests a simple equivalent circuit consisting of a
voltage source in series with some internal resistance. Fitting the I –V curve
in the ohmic region for the fuel cell shown in Fig. 4.29 yields the following
approximate relationship:
V = 0.85 − 0.25J = 0.85 −
0.25
A
I(4.39)
where A is cell area (cm
2
), I is current (amps), and J is current density (A/cm
2
).
Example 4.11 Rough Parameters of a Home-Scale Fuel Cell Stack. A1-kW
fuel cell operating on a continuous basis would provide all of the electrical needs
of a typical U.S. house. If such a fuel cell stack generates 48 V dc with cells
operating at 0.6 V each, how many cells of the type described by (4.39) would
be needed and what should be the membrane area of each cell?

FUEL CELLS 221
Solution. With 0.6-V cells all wired in series, 48/0.6 = 80 cells would be needed
to generate 48 V dc. The current that needs to flow through each cell is
I =
P
V
=
1000 W/80 cells
0.6 V/cell
= 20.83 A
Using (4.39) to find the area of each cell yields
0.6 = 0.85 −
0.25
A
× 20.83 A = 20.83 cm
2
4.6.8 Types of Fuel Cells
To this point in this chapter, the fuel cell reactions and explanations have been
based on the assumption that hydrogen H
2
is the fuel, Eq. (4.18) and (4.19)
describe the reactions, and the electrolyte passes protons from anode to cathode
through a membrane. While it is true that these are the most likely candidates for
vehicles and small, stationary power systems, there are competing technologies
that use other electrolytes and which have other distinctive characteristics that
may make them more suitable in some applications.
Proton Exchange Membrane Fuel Cells (PEMFC) Originally known as
Solid Polymer Electrolyte (SPE) fuel cells, and sometimes now called polymer
electrolyte membrane fuel cells, these are the furthest along in their development,
in part because of the early stimulus provided by the Gemini space program, and
especially now since they are the leading candidates for use in hybrid elec-
tric vehicles (HEVs). Their efficiencies are the highest available at around 45%
(HHV). Currently operating units range in size from 30 W to 250 kW.
PEM cells generate over 0.5 W/cm
2
of membrane at around 0.65 V per cell
and a current density of 1 A/cm
2
. To control water evaporation from the mem-
branes, these cells require active cooling to keep temperatures in the desired
operating range of 50
◦
Cto80
◦
C. With such low temperatures, waste heat cogen-
eration is restricted to simple water or space heating applications, which is fine
for residential power systems. One limitation of PEM cells is their need for very
pure hydrogen as their fuel source. Hydrogen reformed from hydrocarbon fuels
such as methanol (CH
3
OH) or methane CH
4
often contains carbon monoxide
(CO), which can lead to CO poisoning of the catalyst. When CO adsorbs onto
the surface of the anode catalyst, it diminishes the availability of sites where
the hydrogen reactions need to take place. Minimizing CO poisoning, manag-
ing water and heat in the cell stack, and developing lower-cost materials and
manufacturing techniques are current challenges for PEM cells.

222 DISTRIBUTED GENERATION
Direct Methanol Fuel Cells (DMFC) These cells use the same polymer
electrolytes as PEM cells do, but they offer the significant advantage of being
able to utilize a liquid fuel, methanol (CH
3
OH), instead of gaseous hydrogen.
Liquid fuels are much more convenient for portable applications such as motor
vehicles as well as small, portable power sources for everything from cell phones
and lap-top computers to replacements for diesel-engine generators.
The chemical reactions taking place at the anode and cathode are as follows:
CH
3
OH + H
2
O → CO
2
+ 6H
+
+ 6e
−
(Anode) (4.40)
1
2
O
2
+ 2H
+
+ 2e
−
→ H
2
O (Cathode) (4.41)
for an overall reaction of
CH
3
OH +
3
2
O
2
→ CO
2
+ 2H
2
O (Overall)(4.42)
Significant technical challenges remain, including control of excessive fuel
crossover through the membrane concern for methanol toxicity, and reducing
catalyst poisoning by CO and other methanol reaction products. The advantages
of portability and simplified fuel handling, however, almost guarantee that these
will be commercially available in the very near future.
Phosphoric Acid Fuel Cells (PAFC) These fuel cells were introduced into
the marketplace in the 1990s, and there are hundreds of 200-kW units built by
the ONSI division of IFC currently in operation. Their operating temperature
is higher than that of PEMFCs (close to 200
◦
C), which makes the waste heat
more usable for absorption air conditioning as well as water and space heating
in buildings.
The electrochemical reactions taking place in a PAFC are the same ones that
occur in a PEM cell, but the electrolyte in this case is phosphoric acid rather than
a proton exchange membrane. These cells tolerate CO better than PEM cells, but
they are quite sensitive to H
2
S. Although there are already a number of PAFCs in
use, their future will be closely tied to whether higher production levels will be
able to reduce manufacturing costs to the point where they will be competitive
with other cogeneration technologies.
Alkaline Fuel Cells (AFC) These highly efficient and reliable fuel cells were
developed for the Apollo and Space Shuttle programs. Their electrolyte is potas-
sium hydroxide (KOH), and the charge carrier is OH
−
rather than H
+
ions. The
electrochemical reactions are as follows:
H
2
+ 2OH
−
→ 2H
2
O + 2e
−
(Anode) (4.43)
1
2
O
2
+ H
2
O + 2e
−
→ 2OH
−
(Cathode) (4.44)

FUEL CELLS 223
The major problem with alkaline fuel cells is their intolerance for exposure to
CO
2
, even at the low levels found in the atmosphere. Since air is the source of
O
2
for the cathodic reactions, it is unlikely that these will be used in terrestrial
applications.
Molten-Carbonate Fuel Cells (MCFC) These fuel cells operate at very high
temperatures, on the order of 650
◦
C, which means that the waste heat is of high
enough quality that it can be used to generate additional power in accompanying
steam or gas turbines. At this high temperature, there is the potential for the
fuel cell waste heat to be used to directly convert, or reform, a hydrocarbon
fuel, such as methane, into hydrogen by the fuel cell itself. Moreover, the usual
accompanying CO in fuel reforming does not poison the catalyst and, in fact,
becomes part of the fuel. Efficiencies of 50–55% are projected for internal-
reforming MCFCs. With combined-cycle operation, electrical efficiencies of 65%
are projected, and cogeneration efficiencies of over 80% are possible.
In an MCFC the conducting ion is carbonate CO
3
2−
rather than H
+
,andthe
electrolyte is molten lithium, potassium, or sodium carbonate. At the cathode,
CO
2
and O
2
combine to form carbonate ions, which are conducted through the
electrolyte to the anode where they combine with hydrogen to form water and
carbon dioxide as shown in the following electrochemical reactions:
H
2
+ CO
3
2−
→ H
2
O + CO
2
+ 2e
−
(Anode) (4.45)
1
2
O
2
+ CO
2
+ 2e
−
→ CO
3
2−
(Cathode) (4.46)
Notice the overall reaction is the same as that described earlier for a “generic”
fuel cell
H
2
+
1
2
O
2
→ H
2
O (Overall)(4.47)
MCFCs operate in a very corrosive environment, and the challenges associated
with devising appropriate materials that will operate with suitably long lifetimes
are significant.
Solid Oxide Fuel Cells (SOFC) SOFCs and MCFCs are competing for the
future large power station market. Both operate at such high temperatures (MCFC,
650
◦
C; SOFC, 750–1000
◦
C) that their waste heat can be used for combined-
cycle steam or combined cycle gas turbines, and both can take advantage of
those temperatures to do internal fuel reforming. The SOFC is physically smaller
than an MCFC for the same power, and it may ultimately have greater longevity.
The electrolyte in an SOFC is a solid ceramic material made of zirconia and
yttria, which is very unlike the liquids and solid polymers used in every other
type of fuel cell. The charge carrier that is transported across the electrolyte is
the oxide O
2−
ion, which is formed at the cathode when oxygen combines with
224 DISTRIBUTED GENERATION
electrons from the anode. At the anode, the oxide ion combines with hydrogen
to form water and electrons, as shown below:
H
2
+ O
2−
→ H
2
O + 2e
−
(Anode) (4.48)
1
2
O
2
+ 2e
−
→ O
2−
(Cathode) (4.49)
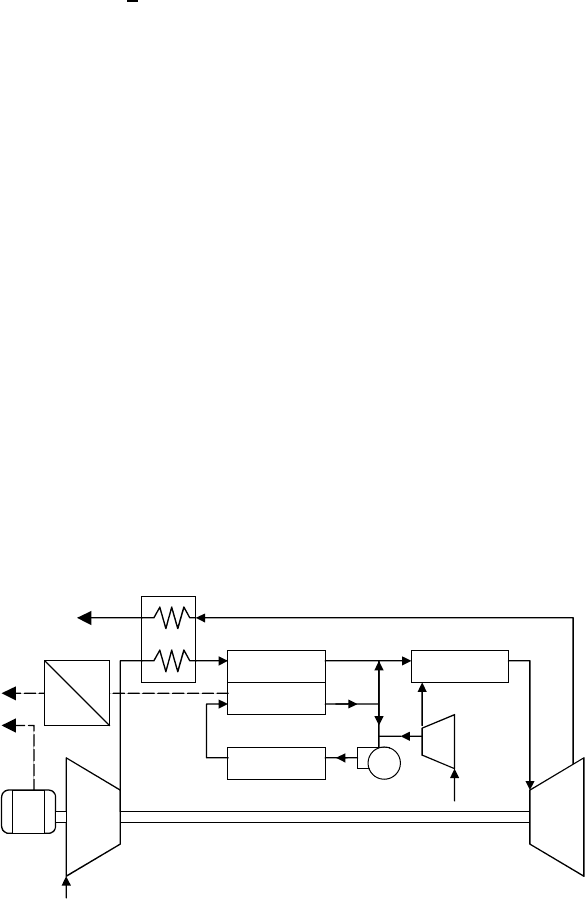
Efficiencies for SOFCs of 60% for electric power and greater than 80%
for cogeneration are projected. When combined with a gas turbine, such as
is suggested in Fig. 4.30, electrical efficiencies approaching 70% (LHV) may
be achievable.
A summary of the main characteristics of these various categories of fuel cells
is presented in Table 4.7.
4.6.9 Hydrogen Production
With the exception of DMFCs, fuel cells require a source of hydrogen H
2
for
the anodic reactions. For those that operate at higher temperatures (MCFCs and
SOFCs), methane may be reformed to yield hydrogen as part of the fuel cell
system itself; but in general, obtaining a supply of hydrogen of sufficient purity
and at a reasonable cost is a major hurdle that must be dealt with before large-
scale commercialization of fuel cells will be achieved.
Hydrogen as a fuel has many desirable attributes. When burned, it yields only
small amounts of NO
X
created when combustion temperatures are high enough to
cause the nitrogen and oxygen in air to combine, and when used in fuel cells, the
only end product is water. Given its low density, it readily escapes from confined
environments so that it is less likely to concentrate into dangerous pools the way
that gasoline fumes, for example, do. It is, however, not an energy source. It is,
Reformer
Anode
Cathode
Blower
SOFC
Compressor
Air
Combustor
Exhaust Recuperator
CH
4
Compressor
Turbine
DC
AC
Power
Inverter
Generator
Figure 4.30 Gas turbines with pressurized solid-oxide fuel cells may be capable of
nearly 70% LHV efficiency.

TA BLE 4.7 Essential Characteristics of Different Types of Fuel Cells
Type of Fuel Cell
a
Characteristic PEMFC DMFC AFC PAFC MCFC SOFC
Electrolyte Proton
exchange
membrane
Proton
exchange
membrane
Potassium
hydroxide
(8–12N)
Phosphoric acid
(85–100%)
Molten
carbonates
(Li, K, Na)
Solid oxide
(ZrO
2
–Y
2
O
3
)
Operating
temperature
50–90
◦
C 50–90
◦
C(≤
130
◦
C)
50–250
◦
C 180–200
◦
C 650
◦
C 750–1050
◦
C
Charge carrier H
+
H
+
OH
−
H
+
CO
2−
3
O
2−
Electrocatalyst Pt Pt Pt, Ni/NiO
x
Pt Ni/LiNiO
x
Ni/Perovskites
Fuel H
2
(pure or
reformed)
CH
3
OH H
2
H
2
(reformed) H
2
and CO
reformed and
CH
4
H
2
and CO
reformed and
CH
4
Poison CO > 10 ppm Adsorbed CO, CO
2
CO > 1% H
2
S > 0.5 ppm H
2
S > 1 ppm
intermediates H
2
S > 50 ppm
Applications Portable,
transportation
Portable,
transportation
Space Power
generation,
cogeneration,
transportation
Power
generation,
cogeneration
Power
generation,
cogeneration
a
PEMFC, proton exchange membrane fuel cell; DMFC, direct methanol fuel cell; AFC, alkaline fuel cell; PAFC, phosphoric acid fuel cell; MCFC,
molten-carbonate fuel cell; SOFC, solid-oxide fuel cell.
Source: Srinivasan et al. (1999).
225

226 DISTRIBUTED GENERATION
like electricity, a high-quality energy carrier that is not naturally available in the
environment. It must be manufactured, which means an energy investment must
be made to create the desired hydrogen fuel.
The main technologies currently in use for hydrogen production are steam
reforming of methane (SMR), partial oxidation (POX), and electrolysis of water.
More exotic methods of production in the future may include photocatalytic,
photoelectrochemical, or biological production of hydrogen using sunlight as the
energy source.
Methane Steam Reforming (MSR) About 5% of U.S. natural gas is already
converted to hydrogen for use in ammonia production, oil refining, and a variety
of other chemical processes. Almost all of that is done with steam methane
reformers. After some gas cleanup, especially to remove sulfur, a mixture of
natural gas and steam is passed through a catalyst at very high temperature
(700–850
◦
C), producing a synthesis gas, or syngas, consisting of CO and H
2
:
CH
4
+ H
2
O → CO + 3H
2
(4.50)
The above reaction is endothermic; that is, heat must be added, which may be
provided in part by burning some of the methane as fuel.
The hydrogen concentration in the syngas is then increased using a water-gas
shift reaction:
CO + H
2
O → CO
2
+ H
2
(4.51)
This reaction is exothermic, which means some of the heat released can be used
to drive (4.50). The resulting syngas in (4.51) is 70–80% H
2
, with most of the
remainder being CO
2
plus small quantities of CO, H
2
O, and CH
4
. Final pro-
cessing includes removal of CO
2
and conversion of remaining CO into methane
in a reverse of reaction (4.50). The overall energy efficiency of SMR hydrogen
production is typically 75–80%, but higher levels are achievable.
Partial Oxidation (POX) These systems are based on methane (or other hydro-
carbon fuels) being partially oxidized in the following exothermic reaction:
CH
4
+
1
2
O
2
→ CO + 2H
2
(4.52)
Since (4.52) is exothermic, it produces its own heat, which makes it potentially
simpler than the MSR process since it can eliminate the MSR heat exchanger
required to transfer heat from (4.51) to (4.50). After the partial oxidation step,
a conventional shift reaction can be used to concentrate the H
2
in the result-
ing syngas.
Gasification of Biomass, Coal, or Wastes As mentioned in Section 4.4,
gasification of biomass or other solid fuels such as coal or municipal wastes by
high-temperature pyrolysis can be used to produce hydrogen. In fact, that was
FUEL CELLS 227
the primary method of hydrogen production before natural gas become so widely
available. With the likelihood of relatively inexpensive technology to remove
CO
2
from the resulting syngas, there is growing interest in coal gasification for
hydrogen production, followed by capture and sequestration of CO
2
in deep saline
aquifers or depleted gas fields. Some researchers hope such carbon sequestration
may provide a w ay to continue to exploit the earth’s large coal resources with
minimal carbon emissions.
Electrolysis of Water In reactions that are the reverse of conventional fuel
cells, electrical current forced through an electrolyte can be used to break apart
water molecules, releasing hydrogen and oxygen gases:
2H
2
O → 2H
2
+ O
2
(4.53)
In fact, the same membranes that are used in PEM cells can be used in low-
temperature electrolyzers. Similarly, solid-oxide electrolytes can be used for high-
temperature electrolysis.
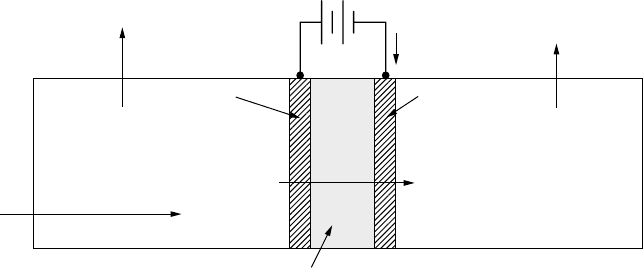
A sketch of an electrolysis cell that uses a proton exchange membrane is
shown in Fig. 4.31. De-ionized water introduced into the oxygen side of the
cell dissociates into protons, electrons, and oxygen. The oxygen is liberated, the
protons pass through the membrane, and the electrons take the external path
through the power source to reach the cathode where they reunite with protons
to form hydrogen gas. Overall efficiency can be as high as 85%.
Hydrogen produced by electrolysis has the advantage of being highly purified,
so the problems of catalytic CO poisoning that some fuel cells are subject to is
not a concern. When the electricity for electrolysis is generated using a renewable
energy system, such as wind, hydro, or photovoltaic power, hydrogen is produced
without emission of any greenhouse gases. And, as Fig. 4.32 suggests, when the
resulting hydrogen is subsequently converted back to electricity using fuel cells,
4H
+
2H
2
O −> 4H
+
+ O
2
+ 4e
−
4e
−
+ 4H
+
−> 2H
2
e
Anode Cathode
+−
Proton exchange membrane (PEM)
4e
−
O
2
2H
2
Water
H
2
O
Power source
Figure 4.31 A proton exchange membrane used to electrolyze water.
