Moser A.P., Folkman S. Buried Pipe Design
Подождите немного. Документ загружается.

3. An analysis of reported data shows that the chance of any problem’s
occurring in PVC water pipe is about twice as high for pipe manu-
factured to the ASTM D 2241 standard as for pipe manufactured to
the AWWA C900 standard.
4. Reported experiences with problems associated with exposure to
ultraviolet light or aggressive chemicals are low in number.
5. Tapping problems associated with PVC pipe are decreasing with
time as utilities gain more experience. Of those utilities reporting
catastrophic tapping failures in the 5-year period, only about 27
percent reported having, on average, more than one failure per year.
In addition, approximately 80 percent of those utilities reporting
catastrophic failures felt they had solved their tapping problems
and have not had any failures in the 2-year period just before the
survey. It was determined that the majority of pipe tappers learn
tapping procedures through an informal apprenticeship program.
6. Some utilities require the use of saddles for tapping of PVC pipe
and feel that this requirement reduces tapping problems. However,
an analysis of data indicates that utilities requiring the use of sad-
dles reported, on average, about the same number of problems as
those using direct tapping.
7. PVC pipe being installed was determined to be of high quality.
8. Some samples of PVC pipe manufactured in the 1970s were of poor
quality as determined by the acetone test for extrusion quality.
(Note: This conclusion is based on results from a small sample of
pipe from a single utility and from only one manufacturer. Thus,
this conclusion should not be generalized. Also, all other PVC pipe
samples were determined to be of high quality.)
Polyethylene (PE) Pipes
Polyethylene used to manufacture pipe is available in several types
and grades as per ASTM D 1248. Some grades of polyethylene may
crack or craze when subjected to certain levels of stress or when in con-
tact with certain chemicals. This degradation is usually accelerated
when high stresses and certain chemicals act simultaneously. This
phenomenon is known as environmental stress cracking. Certain
grades are highly resistant to stress cracking. Type III, class C, cate-
gory 5, grade P34 polyethylene is a high-density, weather resistant,
stress-crack resistant material (Table 7.16).
Polyethylene pipes are available in various sizes and wall configu-
rations for varied applications, some of which are listed in Table 7.17.
Other sizes for specific applications may be available from a particular
manufacturer. See Table 7.18 for polyethylene standards.
Plastic Flexible Pipe Products 459

460 Chapter Seven
TABLE 7.16 Polyethylene Design Properties
Hydrostatic-design basis (HDB) 1250 lb/in
2
Hydrostatic-design stress (HDS) 625 lb/in
2
Elastic modulus 100,000 lb/in
2
Tensile stress (short-time) 3200 lb/in
2
Hazen-Williams coefficient C 150
Manning’s coefficient n 0.009
TABLE 7.17 Polyethylene Pipes
Application Type Size range, in
Industrial (includes gas) Solid wall
3
4
–48
Water (new service) Solid wall
1
2
–3
Water (insertion) Solid wall
1
2
–4
Gravity sewer (lining) Solid wall 4–48
Gravity sewer Profile (ribbed) wall 18–96
TABLE 7.18 Standards for Polyethylene
ASTM D 3287 Biaxially Oriented Polyethylene (PEO) Plastic Pipe (SDR-PR) Based
on Controlled Outside Diameter
ASTM D 3261 Butt Heat Fusion Polyethylene (PE) Plastic Fittings for
Polyethylene Plastic Pipe and Tubing
ASTM D 405 Corrugated Polyethylene Tubing and Fittings
ASTM D 3197 Insert-type Polyethylene Fusion Fittings for SDR 11.0 Polyethylene
Pipe
ASTM D 2609 Plastic Insert Fittings for Polyethylene Plastic Pipe
ASTM D 2104 Polyethylene Plastic Pipe, Schedule 40
ASTM D 2239 Polyethylene Plastic Pipe (SIDR-PR) Based on Controlled Inside
Diameter
ASTM D 3350 Polyethylene Plastics Pipe and Fittings Materials
ASTM F 714 Polyethylene Plastic Pipe (SDR-PR) Based on Outside Diameter
ASTM D 3035 Polyethylene Plastic Pipe (SDR-PR) Based on Controlled Outside
Diameter
ASTM D 2447 Polyethylene Plastic Pipe, Schedules 40 and 80, Based on Outside
Diameter
ASTM D 2737 Polyethylene Plastic Tubing
ASTM F 771 Polyethylene Thermoplastic High-Pressure Irrigation Pipeline
Systems
ASTM D 2683 Socket-Type Polyethylene Fittings for Outside Diameter-Controlled
Polyethylene Pipe and Tubing
ASTM F 810 Smooth-Wall Polyethylene Pipe for Use in Drainage and Waste
Disposal Absorption Fields
ASTM D 1248 Polyethylene Plastics Molding and Extrusion Materials
AWWA C901 Polyethylene Pressure Pipe, Tubing and Fittings,
1
2
in through 3
in, for Water
Plastic Flexible Pipe Products 461
Many of the larger-diameter gravity sewer polyethylene pipes have
pipe stiffness F/y of 10 lb/in
2
and some even lower than 4 lb/in
2
.
Extreme care must be taken during installation of these low-stiffness
pipes because of the possibility of overdeflection and buckling due to
soil load.
Handling factor
Ring stiffness, the pipe’s ability to resist ring deflection, is a function
of EI/D
3
(see Chap. 3). Some literature promoting polyethylene pipe
gives EI/D
2
as the property which is a measure of the pipe’s resistance
to deflection. This idea has absolutely no theoretical or experimental
basis, and if used in the design of a pipe installation could be the direct
cause of pipe overdeflection or collapse.
The term EI/D
2
, called handling stiffness, is sometimes used to rate
the ease of handling without damage. The inverse of this factor, D
2
/(EI),
is called the flexibility factor and is used by the corrugated-steel pipe
industry to rate handling flexibility. These factors may have arisen from
a bending strain consideration as follows:
Bending strain
where C
1
a constant
P pressure
D diameter
PD vertical load
D/2 moment arm
t/2 half-wall thickness
I wall moment of inertia
E modulus of elasticity
One can easily see that D
2
/(EI) is a factor in the above equation. Thus
bending strain for a given soil pressure is directly proportional to this
factor. The inverse of this factor is a measure of the particular product’s
ability to resist bending strain. Of course, ring deflection is not a direct
function of D
2
/(EI), but is a direct function of D
3
/(EI). It matters not
what causes the deflection—handling, installation, concentrated loads,
or soil pressure—the deflection is still a function of D
3
/(EI), not D
2
/(EI).
Also buckling, whether hydrostatic or due to soil pressure, is a function
of D
3
/(EI). Thus, D
2
/(EI) or EI/D
2
should not be used in design calcula-
tions, nor should this factor be used to classify a pipe’s stiffness charac-
teristics for deflection control.
C
1
(PD) (D/2) (t/2)
EI
Mc
EI
462 Chapter Seven
Example 7.12—150 lb/in
2
polyethylene pipe Calculate the required dimen-
sion ratio (DR) for a polyethylene pressure pipe. The maximum working
pressure is 150 lb/in
2
, no surge is anticipated, and the safety factor is to
be 2.5.
1. Calculate the hydrostatic-design stress:
HDS
HDB 1250 lb/in
2
HDS 500 lb/in
2
2. Calculate DR, using Eq. (7.7):
DR 1
where
HDS
DR 1 7.67
Select the next-lower available DR:
DR 7.0
Example 7.13—A 6-in pressure sewer pipe It is proposed to use 6-in polyeth-
ylene pipe for a pressurized sewer line. The maximum pressure including
surge is 50 lb/in
2
, and the maximum depth of cover is 20 ft. (a) Select the
proper wall thickness. (b) What requirements will be necessary concerning
pipe-zone soil type and compaction? (Use safety factor 2.0, OD 6.625,
and deflection limit 5 percent.)
For part (a),
1. Hydrostatic-design stress equals HDB/safety factor.
HDS 625 lb/in
2
2. Use Eq. (7.7) to calculate the dimension ratio:
DR 1
2
P
1250
2
HDB
2.0
2 (500)
150
2
P
1250
2.5
HDB
safety factor
Plastic Flexible Pipe Products 463
1 26
26
Thickness t 0.25 in
solution For part (b),
1. Determine pipe stiffness :
where E 100,000 lb/in
2
I
r mean radius 3.19 in
So
27 lb/in
2
2. Use Spangler’s equation to find required soil modulus E′ [see Eq. (7.4)].
E
′
where PS pipe stiffness F/y, so
E
′
480 lb/in
2
Thus, a granular soil compacted to at least 85 percent Proctor density will
be required in the pipe zone (see Table 3.4).
Example 7.14—A 96-in storm sewer pipe A 96-in storm sewer pipe is to be
installed. The deepest cut will require 14 ft of cover. A profile-wall polyethyl-
ene pipe is to be considered. The wall moment of inertia I of this proposed PE
pipe equals 0.524 in
4
/in. The pipe is to be installed in such a manner that the
resulting vertical deflection is less than 5.0 percent. (1) Calculate the pipe stiff-
ness F/y. (2) If selected, how should this particular type of pipe be installed?
(3) Comment on the suitability of the proposed pipe for this application.
0.56(20) / (0.05) 27
0.41
0.56H/ (y/D) PS
0.41
(6.7)(100,000) (0.25/3.19)
3
12
F
y
OD t
2
(0.25)
3
12
t
3
12
6.7EI
r
3
F
y
F
y
6.625
26
OD
DR
OD
t
2 (625)
50

464 Chapter Seven
1. Calculate pipe stiffness.
where E 100,000 lb/in
2
I 0.524 in
4
/in
r 96/2 48 in
So
3.17 lb/in
2
Note: This is a very low value—pipe will be extremely flexible.
2. For the design installation, use Spangler’s equation to calculate the
required E′.† [See Eq. (7.4).]
E′
374 lb/in
2
It can be determined from Table 3.4 that for E′ 374 lb/in
2
, a granular
material compacted to at least 85 percent Proctor density is required. It
appears that this particular pipe can be made to work under tightly con-
trolled installation conditions. For pipes with such low stiffnesses, buck-
ling due to soil load is much more likely. This failure mode is discussed
in detail in Examples 7.16 and 7.17.
3. Because pipe stiffnesses below 10 lb/in
2
offer little inherent resistance to
deflection, the pipe ring may need to be braced internally while the soil
around the pipe is placed and compacted. After the required soil density
is obtained, the braces (stills) may be removed. For plastic pipe, bracing
may penetrate the pipe wall unless the bracing is carefully designed and
positioned. Because of the above concerns, this pipe should be selected
only if the above concerns can be addressed. Also, a granular material
compacted to at least 85 percent standard Proctor density should be spec-
ified for the pipe zone.
0.56H/0.05 3.17
0.41
0.56H/(y/D) PS
0.41
(6.7) (100,000) (0.524)
(48)
3
F
y
6.7EI
r
3
F
y
†Note: This equation is derived directly from Spangler’s Iowa formula. The Iowa for-
mula is not accurate for very low pipe stiffnesses. Test data at Utah State University
indicate that this equation is nonconservative for a pipe stiffness F/y 10 lb/in
2
and
may not be appropriate for F/y 3.17. A quick examination of the above equation will
show that it cannot hold in the limit as pipe stiffness approaches zero since it indicates
a 0 lb/in
2
pipe will perform essentially the same as, say, a 10 lb/in
2
pipe. Thus, the above
equation can be used for pipe stiffness of 10 lb/in
2
and higher. The error involved is a
function of other parameters as well as pipe stiffness. However, the error is within
acceptable limits for pipe stiffnesses of 10 lb/in
2
or greater. Pipes with 3.17 lb/in
2
pipe
stiffness have virtually no inherent strength and stiffness compared with soil. Thus, the
pipe in this example should be installed in a well-compacted granular material.
Long-term ductility of polyethylene
materials
Introduction. Plastic pipes derive their outstanding performance char-
acteristics from their ability to deform and transfer the earth load to
the surrounding soil. The design rationale is based on its ductility—its
ability to undergo localized strains and deformations without cracking
or structural failure. This is true for all thermoplastics and is espe-
cially true for polyethylene.
A principal advantage of thermoplastic piping for buried applica-
tions is that it allows for the pipe-soil interaction which stabilizes and
strengthens buried pipe; it safely reduces stress concentrations; it
facilitates handling and installation; it simplifies product and instal-
lation design; and it results in more forgiving and durable installa-
tions. Design protocols and construction recommendations for
thermoplastics buried piping have been developed on the assumption
of ductile behavior. Standards for thermoplastics piping include mate-
rial and product requirements intended to ensure that the product is
made from materials with high strain capacity.
Ductility. Ductile materials are able to tolerate marked deformation
before failure. This allows for the redistribution and possible reduction
of stresses. Thus, ductile structures can safely shed stress concentra-
tions. Designs using ductile materials can be based on average stress
and, therefore, can be greatly simplified. On the other hand, designers
using brittle materials must anticipate the maximum strains and
stresses and know the points at which they act. By necessity, brittle
materials require designs procedures that are more complex.
Engineers who design piping systems are well aware of the better
performance of ductile iron pipe over the old gray cast iron pipe.
Ductile iron pipe is a flexible pipe, and installation demands are less
stringent. The better performance characteristics are directly attribut-
able to ductility. Engineers are also aware that more ductile, milder
steels have simpler design procedures that usually result in improved
field performance when compared to higher-strength, more brittle
steels. Ductile structures are more forgiving in regard to stresses that
are often not considered by designers. Such stresses include, but are
not limited to, those induced by improper handling and installation
and those locked in during the manufacturing process.
Ductile—but not always. A designer would like to use a material whose
properties are known and do not change with time. Some polyethylene
pipes have been reported to have failed in a brittlelike manner at low
strain levels. This transformation from a ductile to a brittlelike mate-
rial is the consequence of the formation and propagation of slowly
Plastic Flexible Pipe Products 465
growing cracks. For these cracks to initiate, the PE material must have
a crystalline structure. The more crystalline the structure, the easier
it is for the cracks to initiate. The transition from the ductile to the
brittlelike state results in not only lower longer-term strain capacity,
but also lower longer-term strength and less endurance to cyclic
stressing.
For nonpressure pipes, it is possible for cracks to form and grow to
such extent as to eventually compromise the infiltration and exfiltra-
tion requirements on the pipe and may also destroy the structural
integrity of the pipe-soil system. The possibility of the development
and continued growth of cracks in a pressure pipe is usually unac-
ceptable, particularly when the pipe is carrying a dangerous material
such as natural gas.
PE materials used to manufacture pipes must offer adequately high
resistance to crack initiation and propagation. PE polymer materials
are partially crystalline and partially amorphous. Density and molec-
ular weight have tremendous influence on properties of the particular
polyethylene. The reader should understand that high molecular
weight and high density are not the same thing, nor are they always
mutually beneficial. Higher-density polyethylene materials are more
crystalline in structure, which results in higher stiffness, tensile
strength, and hardness. Increases in these properties are often consid-
ered beneficial. However, these benefits are accompanied by decreases
in toughness, impact strength at lower temperatures, and long-term
crack resistance. One may somewhat compensate for these losses by
increasing the molecular weight of the PE. The downside of increasing
the molecular weight is a simultaneous increase in the melt viscosity.
Manufacturers of pipe are concerned because high melt viscosities
mean the ease of processability is diminished, and it becomes more dif-
ficult to manufacture pipe. The challenge is to balance density and
molecular weight to offer long-term ductility, and resistance to stress
cracking, and still be able to process the material into a pipe.
To meet this challenge, resin suppliers have copolymerized ethylene
with small amounts of other monomers. Extreme care must be taken
by polymer chemists because experience has shown that such copoly-
mers can become more crystalline with time. The rate in the process in
moving away from an amorphous structure to a crystalline structure
is a function of temperature. This is more of a concern for commercially
available PE polymers (compared to simple homopolymers such as
PVC) because of the greater diversity in the molecular structure. For
PVC, it is possible to fairly precisely link basic polymer characteristics
such as density, molecular weight, and melt viscosity with resultant
mechanical properties, such as strain capacity and long-term strength.
466 Chapter Seven
Plastic Flexible Pipe Products 467
The percentage of elongation in a tensile test is commonly used as a
measure of ductility for metals. However, it is well known that even
ductile materials can fail at low strains and in a brittlelike fashion if
subjected to a multiaxis stress field and if a small material flaw (crack)
is present. With this said, it is still important to understand that the
more ductile the pipe material, the more “forgiving” will be the end
product. Thus, for materials like PE, it is necessary to have test
requirements for ensuring that only materials of adequate long-term
strain capacity are used for buried piping, whether it be intended for
pressure or nonpressure uses.
Such a test will only determine properties of the material at the time
of the test. Therefore, there must also be some assurance that the
material will not become more crystalline with time and thus make a
transition from ductile to brittle behavior. For PE materials the test-
ing required is by no means a simple matter because ductility can be
compromised by slow-acting crack initiation and crack growth.
The ESCR test
In 1959, ASTM standard test method D 1693 was issued. Soon after,
an environmental stress crack resistance (ESCR) criterion was added
to the ASTM specification for the classifying of PE compositions. Other
PE specifications followed based on the ESCR test. The ESCR test is
imprecise and is recognized as giving only a rough measure of long-
term crack resistance. The bent-strip ESCR test has never been
adopted for the defining of minimum crack resistance requirements for
the pipe material. Instead, industry adopted the hydrostatic-design
basis requirement to ensure adequate strength (which also takes into
account the adequacy of long-term crack resistance). HDB-rated resins
are required for pressure pipe.
The HDB requirement for PE
PE materials that pass the HDB requirement when manufactured
into pipe will perform well. This is true for both pressure and non-
pressure applications. However, the HDB requirement excludes mate-
rials from nonpressure uses with lower but quite adequate long-term
crack resistance.
The procedure by which the HDB method excludes materials of low
crack resistance may be described by referring to Fig. 7.42. This figure
depicts the characteristic stress-rupture behavior for PE pipe at ambi-
ent temperature which results in two zones: an initial zone of gradual
regression of rupture strength with increased time to fail (in which the
pipes fail by ductile yielding) and a zone of more rapid regression of
strength (where failures are small brittlelike slits with no evidence of
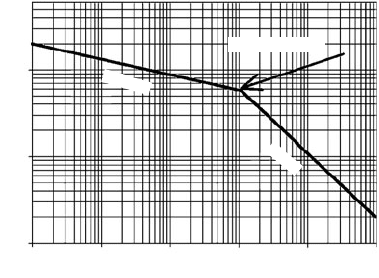
468 Chapter Seven
any yielding). The later this transition occurs, the more resistant the
material is to crack initiation and growth. In 1986, a requirement was
added to ASTM D 2837. This new requirement stated that no HDB
may be awarded to a PE unless it is demonstrated (through a specially
devised elevated-temperature test protocol) that the transition from
ductile to brittlelike zone in the stress-rupture properties occurs
beyond 100,000 h. While this requirement has resulted in very strong
assurance of permanence of ductility, it is believed by some to be too
demanding to apply in the case of PE materials used in gravity pipe.
The NCTL test
There is an obvious need for a fairly simple and quick test that gives a
reliable relative measure of a PE pipe’s capacity to safely tolerate sus-
tained straining. Dr. Grace Hsuan of Drexel University has reported on
a research program to find such a test.
24
A preliminary evaluation indi-
cates that an ASTM method, the notched constant tensile load (NCTL)
test, can fulfill this need. This evaluation also shows that this test can be
used on PE materials containing postconsumer (i.e., recycled) resins. The
ASTM D 5397 (NCTL) test was developed to evaluate the stress crack
resistance (SCR) of medium-density polyethylene geomembranes. The
test is performed using notched dumbbell specimens under constant ten-
sile stress in a controlled-temperature surface active solution.
Dr. Hsuan states the following:
Unfortunately, this test also requires a relatively long testing time to
obtain the full NCTL stress rupture characterization curve, typically on
the order of a few weeks. Thus it is not well suited for a MQC test.
However, an abbreviated version of the test has been developed based on
a single point evaluation, hence it is referred to as a SP-NCTL test.
Because stress cracking is caused by the slow crack growth mechanism,
Failure Time (Hours)
Hoop Stress
Transition Zone
Brittle
Ductile
Figure 7.42 Schematic of stress regression for PE
pipe.
