Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.

chromium metal to the other. The oxygen activity in the Cr/Cr
2
O
3
mix is known
from thermochemical data relating to the equilibrium reaction:
Cr
2
O
3
!2Cr þ 3=2O
2
(4:40Þ
A common form of disposable sensor is a long cardboard tube with the sensor
elements built into one end. The tube is plunged into the molten steel and the
measurements made within a few seconds.
Amperometric mode An advantage of the lambda sensor as described above is
its sensitivity close to the l ¼ 1 point where the oxygen activity in the exhaust
changes rapidly with burn conditions and consequently so too does the cell e.m.f.
The disadvantage is that under ‘lean-burn’ (oxygen-rich) conditions the log
dependence on oxygen activity renders the cell insufficiently sensitive to provide
effective control. This shortcoming has led to the introduction of sensors used
in the amperometric mode discussed below and illustrated schematically in
Fig. 4.40.
The requirement is to measure the concentration (of order 1 vol.%) of oxygen
in a gas. The oxygen diffuses down the capillary and by Fick’s Law the flux, J,is
J ¼DAdc=l (4:41Þ
in which D is the oxygen gas diffusion coefficient, l the capillary length and A its
cross-section; dc is the concentration difference between the capillary ends.
The oxygen ions are driven into the electrolyte by an applied e.m.f. and the
current (I), limited by the supply of oxygen ions, measured. It follows that
I ¼ 4eJ (there are four electrons carried by each oxygen molecule)
202 CERAMIC CONDUCTORS
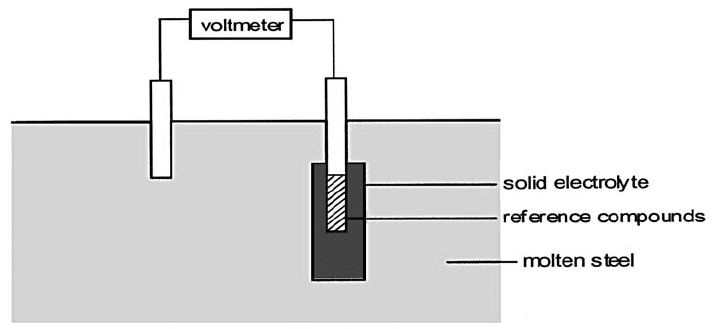
Fig. 4.39 Schematic of an oxygen meter for monitoring the oxygen content of steel during
refining (adapted from [15]).
TEAMFLY
Team-Fly
®
or
I ¼ 4eDAdc=l (4:42Þ
Assuming that the oxygen concentration at the surface of the electrolyte is zero,
and with D, A and l all constant, then
C
ðoxygenÞ
/ I (4:43Þ
where C
(oxygen)
is the oxygen concentration in the gas.
The capillary tube can be replaced by a porous ceramic appropriately limiting
gas flow. Because of the constant urge to reduce component size and cost
multilayer technology is exploited in the fabrication of sensors.
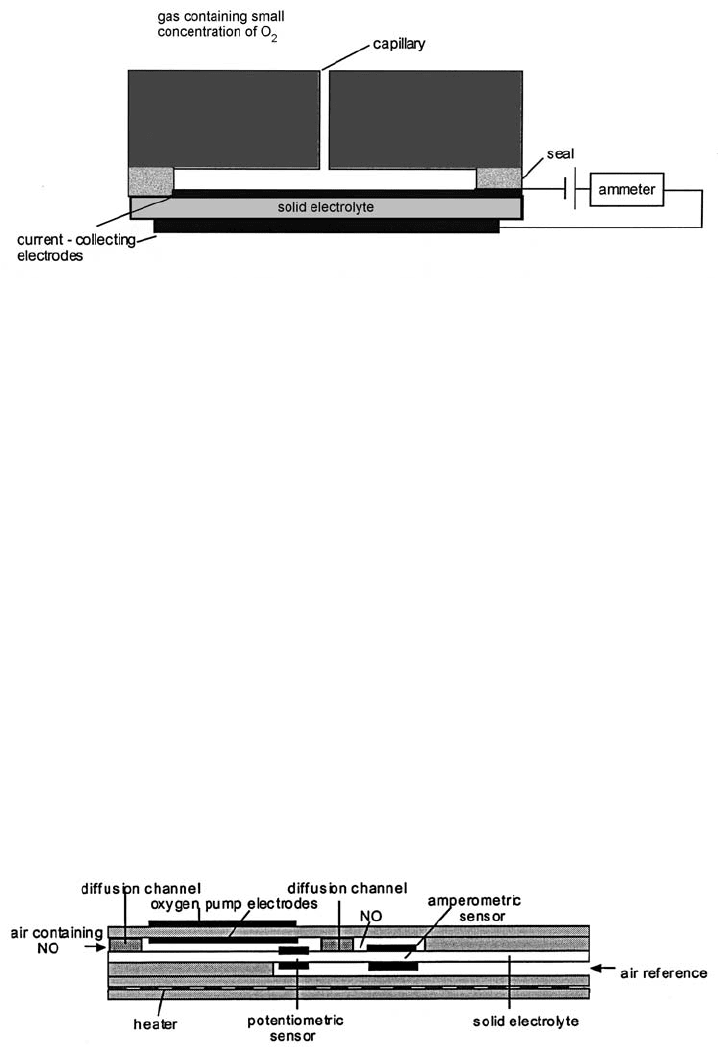
The sensor illustrated in Fig. 4.41 for measuring NO in oxygen well illustrates
the complexities in design which multilayer technology can readily cope with. In
the first stage the gas mixture enters the sensor where the oxygen is pumped
away. The potentiometric sensor monitors the oxygen level and controls the
pumping. The NO diffuses through the second barrier and dissociates at the
amperometric sensor electrode, the oxygen ions being driven through the sensor
into the air reference compartment.
CERAMICS-BASED CHEMICAL SENSORS 203
Fig. 4.40 Schematic illustrating the principle of the amperometric mode.
Fig. 4.41 A planar form of NO sensor the entire fabrication exploiting multilayer technology
(adapted from [16]).
As an alternative to the limited gas diffusion technique described above, high
oxygen activities can also be measured by driving a current though the wall of a
membrane separating the test and reference gases (e.g. Fig. 4.36). The driving
potential difference is reversed so that the current is measured under the applied
voltage reinforced by the cell e.m.f. for one half of the cycle and by the applied
voltage minus the cell e.m.f. for the next half cycle. It can be shown that the test
oxygen activity is directly related to the ratio of the currents flowing under the
two different voltage gradients. The technique is suited to measuring oxygen
concentrations over a wide range [17].
Sensors for chemical species other than oxygen
The following examples serve to illustrate the versatility of the experimental
approach provided suitable electrolytes can be found. The technology is reviewed
by D.J. Fray [15,18].
Sensing hydrogen in molten aluminium Because of the detrimental effect
dissolved hydrogen can have on the mechanical properties of many metals it is
monitored in metallurgical processing. For this the requirement is for a proton
conductor and suitable ceramics have been identified.
It has been known for many years [19] that ‘water’ can be incorporated into
silica in the form of hydroxyl groups which have a characteristic absorption in
the infrared. This was a problem encountered in the early days of fibre optics
communications’ technology and solved by excluding water from the processing
atmospheres. This ‘water’ can be readily introduced by high temperature
annealing in water vapour and removed by heating in a water-free atmosphere.
More recently, H. Iwahara et al. [20] reported that some compounds having
the perovskite structure (see Section 2.7.3) become proton conductors if
hydrogen is introduced into the crystal, and the solubility of ‘water’ and proton
mobility in ‘perovskites’ are now actively researched topics [21]. The perovskites
which can be tailored to exhibit high protonic conductivity have compositions of
the type
AB
1x
M
x
O
3x
in which M is a trivalent dopant and x the oxygen deficiency per formula unit.
CaZr
0.9
In
0.1
O
3x
is an example, the indium substituting for zirconium and the
compensating defect being oxygen ion vacancies. Other examples are
SrZr
0.95
Y
0.05
O
3x
, BaCe
0.8
Y
0.2
O
3x
, BaCe
0.9
Nd
0.1
O
3x
and SrCe
0.95
Yb
0.05
O
3x
.
When these compositionally modified perovskites, which are p-type semi-
conductors, are heated to high temperatures in a water vapour atmosphere
protons are incorporated into the structure, most probably is the form of
hydroxyl groups, with the proton able to ‘hop’ from oxygen to oxygen. The
204 CERAMIC CONDUCTORS
proton mobility will depend upon the strength of the hydrogen bond which will,
in turn, depend critically on structural details. The proton transport number can
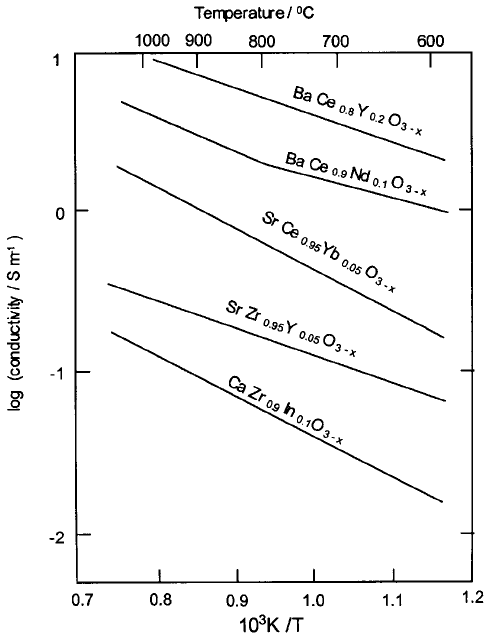
be as high as 0.99. Arrhenius plots of the conductivities of the perovskites listed
above are shown in Fig. 4.42.
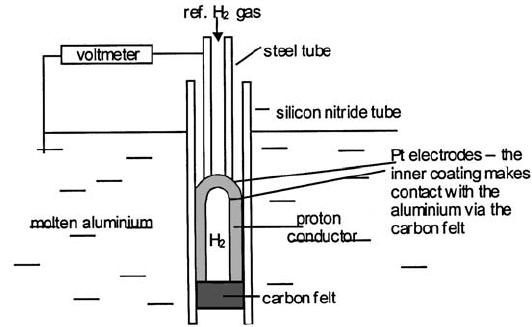
The exploitation of proton conductors is discussed by H. Iwahara [22]. Figure
4.43 is a schematic of a commercial sensor for measuring the hydrogen content of
molten aluminium [23]. Silicon nitride and carbon are used in the construction
because both are corrosion-resistant to molten aluminium. The electrolyte tube is
CaZr
0.9
In
0.1
O
3x
ceramic. The reference hydrogen activity is fixed by a mixture of
1 vol.% hydrogen in argon fed to the platinised outer sensor surface via the
spring-loaded steel tube. The hydrogen gas collected in the inverted sensor tube is
in equilibrium with the hydrogen dissolved in the molten aluminium. The
measured e.m.f. is given by the Nernst equation
CERAMICS-BASED CHEMICAL SENSORS 205
Fig. 4.42 Electrical conductivities of modified ‘perovskite’ proton conductors (adapted from
[22]).
E ¼ RT=2F flnða@
H
2
=a
0
H
2
Þg (4:44Þ
in which a@
H
2
and a
0
H
2
are respectively the hydrogen activities on the measuring
and reference sides of the cell. The factor 2 arises because two electrons are
associated with the hydrogen molecule.
Sensing sodium The b-aluminas (see Section 4.5.3) can be successfully
exploited as a sodium sensor. Sodium is widely used in the metallurgical
industry, for example for removing arsenic and antimony from zinc and lead, for
removing the same two elements and oxygen from copper, and phosphorus from
iron. It is also added to aluminium–silicon alloys to control microstructure and,
in consequence, mechanical properties.
The very high vapour pressure of sodium at high temperatures poses problems
for controlling the sodium content of a melt, necessitating its constant and
accurate monitoring.
There are no new principles involved as far as an electrolytic sensor is
concerned. The electrolyte membrane is b-alumina and the reference sodium
activity fixed, at constant oxygen activity, by a mixture of sodium ferrite
(Na
10
Fe
16
O
29
, which can be written 5Na
2
O 8Fe
2
O
3
) and Fe
2
O
3
, just as
it is common practice to fix oxygen activity by a mixture of a metal and its
oxide.
Sensing CO
2
ACO
2
sensor developed by G.M. Kale et al. [24] illustrates a
principle permitting the sensing of species other than that involving the mobile
ion in a solid electrolyte. For example sensing CO
2
employing a Na-ion
206 CERAMIC CONDUCTORS
Fig. 4.43 A protonic electrolyte used for measuring the hydrogen content of molten
aluminium (adapted from TYK Corporation (Japan) literature; see [23]).

conductor, in this case ‘Nasicon’*. The experimental arrangement is shown in
Fig. 4.44. The Nasicon tube is packed with Na
2
CO
3
which serves as an ‘activity-
linking’ compound. Because of the equilibrium reaction
Na
2
CO
3
!Na
2
O þ CO
2
(4:45Þ
any change in CO
2
activity in the gas phase in contact with the solid Na
2
CO
3
causes a change in Na
2
O activity. At the surface of the electrolyte the activity of
Na
2
O in the Nasicon changes to the same value. This change in activity can be
sensed through the corresponding change in cell e.m.f.
At the platinum contact with the solid Na
2
CO
3
the electrochemical reaction is:
Na
2
CO
3
! 2Na
.
þ 2e
0
þ CO
2
þ
1
2
O
2
(4:46Þ
And at the Nasicon/gas interface,
2Na
.
þ 2e
0
þ
1
2
O
2
! Na
2
O(4:47Þ
The oxygen partial pressures are the same on both sides of the cell so that the
overall cell reaction is given by Eq. (4.45).
4.6.2 Gas-sensors based on electronically conducting
ceramics
The science of gas-sensors based on changes in electronic conductivity of a
semiconducting ceramic is a complex matter depending upon a combination of
surface chemistry and electron transport in mainly transition metal oxides. As is
usual in exploiting electroceramics, the applications’ technology runs ahead of
CERAMICS-BASED CHEMICAL SENSORS 207
Fig. 4.44 Schematic of a CO
2
sensor incorporating sodium carbonate as a ‘linking’
compound. N.B. Because of the small size of the sensor (the outside diameter of the tube is
only approx. 4 mm) equilibration between the atmosphere and sodium carbonate is rapid at
400 8C.
* Nasicon is a generic term for sodium ion conducting oxides containing elements such as Zr, Si, and P.
The values of their conductivities are similar to that of b-alumina.
the basic understanding. The following discussion provides the base for further
study for which the literature sources [25–29] are recommended.
Many semiconducting oxides, for example doped SnO
2
, ZnO, and TiO
2
, show
changes in electrical resistivity in the presence of small concentrations of
flammable gases such as propane, and toxic gases such as carbon monoxide. This
phenomenon has led to the development of a wide variety of gas-sensors which
are finding a rapidly expanding range of applications.
Because of a combination of unique physical properties, namely high
transparency in the visible region of the spectrum, semiconducting characteristics
and good thermal, mechanical and chemical stabilities, tin oxide is exploited for
resistors (see Section 4.2.1), furnace electrodes (see Section 4.1.4) and in
optoelectronic devices (see Chapter 8) as well as dominating in gas-sensor
technology. Unless otherwise stated the discussion is based upon n-type
semiconducting SnO
2
and the reader is referred to Section 4.1.4 for introductory
remarks relating to its solid state science.
The sensors detect gases because of a change in electrical resistance which
accompanies the reaction between adsorbed species such as O
2
,O
and O
2
and
the ambient gases being sensed, the ‘target’ gases. Figure 4.45 illustrates how the
electron distribution in a semiconductor is modified as oxygen interacts with the
surface leading to the adsorption of the most likely species, O
.
(Note. The Debye length (L
D
), although not introduced into the present
simplified discussion, is a parameter frequently referred to in the gas-sensor
literature. It was originally introduced into ionic solution theory and later
applied to semiconductor theory where it is especially applicable to semicon-
ductor/metal and semiconductor/semiconductor junctions. It is a measure of the
distance beyond which the disturbance at the junction has effectively no influence
on the electron distribution and therefore closely related to ‘d’ (see Eq. (4.49)). It
is a material parameter given by L
D
¼(ekT=e
2
c
o
Þ
1=2
where c
o
is the undisturbed
electron concentration, essentially the extrinsic electron concentration in the case
of doped n-type tin oxide, and the other symbols have their usual meaning.)
208 CERAMIC CONDUCTORS
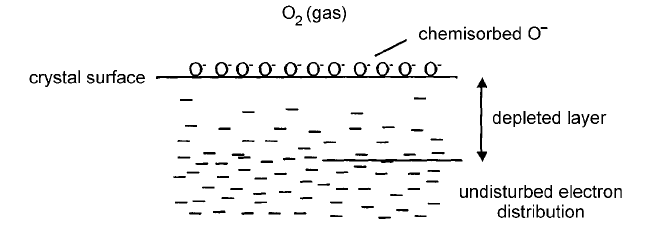
Fig. 4.45 Schematic illustrating the electron-depleted layer at a surface of SnO
2
carrying
chemisorbed oxygen.
With the chemisorption of, for example, oxygen a surface density of electron
acceptor states leads to the establishment of a Schottky barrier. The process is
essentially the same as that which occurs in the case of the PTC thermistor (see
Figs 4.21 and 4.10). The electron potential barrier height (’) is given by
’ ¼ e
2
N
2
s
=2eN
d
(4:48Þ
where N
s
and N
d
are respectively the surface density of ‘acceptors’ and the
volume density of ‘donors’.
Under the same simplifying assumptions as for the PTC thermistor (where the
surface states are shared between two grains) the depleted layer thickness d is
given by
d ¼ð2e’=e
2
N
d
Þ
1=2
¼ N
s
=N
d
(4:49Þ
Summarizing the basics of the gas-sensing mechanism, oxygen molecules from
the surroundings become chemisorbed (that is chemically bonded) to the surface
atoms, the process being described by the reaction:
O
2
ðgÞþ2e
0
¼ 2O
ðadÞ (4:50Þ
As the adsorption proceeds a positive space charge develops in the surface layer
of the oxide as electrons transfer from donor defects to the adsorbed oxygen. The
electrostatic field generated in the surface regions of the crystal tends to oppose
the charge transfer process and eventually it stops. The thickness of the charge-
depleted layer is determined by the availability of electrons able to transfer to the
surface states and by the surface concentration of adsorbed oxygen. Measure-
ments indicate the thickness of depletion layers to lie in the range of a few
nanometres to approximately 500 nm. Reaction of reducing gases at the surface
removes some of the adsorbed oxygen and releases electrons which are then
available for conduction.
The above description is on the basis of the semiconductor being n-type. In the
case of a p-type semiconductor (e.g. Cr
2x
Ti
x
O
3
;0:01 5 x 5 0:45 [29]) then a
decrease in resistance would accompany an increase in the amount of adsorbed
oxygen.
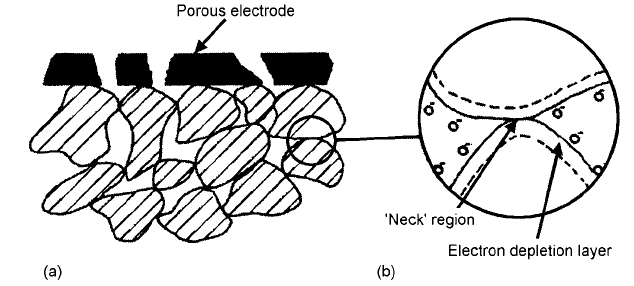
The most common form of gas sensor is based on a porous, sintered ceramic as
shown schematically in Fig. 4.46. As adsorption occurs over the surface of the
semiconductor grains, the barriers to charge transport develop, especially at the
grain boundaries and at particle contact areas (the ‘neck’ regions).
The ideal sensor would have high ‘sensitivity’ and be ‘selective’. That is, it
would show a large resistance change for a small change in the target gas
concentration, and have the ability to discriminate between different gases. It
would also show a reproducible response over the required life-time and be
economically viable.
CERAMICS-BASED CHEMICAL SENSORS 209
The sensitivity of a polycrystalline gas-sensing ceramic depends upon sensor
composition, ceramic particle size (since this determines reactive surface area,
and particularly its relationship to L
D
), sensor geometry, temperature (which
strongly affects adsorption processes and oxide conductivity) and accessibility of
the sensor bulk material (i.e. permeability) to the gas or gases.
It would be inappropriate here to consider the effects of these variables in
detail since optimising sensor performance is still essentially a matter of
‘technical development’ involving not only them but also the sensor housing and
associated circuitry. Optimisation must, of course, be achieved within the
inevitable cost constraints.
The sensor compositions usually include noble metals, particularly palladium
and platinum, since they are found to increase sensitivity through catalysing the
reaction between the oxygen adsorbate and reducing gases. In this role the
additive is said to be acting as a ‘chemical sensitizer’.
The noble metal might also act in a quite different way, as an ‘electron
sensitizer’. Because of its very fine particle size (510 nm) relative to the typical
SnO
2
grain size there are many metal/semiconductor contact points. Also,
because of the high specific surface area (50 m
2
g
1
) of the particulate metal it,
too, presents a large adsorbent surface available to oxygen species, the
adsorption process removing electrons from the metal. Because the work
function of the metal (5.12 eV) is larger than the electron affinity of the
semiconductor (4.45 eV) electrons will flow out of the semiconductor into the
metal, increasing the depletion layer thickness. This is very likely an over-
simplification of what is a complex charge transfer process, and the role of Pt in
influencing the sensitivity to CO of Sb-doped SnO
2
is addressed in detail by L.
Morris and D.E. Williams [29].
210 CERAMIC CONDUCTORS
Fig. 4.46 (a) Schematic diagram of a section through a porous SnO
2
compact with a porous
electrode; (b) detail of the ‘neck’ region between the grains showing the effect of adsorbed
oxygen.
‘Selectivity’ is mainly achieved by ‘trial and error’. It can be engineered
through specific dopants to the tin oxide, for example Fe
2
O
3
to sensitize for NO
2
or V
2
O
5
and Pd to sensitize for CO and other combustible gases, as illustrated
below. It can also be engineered by interposing selective diffusion barriers
between the sensor and the ambient.
‘Sensitivity’ is a strongly microstructure-dependent property, the depletion
layer thickness relative to the particle size of the sensing ceramic being the
particularly important parameter.
It seems likely from Fig. 4.46 that the greatest sensitivity would be achieved
when the particle size is close to twice the depletion layer thickness. Sensor
ceramics therefore have a particle size typically well into the sub-micron range,
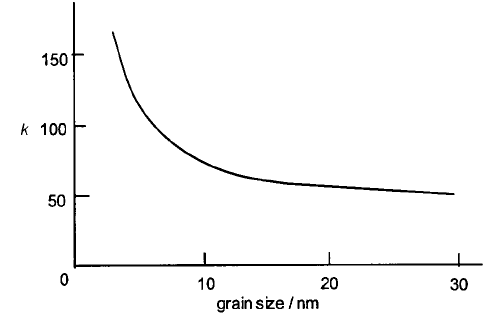
and often in the range 10–100 nm. Figure 4.47 demonstrates the dependence of
sensitivity on grain size, the onset of the steep rise at around 10 nm being
associated with the grain size approximating to the depletion layer thickness.
The now classic ‘Taguchi’ sensor design, dating from the 1960s, is shown in
Fig. 4.48(a) and a present day format in Fig. 4.48(b).
The planar format sensor is most likely to be fabricated by screen-printing, the
sensing layer being typically 20–30 mm thick. The doped tin oxide can be
prepared chemically, for example by precipitating Sn(OH)
4
, together with
hydroxides of the dopants, by passing NH
3
through an aqueous solution of
SnCl
4
together with dopant chlorides. The grain size will depend on the
subsequent calcining temperature and time, typically 800 8C for 1 h.
The screen-printing paste is made up in the manner described in Section 4.2.2
and the patterns printed. The heater element and its leads would typically be
platinum and the interdigitated electrodes gold. As illustrated below (Fig. 4.49)
the heater might also be a composition based on ruthenium oxide (see Section
4.2.2). The contacting tabs are also screen-printed gold.
CERAMICS-BASED CHEMICAL SENSORS 211
Fig. 4.47 Dependence on grain size of the sensitivity (k) of a tin oxide sensor to 100 p.p.m.
hydrogen in air. Adapted from [28]. (k is defined as the sensor resistance measured in air to
that measured in the hydrogen-containing air.)
