Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.

exceed the breakdown strength of air ( 3MVm
71
) causing an arc to form
across the dry band which may grow until ‘flash over’ occurs from line to earth.
This may cause an interruption of power supply to the consumer, or reduced
voltage, and can also result in serious damage to the conductors, towers and
insulators. Both RI and ‘flash-over’ can be alleviated by using glazes having
surface resistivities in the range 1–100 MO/
&
(see Section 4.2).
Radio interference is reduced by coating the insulator surface in the line-
insulator contact area with a semiconducting glaze to smooth the voltage
gradient. Glazes for this purpose usually have a zinc ferrite (see Section 4.4.1)
conducting phase, although antimony-doped tin oxide (see Section 4.1.4) is
sometimes used. Close control of surface resistivity is not necessary.
The effective use of semiconducting glazes to avoid pollution-induced flash-
over is less simple. The glaze needs to have a closely controlled resistivity,
designed to pass a leakage current of approximately 1 mA under the service
voltage. This ensures that the voltage drop across the dry band does not exceed
the air breakdown voltage. The temperature coefficient of resistance must be
small (semiconducting glazes usually have a large negative temperature
coefficient of resisitivity) and the glaze must be very durable. Where insulators
are, broadly speaking, disc-shaped, the current density will vary with the radius
272 DIELECTRICS AND INSULATORS
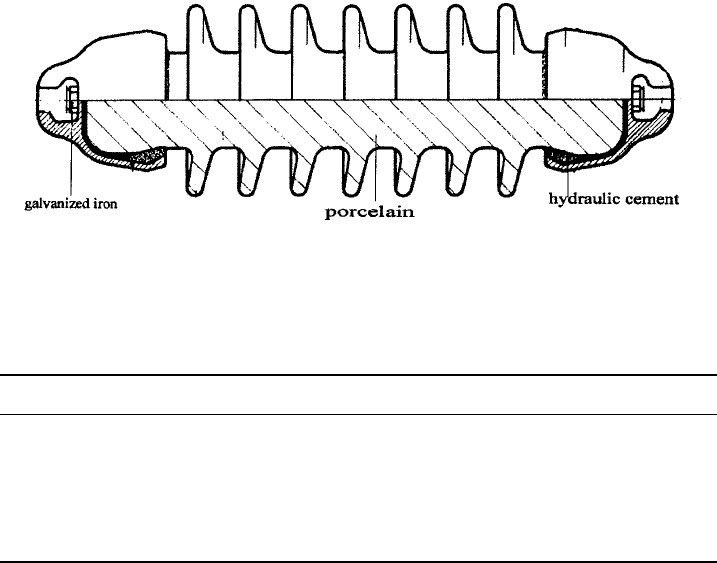
Fig. 5.17 A 36 kV porcelain rod insulator of the variety used to position the overhead power
lines for railways. Overall length 500 mm. (Courtesy of New Zealand Insulators, Temuka,
NZ.)
Table 5.2 Physical properties of electrical porcelains
Property Siliceous Aluminous
Density/Mg m
73
2.4 2.6–2.8
Cross-breaking strength/MPa 100 150
Coefficient of linear expansion/MK
71
6 5–6
Dielectric Strength/MV m
71
25 25
Volume resistivity/O mat208C 10
10
10
10
Tan d/10
4
at 20 8C 150 150
TEAMFLY
Team-Fly
®
leading to a non-linear voltage gradient, and grading of the glaze thickness is
needed to compensate for this. Glazes with an antimony-doped tin oxide
semiconducting phase are almost invariably used. Although they impart to the
insulators better pollution performance than any other type, they are expensive
and produced by only a few manufacturers.
Recently insulators comprising a core of glass fibre-reinforced polymer and a
sheath of a weather-resistant elastomer (‘composite insulators’) have become
available and now have a promising performance history. In particular, some
silicone elastomers have strongly hydrophobic surface characteristics which are
maintained for long periods (420 years) and these give good performance in
polluted conditions because the surface never gets wet. Such insulators are
replacing porcelain for very high voltage transmission, but at lower voltages
(566 kV) are uneconomic unless pollution conditions are severe. Insulators of
toughened glass are also popular, and where a standard design in very large
numbers is demanded have a significant price advantage over porcelain.
Nevertheless, in excess of 500 000 tonnes of porcelain insulators are manufac-
tured world wide each year.
Varistors play an important role in protecting high voltage distribution
equipment (see Section 4.3) against voltage surges caused by lightning strikes,
switching operations and similar hazards. Superconducting fault current-limiters
(see Section 4.7.4) are also used to protect equipment from current surges.
Low-voltage applications of electrical porcelain exist where an insulating
material unaffected by high temperatures or ultraviolet radiation is required.
Holders for discharge lamps such as those used for street lighting, and housings
for fuses are examples. The porcelain types are typically of similar composition
to those discussed above, but the shapes are commonly formed by pressing damp
(15 wt.% moisture) body granules in a steel die.
Talc-based
Talc-based ceramics are important electrical porcelains that have major
crystalline components of the fired ceramic lying in the ternary phase diagram
shown in Fig. 5.18. The principal raw material used for these ceramics is talc
(Mg
3
Si
4
O
11
H
2
O), which is the softest of minerals (no. 1 on the Mohs scale). Talc
is also called steatite, and ceramics using it as a raw material are termed ‘steatite
porcelains’ although the talc contained in them has been changed in crystal
structure during sintering. Blocks of the mineral steatite, which is also known as
soapstone, can readily be machined to shape and, on firing, undergo a change in
crystal structure that results in a very small overall expansion accompanied by a
large increase in hardness and strength.
LOW-PERMITTIVITY CERAMIC DIELECTRICS AND INSULATORS 273
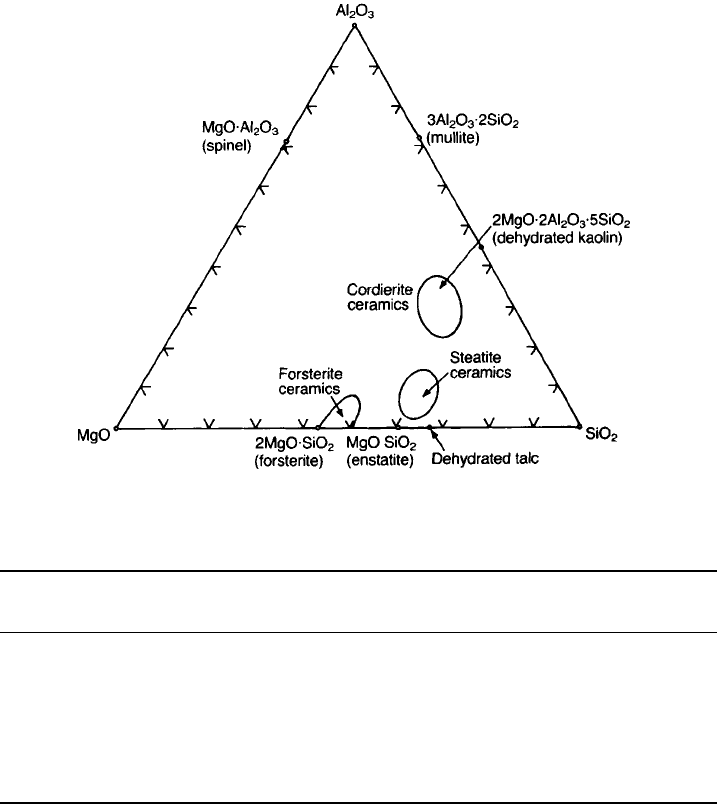
The various porcelain types fall into three regions of the phase diagram and
will be discussed in turn; typical properties are summarized in Table 5.3 and
examples of components are illustrated in Fig. 5.19.
Cordierite ceramics are best known for their low expansion coefficient which
imparts excellent thermal shock resistance. The major phase developed during
firing is cordierite Mg
2
Al
4
Si
5
O
18
(2MgO2Al
2
O
3
5SiO
2
). Clay and talc are the
principal ingredients; the talc content is in the range 20–40 wt% but there are
many modifications to adjust firing temperature and range. Components are
shaped mainly by extrusion and dry pressing, and typical firing temperatures lie
in the range 1150–1400 8C. Uses for the ceramic are many and varied, but it is
274 DIELECTRICS AND INSULATORS
Fig. 5.18 Phases present in the Al
2
O
3
–MgO–SiO
2
system (in wt%).
Table 5.3 Typical properties of common electrical ceramics
Material e
r
tan d/10
74
at 1 MHz
a
L
/MK
71
at 20–1000 8C
l/Wm
71
K
71
at 25 8C
‘Low-loss’ steatite 6.1 7 8.9 3
Cordierite 5.7 80 2.9 2
Forsterite 6.4 2 10.7 3
96Al
2
O
3
9.7 3 8.2 35
99.5BeO 6.8 2 8.8 250
AlN 8.8 5–10 4.5 100
Glass 4–15 2–22 0.8–9 0.5–2.0

particularly suited to applications where there is a need for good thermal shock
resistance combined with high electrical resistivity, for example for high-power
electrical fuse holders and supports for high-power wire-wound resistors and fan-
heater elements.
Steatite ceramics were introduced into electronic components during the 1920s
to meet the demands of the rapidly developing radio industry. Their principal
attribute is low dielectric losses which are necessary for higher frequencies. As for
the cordierite ceramics, the major constituents are clay and talc but with the
composition adjusted so that enstatite, a modification of MgSiO
3
, crystallizes. A
typical composition is about 85 wt% talc, 15 wt% clay and 2 wt% calcium
carbonate (chalk). The calcium carbonate acts as a flux and is used instead of
feldspar to avoid the introduction of alkali metals which would increase dielectric
losses. The shrinkage on sintering can be adjusted by calcining up to three-quarters
of the talc and varying the proportion of calcined material to compensate for
changes in the raw materials. The soft raw talc has a lubricating action that greatly
reduces tool wear during fabrication compared with other ceramic powders.
Most steatite ceramics are either dry mixed and pressed, or wet-mixed, spray-
dried and pressed, but they can also be wet-mixed and extruded. Typical firing
temperatures lie close to 1300 8C. Many small parts are made for the electronics
components industry where low dielectric losses are required, for example for tie-
bars and other parts for ganged capacitors, small trimmer capacitors, high-power
capacitors (see Section 5.6.3), coil formers, lead-throughs and substrates for
LOW-PERMITTIVITY CERAMIC DIELECTRICS AND INSULATORS 275
Fig. 5.19 Selection of magnesium silicate parts (parts supplied by Morgan Advanced
Ceramics).
some types of resistor and circuit. The ceramic is also commonly supplied with
nickel metallizing for terminal connector blocks.
The sintered bodies consist of crystals of protoenstatite (or mesoenstatite),
which is a polymorph of MgSiO
3
, a small amount of cordierite and a continuous
glassy phase surrounding the crystalline phases. Protoenstatite is the thermo-
dynamically stable phase above 985 8C and is stable at room temperature
provided that it is in the form of small crystals (510 mm) covered with a layer of
glass. However, prolonged heating at 500 8C converts protoenstatite into
clinoenstatite, which is the thermodynamically stable form below 985 8C. The
dimensional changes in the crystals due to the transformation result in an overall
expansion and a marked decrease in strength. Badly formulated material, or
material that has been overfired so that excessive crystal growth has occurred
and the amount of glass has been diminished, may be unstable under humid
conditions at room temperature. In bad cases the surfaces of bodies become
white and blotchy, they expand and their strength is reduced. The stability of
steatite components can be tested by exposing them to steam at 100 8C for 24 h
and then checking that their cross-breaking strength is unaltered.
Despite this potential instability, steatite ceramics have been widely used and
adequate precautions in manufacture have prevented all but a very few cases of
deterioration during component lifetimes of many years in a wide variety of
environments.
In forsterite ceramics the mineral forsterite (Mg
2
SiO
4
) crystallizes. They have
excellent low-dielectric-loss characteristics but a high thermal expansion coefficient
which imparts poor thermal shock resistance. During the 1960s they were
manufactured for parts of rather specialized high-power devices constructed from
titanium and forsterite and for which the operating temperature precluded the use
of a glass–metal construction. The close match between the thermal expansion
coefficients of titanium and forsterite made this possible. Today alumina–metal
constructions have completely replaced those based on titanium–forsterite and the
ceramic is now manufactured only to meet the occasional special request.
5.5.2 Alumina
Structure, fabrication and properties
Alumina is a widespread component of siliceous minerals. It occurs as single
crystals in the form of sapphire, and with chromium ‘impurity’ as ruby, and in
large deposits as the hydrated oxide bauxite (Al
2
O
3
2H
2
O). The dehydration of
this and other hydrated oxides at temperatures below 1000 8C leads to the
formation of g-Al
2
O
3
which is converted to a-Al
2
O
3
above 1000 8C. The
transformation is irreversible and the a-polymorph is stable from absolute zero
to its melting point at 2050 8C.
276 DIELECTRICS AND INSULATORS

a-alumina, in powder form, is a byproduct of aluminium production. Bauxite,
which is naturally contaminated with other oxides, principally SiO
2
and Fe
2
O
3
,is
purified by the Bayer process. This involves the dissolution of a low-silica-
content ore in caustic soda (NaOH) solution under pressure, filtering off the
insoluble hydroxides (mostly of iron), and then precipitating Al(OH)
3
by diluting
the solution at atmospheric pressure and adding a small amount of Al(OH)
3
as a
nucleating agent. Most of the silica remains in the solution. The hydroxide is
washed and then calcined at temperatures in excess of 1000 8C to produce a-
Al
2
O
3
. Because of the involvement of sodium in the extraction process the
calcined alumina contains 0.1–0.2% Na
2
O. This impurity has important
consequences as far as the manufacture of electrical ceramics is concerned and
care is exercised to keep it to a minimum. Practically all the powder for the
production of alumina ceramics is produced in this way, but a purer product, for
instance for growing artificial sapphire crystals, can be made by preparing
ammonium alum (NH
4
Al(SO
4
)
2
12H
2
O) by dissolving 99.999% aluminium metal
in sulphuric acid and neutralizing the excess acid with ammonia. The alum is
allowed to crystallize, which helps to purify it, and is then calcined below 1000 8C
to yield g-Al
2
O
3
, which is preferred for some methods of sapphire preparation, or
at higher temperatures to give the a form.
The crystallites in alumina ceramics are mainly a-Al
2
O
3
, mineralogically
known as corundum (a synonym for sapphire). Corundum is 9 on the Mohs scale
of hardness, i.e. it is next hardest to diamond, at 10. Examples of the
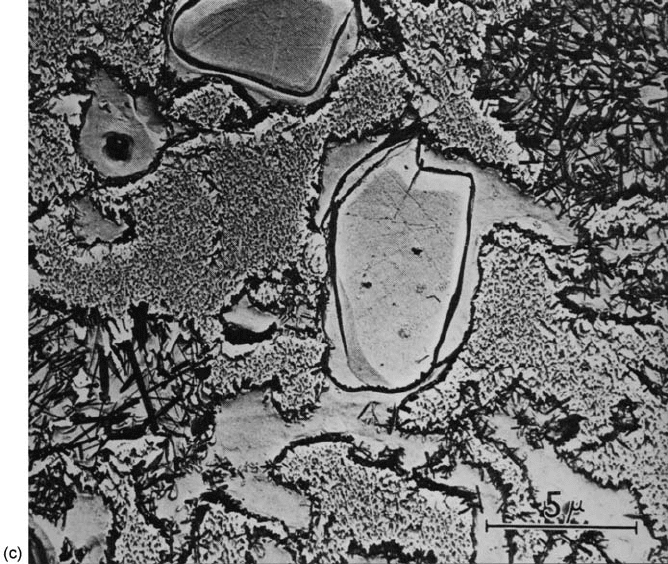
microstructures of a high-purity alumina and a debased alumina are shown in
Figs 5.20(a) and 5.20(b) respectively. The latter consists of a-Al
2
O
3
crystallites
embedded in a glass–crystalline matrix usually composed of calcium and
magnesium silicates.
The less pure aluminas are blended with silicates so that they can be sintered at
1350 8C or less. The highest-purity materials require a temperature of 1750 8Cat
atmospheric pressure or hot-pressing. As cost increases with sintering
temperature, the grade used in practice is usually the least pure that has
adequate properties.
Table 5.4 gives the properties of a range of aluminas and shows the
improvement in properties with purity. The most striking change is in thermal
LOW-PERMITTIVITY CERAMIC DIELECTRICS AND INSULATORS 277
Table 5.4 Typical values of the electrothermal properties of various grades of alumina
ceramic
Alumina content/% 85 90 96 99.5 99.9
e
r
at 1 MHz 8.2 8.8 9.0–9.3 9.7 9.8–10.1
tan d/10
74
at 1 MHz 9 4 1–3 3 0.4–2
Resistivity/O m at 300 8C 4.6 610
8
1.4610
9
3.1610
9
2.0610
9
1.0610
13
Thermal conductivity/
Wm
71
K
71
at 20 8C
14 16 24–35 35 40

278 DIELECTRICS AND INSULATORS
conductivity which indicates the superiority of the purer grades in applications
where the transfer of heat is of importance, e.g. in substrates, discussed below.
The wide range of thermal conductivities reported for 96% Al
2
O
3
requires
some explanation. Thermal conductivity depends on the transfer of lattice
vibrational energy in the form of phonons. Defects in the lattice inhibit this
process, and it is found that the incorporation of impurities much in excess of
0.1 mol.% in the lattice appreciably reduces the thermal conductivity. However,
if impurities are present as separate phases and have not entered the lattice of the
main phase their effect will be in accordance with one of the mixture rules (cf.
Section 2.7.4). Which rule will depend on the distribution of the minor phases,
for example whether they form continuous layers round each grain of the main
phase or are present in separate discrete regions. High conductivity only occurs
in 96% alumina when the raw materials and processing conditions are such that
the additives to lower the sintering temperature do not enter the Al
2
O
3
lattice in
more than minor quantities.
There is considerable uncertainty about the room temperature values of the
electrical resistivity of good insulators; the best estimates are probably derived by
LOW-PERMITTIVITY CERAMIC DIELECTRICS AND INSULATORS 279
Fig. 5.20 Microstructures of (a) 99.9% Al
2
O
3
(Courtesy of E.W. Roberts), (b) 95% Al
2
O
3
(Courtesy of R. Morrell), (c) chemically etched electrical porcelain: note the partially dissolved
quartz grains and mullite precipitates (Courtesy of S.T. Lundin).
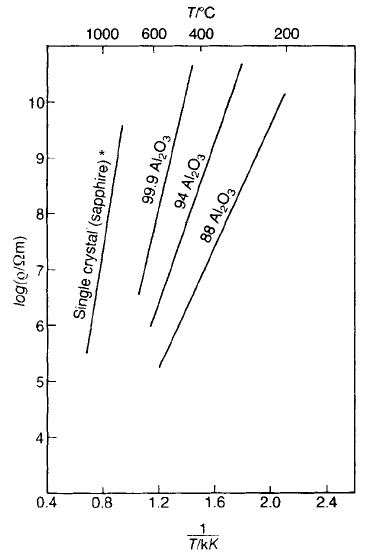
extrapolation of the linear log r–1/T plots of the type shown in Fig. 5.21. Also,
the reliable measurement of the resistance of very high resistance specimens is
made difficult because of the relatively low resistance of the ceramic surface due
to adsorbed impurities, and even that of the surrounding air or other gas. This
can be circumvented by using a properly guarded measurement method.
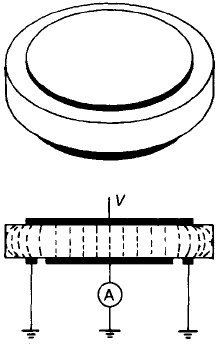
The principle of a guarded measurement is illustrated in Fig. 5.22 which shows
a guard ring around one of the measuring electrodes. When the effects of gas
conduction have to be guarded against the ring must be extended into a cylinder.
The guard ring and one of the centre electrodes are connected to the voltage
supply but only the current through the central electrode is measured.
Satisfactory measurements can be made in this way at 200 8C and above, but
with high resistivities and lower temperatures the polarization and reordering of
defects and impurities result in an initially high current that takes many hours to
fall to a steady state.
Very careful measurements have been made by F.G. Will et al. [4] on high
purity (535 p.p.m. impurities) sapphire over the temperature range 400–1300 8C
and at an oxygen partial pressure of 1 mPa (10
711
atm). They find the
conduction mechanism to be electronic with negligible ionic contribution. The
280 DIELECTRICS AND INSULATORS
Fig. 5.21 Dependence of resistivity on temperature for various grades of Al
2
O
3
. *[4]
measured activation energy of 4.8 eV is consistent with the determined optical
band gap of 9.5 eV.
In the case of polycrystalline alumina the matter is complicated by charge
transport along grain boundaries. It is generally found that s increases with both
large and small p
O
2
values, with a minimum at about 10 mPa (10
4
atm) at about
1600 8C. This behaviour seems to be independent of the type of dopant (acceptor
or donor) or whether the material is single crystal or polycrystalline. Figure 5.23
illustrates the expected general behavioural pattern, but the positions of the
boundaries defining the ‘fields’ for the various mechanisms is largely speculative
and will, of course, depend strongly on the natures and amounts of impurities
present.
The effects of deliberately added donors, such as titanium, and acceptors, such
as iron and magnesium, on electrical conductivity have been studied. Doping
with aliovalent ions affects the concentration of intrinsic defects and, in
consequence, the diffusivity of Al and O. In the case of variable-valency dopants,
changes in p
O
2
change the fraction of dopants in the aliovalent state and the
nature and concentration of the defects. For example, the dopant Ti substitutes
for Al and, in the fully oxidized state, produces the defect Ti
.
Al
, compensated by
V
Al
000
, so that
½Ti
.
Al
¼½3V
Al
000
ð5:16Þ
and ionic conductivity by transport of V
Al
000
is encouraged. However, at low p
O
2
Ti
is in the isovalent state, Ti
3+
, and electron conduction occurs according to
Ti
Al
Ð Ti
.
Al
þ e
0
ð5:17Þ
LOW-PERMITTIVITY CERAMIC DIELECTRICS AND INSULATORS 281
Fig. 5.22 Schematic diagram of a guard ring and circuit for measurements of high resistivity.
