Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

synthesis of ammonia. Much of this synthetic nitrogen is used in the manufacture of fer-
tilizer, which goes directly into increasing the primary productivity of crops.
Many of the transformations that make up the nitrogen cycle are redox reactions.
Nitrogen is present in its most reduced state as organic nitrogen or as ammonia. (The Kjel-
dahl nitrogen determination measures the sum of these two.) The most oxidized form is
nitrate. Intermediate between these extremes are nitrite, NO, N
2
O, N
2
,N
2
H
2
, and N
2
H
4
,in
order of increasing reduction (Figure 13.17). The primary biologically mediated steps in
the nitrogen cycle are discusse d next (Figures 14.7 and 14.8).
Fixation: the conversion of N
2
to ammonia. The prokaryotic aquatic blue-green
bacteria (cyanobacter) are photosynthetic and get their energy directly from the sun.
Bacteria such as Rhizobium an d Bradyrhizobium beneficially infect the roots of
leguminous plants such as alfalfa, clover, peas or beans, and some lichens and marine
algae. Those plants sacrifice some of their growth to share the energy they captured
from the sun with the nitrogen-fixing bacteria, in return for a rich source of available
nitrogen. Azotobacter is a free-living bacterial nitroge n fixer. Fixation requires a
considerable amount of energy. Legume bacteria use about 40 kcal (equivalent to about
10 g of gluc ose) to fix 1 g of nitrogen. Free-living nitrogen fixers require about 10 times
as much.
Ammonification: the conversion of organic nitrogen to ammonia. This is performed by
saprotrophic organisms which release nitrogen in excess of their own n eeds as
ammonium.
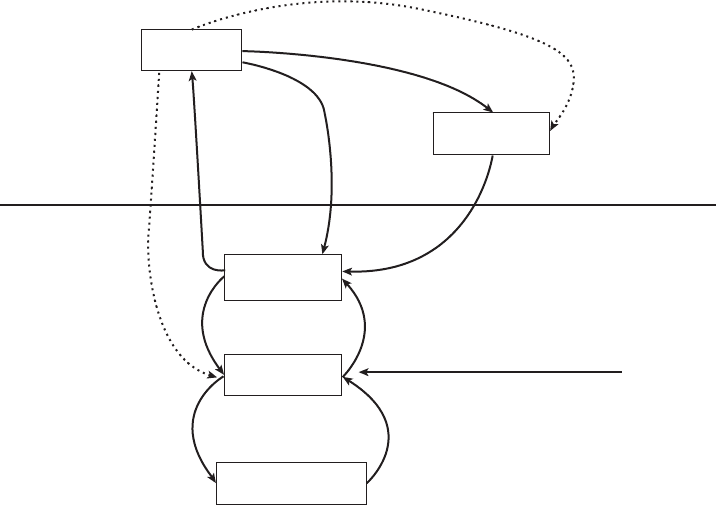
Atmosphere
Land and water
–
NH
3
N
2
NO
3
–
NO
3
NO
x
Organic N
Lightning
4
Industrial
fixation
40
Fossil fuel
Combustion
21
from air to: Land Sea Total
Acid rain 17 9 26
Dry deposition 15 4 19
Forest fires
12
Assimilation
1000
Mineralization
(Ammonification)
Nitrification
Denitrification
Land 107-161
Sea 40-120
Biofixation
Land 139
Sea 10-90
Volcanism 5
Figure 14.7 Fluxes in the global nitrogen cycle. Estimated fluxes in Tg/yr. Ammonia, organic
nitrogen, and other forms also enter the atmosphere and oxidize or fall with rain. Dotted line arrows
represent primarily anthropogenic fluxes. (From Odum, 1987.)
458
ECOLOGY: THE GLOBAL VIEW OF LIFE
Nitrification: the chemoautotrophic conversion of ammonia first to nitrite, and
subsequent conversion of nitrite to nitrate. The first step is accomplished by bacterial
genuses such as Nitrosospira and Nitrosomonas. The second step is done by bacteria such
as Nitrobacter, and in marine environments by Nitrococcus. Nitrification is also
responsible for the production of nitric oxide (NO) and nitrous oxide (N
2
O).
Denitrification: the conversion of nitrate and nitrite, through a series of steps,
ultimately to nitrous oxide and N
2
. This process is performed by numerous facultative
heterotrophic bacteria when oxygen is limiting. Den itrification can occur even in
nominally aerobic environments when anoxic micro environments are present. This can
occur, for example, in clumps of soil where organic matter is present to deplete the
oxygen locally and provide energy for denitr ification, or in the interior of flocs in the
activated sludge wastewater treatment process. Nitrate serves as a substitute for oxygen as
an electron acceptor for heterotrophic respiration.
Assimilation: the conversion of inorganic nitrogen to organic forms such as amino
acids. Plants can absorb ammonia or nitrate, but mostly they use nitrate. Nitrite is toxic to
plants. Plant fertilizers usually contain ammonia or urea, which breaks down to ammonia.
The ammoni a is then nitrified. After absorption, the plant converts the nitrate back into
ammonia before converting it into amino acids or other organic nitrogen compounds.
The nitrous oxide produced by denitrification can be oxidized photochemically in the
stratosphere to nitric oxide, which then reacts with ozone to form NO
2
. Along with the
NO
x
species from nitrification, these ultimately form nitrate again. This is deposited with
precipitation and is part of the problem of acid deposition (Section 15.7). About 25% of
the total NO
x
-N production is thought to originate in lightning and in biological reactions
in soil. The rest is anthropogenic, including fossil-fuel combustion and forest burning.
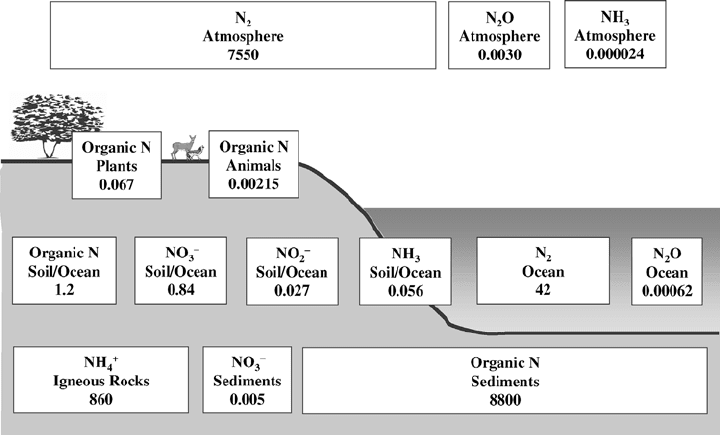
Figure 14.8 Another view of the global nitrogen cycle, showing storage reservoirs of nitrogen.
Values are kg/m
2
of the Earth’s surface. (Based on Whittaker, 1975.)
FLOW OF MATTER IN ECOSYSTEMS 459

Ammonia is also released by microbial activity in the soil. Much is reabsorbed by
plants, which also absorb nitrate deposited on leaves. Ammonia in the air can neutralize
some o f the atmospheric sulfate, the other major acid rain component. Rainfall can also
contain significant quantities of dissolved organic nitrogen (D ON). The source of the
DON may be from ocean spray tranported to the land.
Overall, rainfall contributes about 17 Tg N/yr in the form of nitrate to the land, plus
15 Tg N/yr as ammonia and 9 Tg N/yr as DON. Dry deposition contributes another 16 Tg/
yr, mostly nitrate, for a total flux to land of 57 Tg/yr. The oceans receive a total of at least
20 Tg/yr.
The uncertainty in estimates for global nitrogen fixation and denitrification will need to
be reduced before it can be determined if fixation and denitrification balance each other
(Figure 14.7 and Table 14.3). The biological fixation on land includes an anthropogenic
flux of 44 Tg/yr due to human cultivation of leguminous crops.
The nitrogen cycle is an intere sting example of the Gaia hypothesis at work. Lovelock
points out that one of the effects of life is the maintenance of chemical disequilibrium in
the environment. If you turned off all biotic reactions, chemical equilibrium would even-
tually be achieved among the various species in the environment. In the case of nitrogen,
the atmospheric O
2
would combine with its N
2
to form nitrate, which would be dissolved
in the oceans. It is commonly understood that the oxygen in the atmosphere is maintained
there by photoautotrophs. What is less well known is the fact that most of the nitrogen is
in the atmosphere instead of the ocean, because of denitrifiers. Without life, Earth’s atmo-
sphere might resemble that of Mars, which is 95% CO
2
.
Industrial fixation of nitrogen by the Haber–Bosch process has altered human ecology
significantly. Each year, 175 million tons of nitrogen flow into the world’s croplands, half
of which is assimilated by cultivated plants. Synthetic fertilizers provide about 40% of all
nitrogen taken up by these crops. The crops furnish about 75% of all nitrogen consumed
by humans (the rest comes from fishing and from grazing cattle). Thus, about one-third of
the protein in humanity’s diet depends on synthetic nitrogen fertilizer. This has undoubt-
edly facilitated the tripling of the world’s population that occurred in the twentieth century.
14.2.5 Sulfur Cycle
Roughly 90% of the world’s supply of sulfur (about 1016 metric tons) is held in sediments
and rocks and the remaining fraction is largely found dissolved within our oceans, which
TABLE 14.3 Global Nitrogen Fluxes of Fixation and Denitrification
(Tg N/yr)
Source Fixation Denitrification
Land (biological) 139 107–161
Ocean (biological) 10–90 40–120
Atmosphere (lightning) 4
Forest fires 12
Industrial, including fertilizers 40
Fossil-fuel combustion 22
Total 227–307 147–281
Source: Berner and Berner (1987).
460 ECOLOGY: THE GLOBAL VIEW OF LIFE
in turn represents the dominant reservoir for the majority of sulfur being cycled within our
biosphere. The cycle reactions for sulfur are depicted schematically in Figure 13.25, and
the global cycle in Figure 14.9.
Sulfur is essential for life largely because it is a component of two of the amino
acids, cysteine and methionine. The sulfur cycle may be the cycle that is most affected
by human activities. Human emissions of CO
2
and nitrogen are about 5 to 10% of natural
emissions. However, human activity releases about 160% as much sulfur as nature
does.
Like nitrogen, sulfur cycles through a number of oxidation states. The most oxidized
form, sulfate, serves as an alternative electron acceptor in respiration. It is generally used
only if both oxygen and nitrate are absent, since it is less energetically favorable to organ-
isms. Sulfate is the form of sulfur taken up by the primary producers, and thereby intro-
duced into the food chain.
The most reduced inorganic form is sulfide. The toxic volatile compound hydrogen
sulfide is commonly associated with anaerobic environments that have large amounts
of organic matter, such as marsh sediments, anaerobi c digesters in wastewater treatment
plants, and petroleum deposits. In these the organic matter serves as a reducing agent, first
reducing oxygen to CO
2
, any nitrate is denitrified, and then all sulfate is reduced to H
2
S.
Sulfide is also found in deep groundwater aquifers, having been reduced by either soil
organic matter or by ferrous iron. If ferrous iron is present, the sulfide will form the non-
volatile, low-solubility precipitate iron sulfide (FeS). Sulfide is also used by photoauto-
trophic bacteria as an electron donor in place of H
2
O in a process similar to
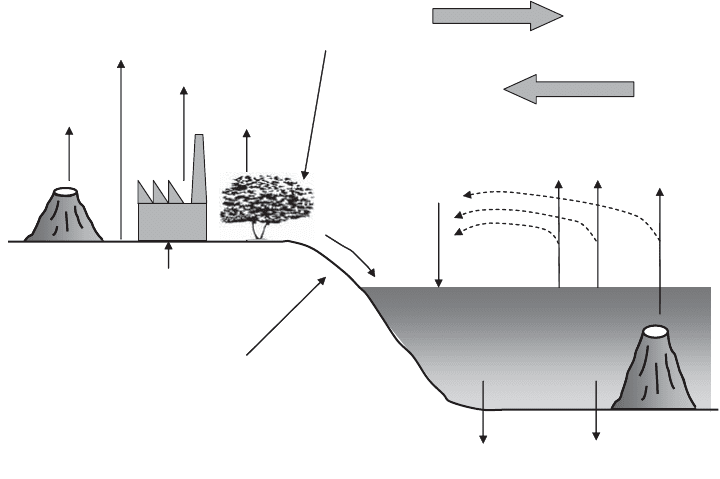
Mining
149
Volcanism
10
Dust
20
Industrial
93
Biogenic gases
22
Wet and dry
deposition
84
Rivers
213
Weathering and erosion
72
Deposition
258
Sea
salt
144
Biogenic
gases
43
Volcanism
10
Pyrite
39
Hydrothermal
sulfides
96
Aerial transport to sea
81
20
Aerial transport to land
Figure 14.9 Global sulfur cycle. (Based on Krebs, 1994.)
FLOW OF MATTER IN ECOSYSTEMS 461
photosynthesis. Sulfur here plays the role of oxygen, and instead of O
2
being produced,
elemental sulfur is the product. In aerobic environments, reduced forms of sulfur are
rapidly oxidized to sulfate by chemoaut otrophic bacteria. Thus, the sulfur cycle is closely
linked to the carbon cycle.
The sulfur cycle is also linked to the phospho rus cycle in aquatic systems (Secti on
14.2.6). When iron sulfide is oxidized in aquatic sediments, sulfate is released and phos-
phorus precipitates with the iron, becoming unavailable to organisms. Under anoxic con-
ditions, the iron is reduced, releasing the phosphorus to the water, and the iron precipitates
again as a sulfide.
Because reduced forms of sulfur are often associated with deposits of fossil fuels and
mineral ores, combustion and smelting of these resources often results in emissions of
sulfur dioxide to the atmosphere. This is rapidly oxidized to sulfate. In the absence of
alkaline species, this is present in the form of sulfuric acid aerosols. These are easily
washed out of the atmosphere by precipitation, which as a result has greatly increased
acidity and reduced pH. The result is called acid rain or acid precipitation. Acid preci-
pitation has several ecological impacts on aquatic and terrestrial ecosystems where it falls.
These are detailed in Section 15.7.
The ocean is also a significant source of atmospheric sulfur, with a biological origin.
Marine phyto plankton produces volatile dimethyl sulfide [(CH
3
)
2
S]. In the air this oxi-
dizes rapidly to sulfate, much of which is washed back into the sea.
14.2.6 Phosphorus Cycle
The phosphorus cycle differs from those of carbon, nitrogen, and sulfur in several ways:
There are fewer steps, there is no change in oxidation state, there is no significant atmo-
spheric component, and it tends to cycle locally. For these reasons, it is easier to study.
Phosphorus is present in three main forms: free or orthophosphate; polyphosphate,
which is a polymer of orthophosphate; and organic phosphate. Although present in living
things in much smaller quantities than carbon or nitrogen, its importance is clear from
biochemistry, as phosphate forms the backbone of the DNA molecule and is central to
energy metabolism in cells. Phosphate is the most common limiting nutrient in aquatic
ecosystem. Humans disturb the phosphorus cycle in aquatic systems by discharging was-
tewater containing phosphate. This stimulates excessive cyanobacter, algae, and plant
growth, which later die and deplete the water of oxygen. Aquatic system s with high nutri-
ent loading are called eutrophic (Section 15.2.6).
Figure 14.10 shows one particular phosphorus cycle based on measurements in a salt
marsh using radioactive tracers. Note that the various compartments do not have to be in
steady state, as defined by equation (14.3). Each compartment may be importing or
exporting phosphorus from the ecosystem. An energy flow diagram was also done for
this ecosystem. An interesting conclusion stemmed from the observation that the filter-
feeding mussels were more important for their role in nutrient cycling than for energy
processing. In other words, they were more import ant for recycling phosphorus than as
a food source for other organisms.
Sediments store and release phosphate to the water, depending on oxygen concentra-
tions in aquatic systems. When oxygen is available, phosphate is absorbed by ferric
hydroxide. When oxygen is limiting, the ferric iron is reduced to ferrous form, and the
phosphate is released. This occurs seasonally in temperate-zone lakes.
462 ECOLOGY: THE GLOBAL VIEW OF LIFE
14.2.7 Cycles of Other Mine rals
Numerous other minerals are required by living things, including potassium, calcium,
and magnesium. Others are considered micronutrients, such as iron, chlorine, manganese,
boron, zinc, copper, and molybdenum. Yet others, such as silicon, sodi um, and cobalt, are
required only by some organisms. Some are toxic, such as mercury and arsenic.
Iron is the sixth most common mineral element in the lithosphere (at approximately
5%), surpassed only by silica, calcium, magnesium, aluminum, and sodium. With an esti-
mated total mass of 2 10
17
metric tons of iron, there is roughly 10 times mor e iron than
carbon in the lithos phere. However, the presence of iron in the biosphere is several thou-
sand times lower than that of the macroelements, and its atmospheric fraction would be nil
were it not for dust entrained by the wind. Its low solubility under aerobic conditions lim-
its its availability. The two main forms are ferric (Fe
3þ
) and ferrous (Fe
2þ
). The solubility
of ferric iron at pH 6.0 in waters of moderate alka linity is about 0.13 mg/L. As a result, it is
a limiting nutrie nt in some ecosystems, such as the open ocean. Ferrous iron is much more
soluble, at about 20 to 30 mg/L, but this form is converted to ferric in the presence of
oxygen. Ferrous iron may be found in deep groundwaters or wetland sediments where
oxygen is depleted. When oxygen is present at pH levels above 6, ferrous iron is oxidized
abiotically. At lower pH levels this occurs very slowly. But chemolithotrophic bacteria
such as Thiobacilus ferrooxidans and Gallionella ferruginea can take over, harvesting
energy in the process. Others, such as Sphaerotilus and Leptothrix perform the reaction
but seem not to obtain energy from it.
One of the most thoroughly studied ecosystems in the world is the Hubbard Brook
Experimental Forest in New Hampshire. A calcium budget developed for this forest
found that it receives 3 kg/ha yr from rainfall and 5 kg/hayr from weathering of bedrock.
The output from the watershed via streamflow carrie s the sum of these, 8 kg/ha yr. The
biota and abiotic reservoirs exchange 50 kg/ha yr between themselves. This shows how
the biological community recycles the mineral many times faster than the overall through-
put of the ecosystem.
Water
30
Sediments
500,000
Filter feeders
175
Spartina
660
Detritus
10,000
9.8
16.4 ( avg)
16.4 ( avg)
6
6
9.8
Figure 14.10 Phosphorus cycle from a Georgia salt marsh. Reservoirs are in mg P/m
2
, fluxes are
in mg P/m
3
day. Uptake by Spartina and release from detritus vary seasonally as shown. (Based on
Odum, 1987.)
FLOW OF MATTER IN ECOSYSTEMS 463
Human activity has greatly affected the transport of many minerals. It has even created
new ones. Radioactive strontium-90 is created by fallout from nucl ear explosions. It
behaves like calcium. Radioactive cesium-137 behaves like potassium and is rapidly
recycled by organisms. Industrial activities have resulted in the release to the environment
of otherwise scarce minerals, including mercury and chromium. Acid rain has increased
leaching of aluminum into aquatic ecosystems, affecting fish life. Aluminum in soil water
also decreases absorption of magnesium by plant roots. Recall that magnesium atoms are
part of the chlorophyll molecule. Aluminum is added in the treatment of drinking water in
the form of alum, although most is then removed.
14.2.8 System Models of Cycles
The cycling of nutrients among compartments can be modeled mathematically using sim-
ple rate expressions. Assuming steady-state nonreactive (conservative) systems, the
expressions become even simpler. We illustrate with an example involving nitrogen bal-
ances, first as a linear steady-state system and then as a nonlinear system in both steady-
state and dynamic conditions. The models can be used to explain and predict the distribu-
tion of nutrients in ecosy stems.
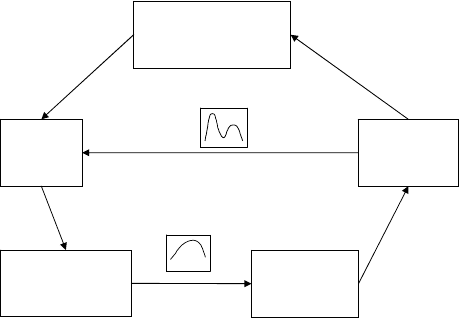
Consider the system in Figure 14.11 which represents major nitrogen components in
an aquat ic system. The four compartment s are: particulate nitrogen, which represents
phytoplankton; dissolved organic nitrogen (DON), which is excreted by the phyto-
plankton; ammonia, produced by mineralization of the DON; and nitrate, produced
by nitrification. Notice that the phytoplankton can utilize both nitrate and ammonia
nitrogen.
The concentration of each species is represented by X and the flux between compart-
ments by J. Each flux is assumed to be proportional to the concentration of the source
species (first order kinetics). For example, the uptake of nitrate is assumed to be propor-
tional to nitrate concentration: J
1
¼ k
1
X
1
. This makes the system linear. The constant of
proportionality is k. The system is set up by writing the mass balance equations for each
X
2
Particulate N
X
4
Ammonia
X
1
Nitrate
X
3
DON
J
1
= k
1
X
1
J
2
= k
2
X
2
J
3
= k
3
X
3
J
4
= k
4
X
4
J
5
= k
5
X
4
Figure 14.11 Simplified nitrogen cycle in the Bay of Quinte. (Based on Ricklefs, 1993.)
464
ECOLOGY: THE GLOBAL VIEW OF LIFE

compartment based on equation (14.3):
Nitrate : J
1
þ J
5
¼ 0 k
1
X
1
þ k
5
X
5
¼ 0
Particulate N : J
1
J
2
þ J
4
¼ 0 k
1
X
1
k
2
X
2
k
4
X
4
¼ 0
DON : J
2
J
3
¼ 0 k
2
X
2
k
3
X
3
¼ 0
Ammonia : J
3
J
4
J
5
¼ 0 k
3
X
3
k
4
X
4
k
5
X
5
¼ 0
ð14:6Þ
These equations form four equations with four unknowns. However, they are not inde-
pendent equations. That is, one equation could be derived by combining the others. To see
how this happens, write the equations for two compartments with two fluxes. You will see
that the two mass balances are identical. Another way to see that the equations as given do
not provide a useful solution is to recognize that one possible solution is to set all the
concentrations to zero. In fact, an infinite number of solutions are possible, depending
on the total concentration in the system. Thus, to find a unique solution we must specify
the total concentration, M; then the individual X’s will represent fractions of the total:
X
1
þ X
2
þ X
3
þ X
4
¼ M ð14:7Þ
If we replace one of the four mass balance expressions, say the nitrate equation, with
this equation, we will have our four independent expressions. To solve we express them in
matrix form:
11 1 1
k
1
k
2
0 k
4
0 k
2
k
3
0
00 k
3
k
4
k
5
2
6
6
4
3
7
7
5
X
1
X
2
X
3
X
4
2
6
6
4
3
7
7
5
¼
M
0
0
0
2
6
6
4
3
7
7
5
ð14:8Þ
The coefficients must be obtained by experimental methods or by fitting the model to
field measurements of the X’s. The system could then be solved by standard matrix solu-
tion methods, such as Gaussian elimination. Matrix systems such as this can also be
solved easily usin g spreadsheet software (see Table 14.4 and Problem 14.6).
The linear system described above is powerful but limited. Suppose that we tried to
make it more realistic by improving the model for flux J
1
, nitrate uptake by phytoplankton.
TABLE 14.4 Nitrogen Cycle Mass Balance Calculation
Process Coefficient
Nitrate uptake (k
1
)16
Excretion (k
2
) 4.1
Mineralization (k
3
)2
Ammonia uptake (k
4
) 1.6
Nitrification (k
5
) 3.1
Form of nitrogen Fraction of total mass
Nitrate-N 0.081
Particulate-N 0.234
Dissolved organic N 0.481
Ammonia-N 0.204
FLOW OF MATTER IN ECOSYSTEMS 465

In the linear model we assumed that this was proportional to nitrate concentration. How-
ever, it would be reasonable to assume that it would also be proportional to phytoplankton
concentration. Thus, a better model would be J
1
¼ k
1
X
1
X
2
. This is a second-order expres-
sion. When we insert it in our system, we can no longer create the matrix as we did above.
We might make similar changes to the equat ions for other fluxes, and might even include
Monod-type expressions.
These changes produce a set of nonlinear simultaneous algebraic equations. Such a
system can be solved by Newton’s method for systems of equations. An alternative and
somewhat easier approach is to write the unsteady-state mass balances for each compart-
ment, then simulate a sufficient period of time until steady- state is practically achieved.
For example, in this case the equation for X
1
would be:
dX
1
dt
¼ k
1
X
1
X
2
þ k
4
X
4
k
2
X
2
ð14:9Þ
The result is a system of nonlinear ordinary differential equations (ODEs), which must
be solved numerically, such as by Euler’s method or Runge–Kutta methods. Initial values
must be specified for the X’s. These can be estimates or solutions obtained from the linear
case. Using the ODE model allows simulation of dynamic effects of disturbances. For
example, the mode l could be used to determine the changes in phytoplankton populations
that would result from a sudden addition of DON and ammonia, such as from a sewage
spill.
Models of these kinds have been developed to simulate complete ecological systems.
For example, models have been created for proposed closed ecological life support sys-
tems (CELSSs) to grow food and recycle wastes in support of human crews on long-term
space missions. Global models have been used to make projections on economic
resources such as food and minerals. Based on current scenarios for population growth
and resource consumption, these models predict a catastrophic resource depletion some
decades in the future. However, they do not take into account social responses to deple-
tion, such as the development of new technologies, reso urce switching, or discovery of
new resource s. Nevertheless, the models make clear that our current world economy is
not sustainable.
14.3 FACTORS THAT CONTROL POPULATIONS
We have just seen how energy and materials move through the ecosystem. Recall that our
primary concern in ecology is to under stand the distribution and abundance of organisms.
Ultimately, this will relate to organisms’ ability to gather and utilize resources. This, in
turn, will depend on the availability of the resources in the environment, on the presence
of other conditions favorable to organism growth and activity, and on their interactions
with other species.
14.3.1 Limiting Factors and Interactions
Resources are factors that are consumed by an organism, resulting in their being removed
from the environment. These include factors such as nutrients, but also less obvious
factors, such as habitats. For example, barnacles require rock surfaces to grow on. As
466 ECOLOGY: THE GLOBAL VIEW OF LIFE

rock surfaces become colonized, growth and recruitment of new individuals is reduced
by the crowding. Factors such as temperature are not resources because they are
not depleted.
Historically, the effect of resources on growth has been described by Liebig’s law of
the minimum, which states that a population will increase until a single resource, called
the limiting factor, becomes insufficient to support further growth. Thus, for every popu-
lation in an ecosystem under steady-state conditions, Liebig’s law predicts that each
would be limited by a single factor. If that factor were known, the population could be
increased by supplementing it.
Although it is still a useful concept and often applies to particular situations, Liebig’s
law is not gener ally true. For example, it may be possible to increase the growth of grasses
in a mea dow by supplementing either nitrogen, phosphorus, or water, and supplementing
two or three of these together could increase growth even more. One of the factors could
become limiting only if the others were present in excess.
Factors can also affect growth in a nonadditive way. For example, growth of Impatiens
parviflora was increased 33% by adding nitrogen fertilizer and 19% by adding phos-
phorus. However, when both were added, the increase was 100%. If there were no inter-
action, adding both nitrogen and phosphorus would be expected to increase growth
33% þ 19% ¼ 52%. This is an example of interaction, which is not accounted for in Lie-
big’s law. The concept of interaction is important: An interaction is when the sensitivity
of a variable to one factor depends on the level of another factor. Here, sensitivity can be
given a prec ise mathematical meaning. It is relat ed to the rate of change of one variable
with respect to another. One way to express the sensitivity of variable a to variable b
(s
ab
)is
s
ab
¼
da
db
’
a
b
ð14:10Þ
This expresses the sensitivity as the amount that a would change for a unit change in b.
Example 14.1 If increasing phosphorus concentration, P, from 10 mg/L to 20 mg/L
results in an increase in chlorophyll concentration, c, in a water body from 3 mg/L to
5 mg/L, the sensitivity of chlorophyll to phosphorus, s
cP
, can be approximated as follows:
By equation (14.10),
s
ab
¼
5 3
20 10
¼ 0:20 mg Chl-a=mgP
That is, each 1-mg/L increase in phosphorus produces a 0.20-mg/L increase in chlorophyll a.
Example 14.2 Peak summer chlorophyll contents in lakes, C
a
, have been empirically
related to phosphorus concentration at spring turnover, P
w
, as in equation (15.1):
C
a
¼ 0:37 P
0:91
w
(both concentrations in mg/L). According to this relation, what is the sen-
sitivity of chlorophyll to phosphorus at a concentration of 15 mg P/L?
Solution First, using equation (15.1), the expected concentration of chlorophyll a in
the lake is
C
a
¼ 0:37 ð15 mg=LÞ
0:91
¼ 4:35 mg=L
FACTORS THAT CONTROL POPULATIONS 467
