Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.


the degrading cells will hydrolyze to ammonia and be nitrified. This oxidative transforma-
tion releases 2 mol of hydrogen ions for every mole of oxidized nitrogen, which corre-
sponds to an alkalinity consumption rate of 7.14 mg alkalinity (as CaCO
3
) per milli gram
of fully oxidized ammonia-nitrogen. With incoming solids levels measured in percentile
figures, and with commensurate ammonia-nitrogen releases of 1000þ mg N/L, it concei-
vable that this sort of pH drop could well shift the reactor to a sufficiently low level (i.e.,
<pH 5.5) that this nitrification process would actually be discontinued.
Without this sort of pH disruption, though, the effluent nitrate-nitrogen levels com-
monly observed in aerobically digested biosolids residuals would be quite high (measured
in 100s if not even the 1000þ mg NO
3
-N/L range). The problem posed by these high
residual nitrate levels is that unlike ammonium -nitrogen, NO
3
-N has no cationic affinity
for soils. As a result, subsequent land application of these aerobically digested solids will
require close attention to the potential migration of these nitrates to the groundwater (for
which the U.S. EPA’s Safe Drinking Water Standard is 10 mg NO
3
-N/L).
Figure 16.47 Aerobic sludge digestion system.
648
BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CONTROL
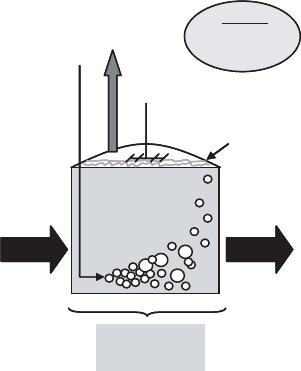
The regulatory stipulations on aerobic digester holding times (i.e., hydraulic retention
times) relative to temperature stem from the necessity to maintain suitable reductions in
pathogens. However, at the mesophilic conditions under which most aerobic digesters are
maintained, retention times on the order of multiweek to multimonth periods are not
uncommonly necessary to achieve volatile solids destruction levels up to or beyond the
38% range [i.e., the benchmark national standard for vector attraction reduction (VAR)
with stable biosolids established by 503 Rule federal regulations (i.e., Code of Federal
Regulations, 1993)]. Furthermore, during cold-weather periods, and with a corr esponding
drop in aerobic digestion efficacy, even longer periods will often be necessary.
As a proactive approach to escalating the standard aerobic digestion process, therefore,
this technology has followed a trend analogous to that of the newer anaerobic schemes
whereby reactor temperatures are increased to secure higher metabolic rates. This new
design strategy, commonly known as autothermal thermophilic aerobic digestion
(ATAD) (Figure 16.48) involves reactor temperatures starting in the mid-50
C range
and in most cases reaching the 60þ
C range, at which point these processes experience
far faster rates of lysis and oxidation. In addition, these ATAD operations also realize a
sizable acceleration in their rates of overall disinfection.
Relatively little is known as yet about the microbial character and makeup of these
types of systems, but it does appear that they may offer several new avenues for degrading
a numb er of organic, possibly even hazardous compounds in addition to that of conven-
tional wastewater sludge. In both cases, the levels of oxygen tension at which these high-
temperature system s appear to operate tends to be much lower than what is usually seen
with standard aerobic treatment reactors (e.g., 0.5 mg of dissolved oxygen per liter with
ATAD vs. 2þ mg/L in standard mesophilic aerobic digesters). In fact, at these DO levels,
it is rather likely that the microbial consortia involved includes both aerobic and quasi-
anaerobic microbes which live and work in metabolic harmony. Rather interestingly, the
issue of nitrate buildup and release is also a moot concern at these thermophilic levels.
Not only do the autotrophic bacteria responsible for nitrification effectively stop working
at temperatures of about 40
C, but there is also a pronounced tendency to volatilize free
12-14 d ay
HRT
(~55→ 75
o
C)
ATAD
Total HRT
~12-
Raw
sludge
Digested
sludge
Tank
cover
High
efficiency
aeration
system
Off-gas
scrubbing
(for ammonia,
Foam
cutter
12-14 day
HRT
Thermophilic
(~55→75
°
C)
ATAD
Total HRT
~12-14 days
H
2
S and mercaptans)
Figure 16.48 Autothermal thermophilic aerobic sludge digestion process schematic.
SLUDGE TREATMENT 649

ammonia (NH
3
) at this thermophilic level. Off-gas treatment (e.g., biofiltration) may,
however, be required to deal with this released ammonia, let alone the release of other
odorous compounds (e.g., reduced sulfur gases such as mercaptans and hydrogen sulfide).
Aerobic digestion is widely used as a sludge stabilization process at smaller waste-
water plants (i.e., at flow rates below about 20,000 m
3
/day). Batchwise opera ting regimes
are common, but it is also possible to use intermittent batch (with cyclic settle, decant, and
refill steps) and continuous-flow formats as well. Compared to anaerobic digestion, this
aerobic option tends to be easier to maintain, and in the case of mesophilic systems has
considerably less potential to release troublesome odors. These aerobically digested solids
are also biochemically stable (i.e., resistant to further decay) and low in residual ammo-
nia-nitrogen.
The key metabolic factor with aerobic digestion is that of the endogenous decay rate of
the solids involved, which for most cells is in the neighborhood of 0.05 day
1
(i.e., 5%
solids decay/day) at a temperature of 20
C. Of course, this rate of decay varies according
to temperature and the degradable characteristics of the processed solids.
The design criteria typically used for sizing these units is therefore that of the retention
time for these solids, for which values of 20þ days would accordingly provide volatile
solids reductions of approximately 63% (37% remaining) when completed under batch
conditions:
X
20
¼ X
0
e
ð0:05 day
1
Þð20 daysÞ
X
20
X
0
¼ 0:37
ð16:4Þ
where X
0
and X
20
are, respectively, the initial and final (after 20 days) volatile solids con-
centrations found within the reactor. However, despite this theoretical level of perfor-
mance, 503 Rule regulations require solids retention times (SRTs) of 40 days at 20
C
and 60 days at 15
C before the product of this mesophilic aerobic digestion process is
considered suitable for widespread use as a soil amendment based on desired VSS
destruction. Here again, the latter increase in SRT reflects the fact that metabolic solids
degradation slows down at colder temperatures.
Yet another important factor with aerobic digestion is that of its higher energy require-
ment, for both mixing and aeration, vs. that of anaerobic digesters, which not only require
far less energy for mixing but also generate an energy-rich methane gas product. Aeration
and mixing within an aerobic digester must therefore be provided using either mixers that
entrain oxygen mechanically or compressed air blowers that diffuse oxygen into these
tanks through bubble transfer. Mixing intensities with mechanical mixers are usually
designed to provide 10 to 100 W/m
3
(0.4 to 0.5 hp per 1000 gal), while the diffused aera-
tion systems are typically sized to provide 20 to 40 m
3
/min of air per 1000-m
3
tank
volume.
As mentioned previously, one variation to conventional aerobic digestion that has
recently drawn considerable attention due to its inherent ability to pasteurize sludge effec-
tively is that of the autothermal thermophilic aerobic digestion (ATAD) process, by which
the heat released exothermically from the digesting sludge naturally causes the reactor
temperature to rise into the thermophilic range (i.e., to values typically between 55 and
65
C, and sometimes well in the 70s). Federal regulations specifically stipulate a holding
time of only 24 hours for those systems able consistently to maintain a temperature at or
above 55
C.
650 BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CO NTROL
In comparison to standard mesophilic operations, these ATAD systems are able to take
advantage of a considerable increase in the available rate of endogenous decay, with
values believed to be many times greater than thos e experienced at mesophi lic tempera-
tures. A properly designed and operated ATAD system does not need external heating.
Heat is provided by the exothermic biodegradation reaction. A clear advantage of these
ATAD systems is that their solids loading rates are considerably higher than either the
mesophilic aerobic or anaerobic options, with values typically in the range 4.5 to 5 kg
TSS/m
3
day. In turn, the design and operating SRT values used with ATAD systems
can be decreased sizably, to values of a few weeks and possibly even less.
Example 16.6: Preliminary Autothermal Thermophilic Aerobic Digester Design Due
to the volume estimated for the standard anaerobic digester system in Example 16.5,
yet another request is then presented to the Deer Creek, Illinois, consulting engineer to
prepare a second preliminary sludge processing design for a more advanced, and
assumedly smaller, autothermal thermophilic aerobic digester. This ATAD design
makes use of all four assumptions regarding sludge production and character (that were
presented in Example 16.5).
Preliminary Design Details
Sludge solids mass
and volume,
reactor sizing,
and retention
time
Estimated daily total sludge solids mass: 151.43 kg TSS/day
Estimated raw wet sludge volume: 2.5 m
3
/day
Design anaerobic digester solids loading rate: 4.8 kg TSS/m
3
day
Design ATAD digester volume:
31:5m
3
¼ð151:4kg=dayÞ=ð4:8kg=m
3
dayÞ
Design anaerobic digester HRT:
12:6 days ¼ð31 :5m
3
Þ=ð2:5m
3
dayÞ
Related notes 1. ATAD loading rates are sizably higher than those of even the
high-rate anaerobic digester, and as a result these systems
will require a distinctly smaller volume (i.e., in this case, the
projected ATAD sizing is only one-third that of the anaerobic
digester).
2. ATAD solids loading rates are generally based on the total vs.
the volatile solids content of the sludge.
3. Prethickening of the incoming to sludge to values at or above
6% total suspended solids levels is very important with ATAD
systems in order to secure autothermal (i.e., self-heating)
operations.
4. Here again, this single-reactor design does not afford the
desired system redundancy associated with multiple units.
To generate the level of heat output required to incur this temperature increase (i.e.,
raising the reactor temperature autothermally), the incoming total solids content of the
raw sludge supply must be routinely prethickened (using thickeners, gravity belt thick-
eners, etc.) to values of 6% or highe r. The corresponding density of these solids, and
the fact that oxygen solubility drops considerably at higher temperatures, presents a
SLUDGE TREATMENT 651
distinct challenge in terms of providing the oxygen necessary to maintain aerobic
conditions without unacceptably stripping heat away from the reactor at a rate that
would negate the thermophilic condition desired. However, high-efficiency aerators
(e.g., jet-type mixing units) have proven to be a suitable aeration technology for this
type of application.
Despite the apparent technical benefits afforded by ATAD processi ng, a number of
important operating details have yet to be fully resolved. First, instrumentation for mea-
suring dissolved oxygen levels at these thermophilic operating temperatures has only
recently been developed and has limited field experience. Preliminary testing has there-
fore been conducted using measurement of oxidation–reduction potential (ORP) as an
alternative indication of the apparent aerobic nature of these reactors, and it does appear
that holding this parameter within an approximate range of about 50 to 350 mV can
subsequently be used to regulate aeration rates in a fashion that will obviate, or at least
minimize, undesired shifts toward fully anaerobic conditions (with ORP dropping much
below 400 mV), especially following intermittent loading events. Second, excessive
foaming events have been observed at a number of early full-scale plants. Here again,
this phenomenon is not fully understood, but it does appear that low-SRT opera tions
are particularly prone to this problem, perhaps due to load-related swings in the cyclic
level of soluble proteins. On the one hand, a limited amount of foam (i.e., 10 to 30 cm)
is actually beneficial, in that it helps to provide an insulating blanket across the top of the
tank. However, excessive foam production can lead to undesirable reactor overfoaming
conditions, to the point where foam-cutter (essentially a coarsely toothed disk rotating
just above the desired foam height) or foam-aspirator mechanisms are used to minimize
this condition. Finally, it is important to note that the microbial behavior, temperature
range, and environmental circumstance of ATAD processes is equivalent to that of bioso-
lids composting operations, or at least the interior zones of the actively composting piles,
as described in the following section.
16.2.3 Composting
Sludge residuals may also be biochemically digested, stabilized, and perhaps even parti-
ally or fully pasteurized using composting operations of the sort shown in Figure 16.49.
When exposed to moisture and appropriate environmental conditions, organic, nutrient-
rich surfaces will quickly become colonized by bacteria and other microorganisms.
Since no organism can be 100% efficient in its metabolism, during the ensui ng degrada-
tion of the organics, some chemical energy is wasted and given off as an exothermic heat
release. Ordinarily, this release of heat would not be noticed, since it quickly dissipates
into the environment. When solid-phase organic materials are held in a large pile, how-
ever, the pile itself acts as insulation and traps some of the heat. This effect can then lead
to a noticeable increase in temperature of the material and is thus referred to as biological
self-heating or autothermal metabolism. The intensity of self-heating can be surprising,
particularly if the pile is sufficiently porous to allow oxygen penetration and if the avail-
able moisture content remains sufficiently high to sustain continued biodegradation.
Temperatures of up to 80
C (176
F) can be reached with many materials, at which
point subsequent chem ical self-heating and eventual combustion may occur if moisture
is still present. In fact, important early research on this process was completed in New
Zealand during the 1960s, leading to documented reports of spontaneous ignition in
piles of wool! In much the same fashion, spontaneous ignition is a familiar phenomenon
652 BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CO NTROL

to farmers, who must, for example, carefully aerate and cool hay stored in barns to prevent
disastrous fires.
This sort of heat production is, in fact, a natural occurrence with any biochemical sys-
tem, and in our own case provides the thermochemical basis by which we intrinsically
maintain an optimal body temperat ure. Exothermic biochemical energy is also released
within aqueous-phase wastewater treatment processes, whether they be suspended-growth
activated sludge or attached-growth biofilm processes, but the high thermal mass of the
water and the cooling effect of aeration (by evaporation and conduction) typically offset
the metabolic heat release to such a degree that it does not affect system temperature. In
rather rare instances, high-strength industrial wastewater operations have experienced ele-
vated temperatures due to their elevated levels of heat release. Similarly, the ATAD sys-
tems described previously are also designed to benefit specifically from this mechanism,
in which aeration is maintained inside insulated reactors at rates close to stoichiometric
levels such that their exothermic heat release is not excessively offset by off-gas heat loss.
As applied to the high-temperature degradation of wastes bearing high-level biode-
gradable solids, composting is an engineered process that utilizes self-heating for
waste treatment purposes. By definition, it would be described as a mainly aerobic,
self-heating, solid-phase biological treatment process. The goals of composting include
stabilization, reduction, drying, and pathogen destruction. Traditionally, composting has
been used to treat agricultural wastes such as crop residues and animal manures, and
large-scale composting of separately collec ted yard wastes is a common practice in the
United States, particularly in the northeas t and midwest, where the fall leaf collection
can be sizable (often 10 to 20% of the total MSW for the year!). In the broader context
of environmental waste management, composting also has wide applicability for waste-
water treatment sludges, municipal solid waste (MSW) fractions, and some industrial
(including hazardous) wastes.
The primary objective of composting is to stabilize the waste material being treated.
This results in a reduction of mass and volume as well as stabilization, destruction of
putrescibles (rapidly degrading, odor-producing compounds). Another potential benefit
of composting is that in many cases the final residue is a loamy, soil-like material with
Figure 16.49 Windrow biosolids composting piles.
SLUDGE TREATMENT 653
an earthy smell, called compost, which may be used beneficially as a soil amendment or
surface mulch. The complementary fact that composting is capable of substantia l patho-
gen and weed seed destruction is also quite beneficial. As a soil amendment, compost
adds to the organic content of a soil, increasing its friability and its ability to adsorb
water and nutr ients. Uncomposted wastes are not as suitable for this purpose because
they will degrade in the soil, depleting soil oxygen, which can harm plant roots.
Waste materials subjected to com posting will typically undergo a succession of micro-
organisms in relation to progressive changes in waste character and environmental condi-
tions. At first, mesophilic organisms originally present in the material, along with early
invaders, will be dominant. This community may be highly diverse, including fungi, pro-
tozoans, and even invertebrate animals, such as earthworms, insects, and sow bugs, in
addition to bacteria. However, as the ongoing heat release moves temperatures above
40
C, many of these original inhabitants are inhibited, and eventually, most are killed
by the heat. At this point, having shifted into a thermophilic realm, small numbers of ther-
mophiles that were present find suitable conditions and grow rapidly. This is a more select
group, as few eukaryotes can survive at temperatures above 50
C. Above 62
C, the last
fungi are unable to grow, and only bacteria (and perhaps archaea) are left. The known
organisms in thermophilic composting include mainly Bacillus species, such as B. stear-
othermophilus and B. coagulans, and some actinomycetes, although there is evidence
from molecular techniques that other groups may also be present. Eventually, as substrate
is used up, the material will cool again, be recolonized by the germination of spores that
survived the high temperatures, and be reinvaded by mesophiles.
Earlier, some composting enthusiasts seemed to believe ‘‘the hotter, the better.’’ How-
ever, it is now well established that if not controlled properly, mos t composting materials
will overheat, killing or severely inhibiting even the thermophilic microorganisms. This
has led to the failure of many composting facilities, since the subsequent rate of degrada-
tion slows dramatically, and the remaining putrescibles lead to odor problems. Thus, an
important goal of modern composting technologies is to maintain temperatures at a desir-
able level of 60
C (140
F) to maintain the desired high rates of microbial activity. As a
point of comparison, most home hot-water heaters are set at 130
F; reac hing into the
interior of an actively compost pile would lead to a serious burn!
It is also important that the com posting material be kept mainly aerobic. Only aerobic
metabolism releases energy rapidly enough to sustain this degree of self-heating. Also,
avoiding extensive anaerobic conditions help s to minimize odor production. Usually,
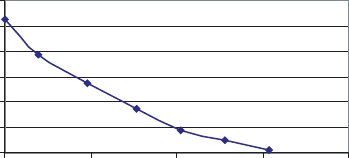
maintenance of at least a 10% oxygen partial pressure in the pore space s within a pile
(compared to the 21% oxygen in ambient air; Figure 16.50) will be adequate, depending
0
4
8
12
16
20
24
020406080
Pile Depth
(cm)
Oxygen Tension
(%)
Figure 16.50 Biosolids composting pile oxygen tension relative to pile depth.
654
BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CONTROL

on the material and method used. To facilitate the desired transport of oxygen into these
actively composting systems, therefore, the composting solids must be maintained in a
suitably porous form. If the starting waste materials are too wet, as is the case with
most raw sludges, they may need to be partially dewatered and may also need to be
mixed with other materials to improve porosity (bulking agents, such as finished com-
post, MSW, or wood chips). If the pile is too wet, the water filling the pore spaces severely
limits oxygenation, and anaerobic conditions develop. On the other hand, materials such
as municipally collected leaves commonly start out too dry for rapid microbial growth and
thus benefit from water addition. Generally, moisture contents in the range 50 to 70% will
give the best results, but this depends on both the material and the type of composting
system used. Many of the bulking agents used in composting (e.g., wood chips, shredded
tire chips, bark) have an original bulk cost that warrants an attempt to secure their recov-
ery and reuse. As shown in Figure 16.51, it may be possible to screen and recover these
Figure 16.51 Sludge composting: (a) aerated piles; (b) bulking agent screening and recovery.
SLUDGE TREATMENT 655
supplemental bulking agents from the finished, composted product so that they might
then be reused yet again wi th fresh sludge. This effort not only saves money by reducing
the necessary volume of bulking material but also provides an initial seeding of the
coblended material with thermophilic microorganisms, which then promotes faster startup
times.
The relative masses of carbon and nitrogen (the C/N ratio) can be important in com-
posting, with a ratio of about 30 : 1 often considered desirable. Materials such as dry
leaves may have C/N ratios of 80 : 1; under such conditions, nitrogen becomes limiting
and composting rates are slowed. This is normally acceptable for leaf composting, and N
addition is not recommended, but this might be an issue for some industrial waste s. Muni-
cipal sewage sludge, thoug h, tends to have a C/N ratio of 8 : 1, indicating excess nitro-
gen. This will not decrease composting rates, but can lead to the release of nitrogen as
either ammonia gas (making odors more of a problem) or as a potential water pollutant.
Addition of a carbonaceous material may therefore be desirable. However, although a
bulking agent such as wood chips will increase the calculated C/N ratio, most of the C
is unavailable to microorganisms and hence may not produce the full beneficial effect
expected.
As with other microbially based treatment systems, the presence of a large number and
variety of microorganisms is desirable for rapid composting. However, waste materials
typically already contain a high concentration and diversity of appropriate organisms,
and under proper conditions their growth will be very rapid. In the few cases where the
deliberate addition of microorganisms may be warranted, such as for some pasteurized
food-processing residues, this is usually best accomplished by adding small amounts of
finished compost or soil to the initial mixture. There is no scientific evidence that the addi-
tion of commercially available inocula or ‘‘compost starters’’ is beneficial.
On the other hand, waste materials may start out with numerous undesirable biological
agents present, such as pathogens, parasites, and weed seeds. Composting can be extre-
mely effective (better even than chemical disinfection, and probably second only to incin-
eration among treatment processes) at inactivation of these undesirables. This is a result
mainly of the high temperatures achieved (e.g., Salmonella will be reduced significantly
within an hour or so at temperatures much above 60
C), but is also aided by the vigorous
microbial activity that occurs. Thus, decreases of well above 99.99% are expected in
properly run systems. In fact, a common criterion, maintenance of 55
C for 3 days, is
predicted to give a minimum of 15 ‘‘9’s’’ (i.e., 99.9999999999999%) reduction of even
the most resistant pathogens. Federal (i.e., 503 Rule) regulations in the United States for
pathogen control with composting systems stipulate specifically that the temperature of
these piles must be held above 40
C or higher for a period of 5 days. This standard
also requires that a temperature of 55
C or higher must be reached for a period of at
least 4 hours in such piles to maintai n the necessary reduction in pathogens.
One special concern with pathogens is that a few may actually grow during some com-
posting processes. The best known example is Aspergillus fumigatus. This thermotolerant
fungus is cellul olytic (degrades cellulose) and thus very common in nature and agriculture
in soil and decaying vegetative material. However, it produces large numbers of spores
that can cause a mild to severe allergic reaction in susceptible people. In a few cases,
it is also able to opportunistically invade people with severely weakened immune systems,
leading to potentially lethal infections. Sludge composting operations using wood chips as
a bulking agent may release very high levels of A. fumigatus spores during the final
screening step to remove the wood chips from the compost. Reuse of the wood chips
656 BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CO NTROL
then serves to heavily reinoculate the new pile with A. fumigatus. Elevated levels have
also been observed at some leaf composting sites during the turning of windrows.
The overall composting reaction can be described using a modified form of the basic
equation for aerobic respiration:
organic matter þ O
2
! CO
2
þ H
2
O þ compost þ heat ð16:5Þ
From this expression it can be seen that the rate of organic matter stabilization is propor-
tional to the rate of heat production. Thus, maximizing the rate of heat produc tion will
maximize the rate of stabilization. However, if the material becomes too hot, rates slow
dramatically. Once active self-heating occurs, therefore, it is necessary to remove heat at
approximately the same rate it is released, so as to avoid exceeding 60
C in the material.
This should be a major concern in system design, as the amount of heat requiring removal
can be substantial. The amount of heat released per mass of oxygen consumed is approxi-
mately 14,000 J/g and is very nearly constant for a wide variety of different organic mate-
rials. At the same time, it would be necessary to provide adequate oxygen to reach this
oxidative heat release, and to keep moisture and other parameters within a desirable range.
The goal of a composting system is to stabilize the particular material being treated in
an efficient, economical, and environmentally sound manner. For some materials, such as
leaves, a low-cost system can be used, even though the conditions it provides do not come
close to maximizing composting rates. This is because the facility, if it is large and iso-
lated enough, can simply allow extra time for completion (e.g., 6 to 18 months), and the
materials can be managed so as not to cause problems during this time . Other materials,
such as sludges, however, usually demand closer control and require composting systems
that much more nearly achieve maximum rates. Otherwise, problems are likely to occur,
and costs may soar.
Although there are many variations, there are really three approaches to large-scale
composting: mixing; forced aeration; and both. Any of the three potentially can be
done out in the open, under a roof, or in an enclosed reactor, although some combinations
are more common and/or logical. Interestingly, almost all systems are operated as batch
processes rather than continuous feed, as is the case with most other waste treatment
systems.
Probably the most common type of composting, such as that shown in Figure 16.49, is
that of windrowing. The sludge-plus-bulking agent windrow piles are constructed in an
elongated, haystack shape (in cross section) up to perhaps 1.3 to 2 m (4 to 6 ft) high
and 4 to 5 m (12 to 15 ft) wide, and lengths reaching up to and beyond 100 m
(300 ft). Periodically (e.g., twice a week initially, monthly later) it is mixed, or turned,
using a front-end loader or specialized turning machine (as can be seen in the background
of Figure 16.49). Windrowing is virtually the only method used for yard waste compost-
ing and is also used occasionally for sludges and solid was tes in areas that are sufficiently
isolated, handling small volumes, or with other special circumstances. Also, it is com-
monly used in a curing stage, a low-rate finishing step after a more active composting
phase. In some cases, the material is enclosed in bins, and mixed there: essentially, ‘‘wind-
rowing’’ within an isolated reactor.
One disadvantage of static piles and other unmixed systems is that stratification occurs
with gradients of temperature and moisture within the pile. Thus, some portions of the
material may not heat sufficiently for pathogen kill, whereas others may dry or overheat
and become inactive. Periodic windrow mixing or turning, therefor e, helps to ensur e
SLUDGE TREATMENT 657
