Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

uniform solids breakdown. Although some heat is lost during turning, and some oxygen
is incorporated in the pile, the increase in microbial activity spurred by the mixing quickly
(within hours) reheats the material and depletes this added oxygen. The height and width
of these windrows must therefore be held to values of less than a few meters, such that
oxygen may adequately diffuse into the pile interior from the surface. In fact, the ongoing
release of heat from windrow piles helps to facilitate this aeration process, whereby the
physical air current induced by heat rising upward and out of these hot piles essentially
drafts or pulls in cooler air, and oxygen, at the pile base.
In lieu of intermittently mixed windrow systems, there are also units designed to pro-
vide nearly continuous mixing and/or aeration for com posting wastes. Forced static pile
aeration, as shown in Figure 16.51, represents yet another method developed by the Agri-
cultural Research Service (U.S. Department of Agriculture) at Beltsville, Maryland. An
aeration system (ducts, or a perforated false floor) in the lower portion of, or under, the
pile is used, and control systems can be provided to either increase aeration (if the pile
starts to get too hot) or decrease aeration (if the pile starts to get too cool), as needed. In
some cases (including that of the piles seen in Figure 16.51) , this supplemental airflow is
drawn into and through the pile by applying a vacuum to the duct work, with the exhaust
air then being routed through a secondary smaller pile of aged compost material in order
to screen out potentially problematic gas-phase odors, fungal spores, and so on. However,
forced blower aeration directly into the pile interior has also proven to be effective, par-
ticularly in terms of securing direct oxygen entry to the pile’s hottest and most active
zone. These types of simple, feedback aeration schemes, based on a thermostatically regu-
lated vacuum or blower, have a number of interesting features. First, the pile itself
demands the amount of aeration it needs to remove enough heat to keep temperatures
in the desirable range. This is important because the rate of activity (heat generation)
changes with time as the readily degradable organics are depleted. Second, most of the
cooling results from evaporation of water within the pile into the supplied air, at which
point the pile starts to dry out. With some materials pile drying may be so extensive that
composting rates decrease and supplemental water will need to be added. Often, however,
the drying is beneficial, as it substant ially reduces the remaining mass and volume and
leads to a more stable final product that is more easily stored, transported, and ultimately
used with gardening, land application, and so on, measures.
A third feature stems from the fact that the amount of oxygen required for biodegrada-
tion of the organic material is closely related to the amount of heat released [as can be
seen from equation (16.5)]. Thus, an air function ratio can be defined for forced aeration
systems as the amount of air required to remove the heat produced compared to the stoi-
chiometric amount required to provide the oxygen necessary for the oxidation reaction
that releases it. This ratio will vary slightly based on materials, ambient conditions, and
pile temperature, but is typically about 8.5 to 9.0. This ensures that sufficient—in fact,
considerably excess—oxygen will automatically be provided by the aeration required
for cooling. It is interesting to note that the air function ratio must in fact be greater
than 1.0 for self-heating to occur. As a practical matter, some aeration, typically provided
by a timer, also is needed to supply oxygen before and after the phase of the process d ur-
ing which aeration for cooling is required: the come-up and cool-down stages.
The most advanced composting systems combine forc ed aeration for temperature con-
trol and oxygenation with mixing to increase rates and uniformity. Such systems include
agitated beds and ‘‘mushroom tunnels’’ (so-called because they were developed in
the mushroom industry to provide the high-quality compost needed for the commercial
658 BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CO NTROL

growing of mushrooms). Tunnels are enclosed, with a small headspace above the material,
and a portion of the used air can be reused for aeration. This requires some cooling of the
air and condensation of the water vapor present but can greatly reduce the amount of fresh
air needed (since based on the air function ratio, little of the oxygen is utilized). This in
turn reduces the amount of spent gas that must be vented, making control of odors and
other volatile compounds simpler.
Example 16.7: Composting Air Function Ratio Sample Calculation Based on equation
(16.5) and the typical energy release of 14,000 J/g O
2
consumed, an air function ratio can
be calculated for specific composting conditions. Assume, for example, that the ambien t
air is at 20
C and 50% relative humidity (RH), and the air exiting the composting pile is
at 60
C and 100% RH. From thermodynamic data (found in psychrometric charts) it can
be determined that this represents a change in enthalpy (heat energy) of 362 J/g dry air
(part from the increase in temperature, and part from the increase in the amount of water
vapor). Thus, the amount of dry air required to remove the heat released from 1 g of O
2
consumption is
14;000 J=gO
2
362 J=g dry air
¼ 38:7 g dry air=gO
2
consumed
The amount of dry air required to provide 1 g of O
2
is only 4.31 g. Thus, the ratio is
air function ratio ¼
air required for cooling
air required for providing oxygen
¼
38:7g=g
4:31 g=g
¼ 8:98
The air function ratio will vary slightly depending mainly on the temperature and RH
of the inlet and outlet air. However, it is usually between 8.5 and 9.0 for conditions that
are likely to be encountered during thermophilic composting.
A number of other composting system designs have also been of initial interest because
of their materials handling approaches, including semicontinuous feed systems such as
silos (in which materials were added to the top and finished material was removed
from the bottom) and rams (in which material was pushed along in a ‘‘tunnel’’). However,
these particular approaches greatly compacted the material, destroying the porosity that
was essential to movement of air through it. It is important to keep in mind that although
efficient materials handling approaches are important for cost-effectiveness, the system
will fail if the basic biological requirements of the composting process are not sufficiently
met.
16.3 POTABLE WATER TREATMENT
There are relatively few engineered applications for biological treatment within potable
water systems. The rare exception to this is that of denitrifying processes intended to
reduce excessive nitrate levels (i.e., above the regulated level of 10 mg/L), as might be
used in association with shallow groundwaters affected by regional far ming and fertiliza-
tion activities. In contrast to beneficial instances of biological treatment with potable
waters, though, biology can, and in many instances often does, become a troublesome
POTABLE WATER TREATMENT 659
issue either in terms of affecting raw water quality or during subse quent dissemination of
these waters via distribution piping.
Excessive biological growth of any sort, whether algal, bacterial, or other, can lead to
elevated levels of turbidity (i.e., cloudiness) that would complicate subsequent treatment
efforts. However, one of the largest biological problems in potable processing of surface
waters is that of the formation of various taste- and odor-inducing chemicals. Two such
compounds, geosmin and methylisoborneol (often referred to as MIB), are depicted in le
16.14, and it is these particular biochemical products that are most frequently cited as the
chemical culprits behind serious aesthetic complaints. These compounds are produced
biochemically by a variety of cyanobacteria (including Oscillatoria and Anabaena), acti-
nomycetes (including Streptomyces, Micromonospora, and Nocardia), and algae (e.g.,
Asterionella), leading to taste and odor complaints that have been likened to ‘‘musty’’
or ‘‘fishy’’ (Darleym, 1982).
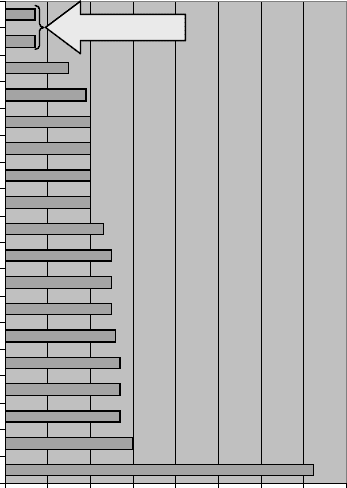
The ability of either compound to create problems is readily demonstrated by their
respective aqueous-phase ‘‘threshold odor’’ values, which quantifies the concentration
value at which humans may perceive their presence. As shown in Figure 16.52, geosmin
and methyl isoborneol have remarkably low ‘‘threshold’’ values, in the single-digit parts
per trillion range. This level is nearly 1000 times lower than that of the next-highest com-
pound (i.e., methyl sulfide, otherwise known as methyl mercaptan, which as described
earlier, has a considerable odor of its own!).
The typical circumstance of these two products creating a problem is that of seasonal
blooms of cyanobacteria and algae in reservoirs, lakes, and rivers, tied to changes in water
temperature and nutrient availability. One such commonplace trigger is that of diurnal fall
and spring overturns in deeper reservoirs and lakes, at which point the underlying phos-
phate-rich hypolimnion waters are returned to the surface to stimulate phototrophic
microbial growth. Even without the bacterial release of geosmin or MIB, algal activity
stimulated in much the same fashion may also result in the release of various phenolic-
type organics, which can then react with chlorine during the treatment process to produce
the strong taste- and odor-causing chlorinated phenols.
A second biological problem in potable water systems that may occur is that of bac-
terial colonization along the interior surfaces of distribution piping (Bitton and Gerba,
TABLE 16.14 Taste- and Odor-Producing Chemicals Generated by Bacteria and Algae
Chemical Name Chemical Structure Generating Microorganisms
Methylisoborneol (MIB)
OH
Actinomycetes
Oscillatoria
Geosmin
OH
Actinomycetes
Anabaena
Oscillatoria
660
BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CONTROL
1984). These microbes, such as Aeromonas, use low-level substrates in the form of bio-
degradable and assimilable organic carbon that in many instances are produced by the
oxidative conversion of otherwise recalcitrant materials (e.g., naturally occurring humic
acids) by chlorine. More often than not, these growths do not reach population densities
that would unduly affect the healthful quality of the water. Their presence can nonetheless
lead to potential corrosion problems, particularly with cast iron piping. Isolated patches of
bacterial biofilm can accordingly create microsite niches whose isolated underlying strata
may effect a reducing environment that, in time, corrosively degrades the pipe wall.
Neither geosmin nor MIB can be removed easily during the treatme nt of surface
waters, at least using the conventional technology (e.g., coagulation, filtration, disinfec-
tion) employed at most municipal operations. As a result, the only approach to dealing
with this problem is that of attempting to eliminate the microbial source of the problem,
either by switching water sources or by working to eliminate the biological agent respon-
sible. Reservoirs are often treated with copper salts, which inhibit algae growth at levels
of 0.5 to 1.0 mg Cu/L.
The key to preventing growth of the organisms in the first place is restricting or elim-
inating the presence of nutrients, such as phosphate, which contribute to the offending
phototrophic bloom. Within many reservoir and lake settings, these nutrients are intro-
duced through a variety of nonpoint sources that are not particularly easy to control or
eliminate, including fertilizer use on lawns bordering homes and cottages, septic tank lea-
chates, or agricultural inputs received via upstream drainage . However, should the domi-
nant phosphorus source be that of diurnal overturn and exchange, this mechanism may be
0.001 0.01 0.1 1 10 100 1000 10000 10000
0
Ammonia
Skatole
Hydrogen sulfide
Methyl mercaptan
Propyl mercaptan
Trimethylamine
Amyl mercaptan
Ethyl mercaptan
Phenyl mercaptan
Benzyl mercaptan
Allyl mercaptan
Diphenyl sulfide
Indole
Thiocresol
Tert-butyl mercaptan
Crotyl mercaptan
Methylisoborneol (MIB)
Geosmin
Threshold Odor (µg/L)
5 parts per trillion
5 parts per trillion
Figure 16.52 Threshold odor levels for various aqueous-phase chemicals.
POTABLE WATER TREATMENT 661
controlled using low-level aeration of the hypolimnion region in a fashion that effectively
ties up the available phosphate ions through precipitation with iron or calcium. This
strategy can be relatively simple to employ, using flexible plastic piping, laid across the
deepest reach of a lake, that is drilled with holes and supplied with compressed air such
that it releases a steady current of bubbles. Another approach is somewhat more compli-
cated, using skirted mechanical mixers or turbines that create hydraulic drafts to routinely
mix the upper and lower reaches of the lake, but in either case the end result is that of
inducing a degree of supplemental aeration that reduces or eliminates the presence of
soluble phosphates.
16.4 WATER AND WASTEWATER DISINFECTION TREATMENT
One of the most significant public health advances over the past century was that of devel-
oping, and then routinely applying, suitable engineering methods for disinfecting potable
waters that could retard, and ideally obviate, the transmission of waterborne disease.
Rudimental disinfection measures based on water filtration (used by the ancient
Egyptians) and heat treatment have long been practiced, but the advent of commercially
available chlorine during the late nineteenth and early twentieth centuries effectively
revolutionized the wide-scale utility and efficacy of this practice (Baker, 1948).
Indeed, chlorination has subsequently served as the dominant disinf ectant ‘‘tool’’ for
potable waters since the early twentieth century, but there are now at least six strategies by
which waters, wastewaters, or sludges might be duly processed to achieve a desired reduc-
tion in their microbial content (Bryant et al., 1992; U.S. EPA, 1999):
1. Physical filtration (e.g., using ultrafiltration or reverse osmosis)
2. Heat treatme nt (e.g., boiling and pasteurization)
3. Physical sonication
4. Strong oxidant chemical treatment (e.g., using halogens such as chlorine, bromine
or iodine, or ozone)
5. Nonoxidizing chemical treatment
6. Radiation treatment
These disinfection strategies can largely be subdivided into the following four options,
although in many instances there are likely to be significant overlaps with the causative
impacts imposed by any one disinf ection procedure:
1. Remove cells physically
2. Alter and disrupt cell membrane permeability
3. Alter and disrupt metabolically essential proteins and enzymes
4. Alter and disrupt genetic ally essential nucleic material
The first such approach to using physical filtration to remove microbes depends on cell
size, which is rather fortunate since chemical-based disinfection tends to become proble-
matic with large cells or cysts, such as might be experienced with protozoan forms of
Giardia lamblia and Cryptosporidium. Conventional slow- and rapid-sand filters have
been used for more than a century to effectively reduce, if not completely eliminates,
662 BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CO NTROL
these larger microbes, and as a result these types of filters have been widely used for pro-
cessing surface water sources susceptible to microbial contamination. However, the suc-
cess of these operations can be compromised by human and technical shortcomings (e.g.,
inadequate filter backwashing regimes), as has been demonstrated in several U.S. cities
over the past few decades. One such well-publicized incident with mismanaged water fil-
ter operations in Milwaukee, Wisconsin, during 1994 affected 400,000þ residen ts. The
effectiveness of these operations has been improved with tightened stipulations (i.e.,
the U.S. EPA Interim Enhanced Surface Water Treatment Rule) on routine filter monitor-
ing (U.S. EPA, 1998). In recent years, the disinfection effectiveness of media-based filtra-
tion has been eclipsed by the use of micro- and ultrafiltration systems with pore sizes in
the double-digit nanometer range (e.g., typically 30 to 100 nm), which are small enough
to prevent the passage of any pathogens.
As for those disinfection strat egies intended to negatively change the permea bility or
perhaps water content of cells, numerous examples can be seen with foods prepared in
percentile-level salt, sugar, or organic-acid-rich conditions (e.g., pickles, candied fruits,
cheeses, vinegar, tomato catsup). These preservation environments, many of which
yield osm otic pres sures intole rable to active cell growth, facilitate a bacteriostatic condi-
tion in which microbial growth has been effectively stopped without specifically killing
the original cells. Large-scale adjustments to the osmotic pressure within water, waste-
water, or sludge treatment processes would, of course, be infeasible given the necessary
chemical dosage requirement (i.e., at expensive, high percentage levels).
Chemical disinfection agents that alter the form and function of membrane-bound
transport enzymes could disrupt the transmembrane passage of essential substrates or
nutrients. Whether or not the latter membrane-specific impact is realized in conjunction
with the use of the more widely used antimicrobial chemicals (e.g., chlorine, ozone),
enzyme disruption and denaturation are widely considered to be their dominant disinfec-
tion mechanism. These agents, and particularly those involving either strong-oxidant (e.g.,
chlorine) or superoxide (generated by means of irradiation) chemicals, readily disrupt a
cell’s hydrogen- and covalently bonded three-dimensional enzyme conformation. Having
lost the enzyme’s catal ytic contribution to energy-yielding catabolism, these disinfected
cells subsequently lack sufficient energetic resources to reproduce effectively.
Four different strong oxidants are widely used for disinfection: (1) halogens (chlorine,
bromine, and iodine), (2) halogen-containing compounds (e.g., chlorine dioxide, chlora-
mines, bromochlorodimethylhydantoin), (3) ozone, and (4) hydrogen peroxide (H
2
O
2
). In
each case, the standard engineering application is that of dosing the applied chemical into
a short-term contact chamber (i.e., typical ly designed for 15 minutes’ retention) using a
chemical delivery system preset to achieve a desired disinfectant concentration relative to
measured flow. As shown in Figure 16.53, these disinfection contact chambers are often
designed with a serpentine configuration in an attempt to secure a quasi-plug-flow regime.
Chlorine has been, and remains, the dominant disinfectant chemical with waters and
wastewaters in the United States, applied either in gas (Cl
2
), liquid (NaOCl), or solid
[Ca(OCl)
2
] form at what is likely to be the least possible cost (i.e., in the range of pennies
per pound) for any disinfection option (White, 1992). Aside from cost, chlorine’s advan-
tages include its range of delivery options and expected efficiency. However, there are also
shortcomings with its use, including the fact that there are significant safety issues to be
addressed when storing and metering chlorine gas.
One key aspect of chlorine use is that of its sensitivity to pH. Above pH 7.5 the desired
hypochlorous acid (HOCl) species found in aqueous environments (e.g., produced by the
WATER AND WASTEWATER DISINFECTION TREATMENT 663

hydration of chlorine: Cl
2
þ H
2
O ! HOCl þ H
þ
) will disassociate into a hypochlorite
(OCl
) anion (HOCl ! OCl
þ H
þ
) whose antimicrobial efficiency is far lower than
that of the hypochlorous acid (i.e., HOCl) form.
Another important fact is that hypochlorous acid reacts read ily wi th reduced ammonia
(NH
3
), leading to a series of amination reactions and products [i. e., monochloramine,
NH
2
OCl; dichloramine, NH(OCl)
2
; and nitrogen trichloride, NCl
3
] whose bacteriocidal
efficacy is again less that of the original HOCl.
Bromine use as a disinfectant also has its own set of unit features in terms of chemistry,
benefits, and shortcomings (Water Environment Federation, 1996). First, the operative
hypobromous acid (HOBr) species does not disassociate until it reaches a pH of about
8.5, so offering a wider range of serviceability them chlorine. Second, bromine tends to
have a higher level of efficacy at equivalent concentrations, so lower dosage levels might
be used. Third, the bromamine forms are all far better disinfectants than are the chlora-
mines. Indeed, a solid-phase, bromine-bearing compound called bromochlorodimethylhy-
dantoin (BCDMH) is widely marketed for hot tub and spa applications, given the
particular prevalence of urinary ammonia release. One clear disadvantage, though, is
that of cost, which tends to be several times higher than that of chlorine if the latter’s
efficacy attributes are ignored.
Hydrogen peroxide has little, if any, credible role as a disinfectant in environmental
engineering systems, but ozone has found widespread acceptance, particularly in Europe,
as a potable water disinfectant . As compared to any of the halogen-based options, ozone
has the unique ability to dissipate shortly after its addition, without any semblance of a
lingering residual. This lack of a residual is considered a disadvantage in the United
States, where residual chlorine levels are maintained routinely in potable water delivery
systems as a safeguard against subsequent contamination that can occur within the distri-
bution system. However, the conventional wisdom in Europe is that disinfecting chemical
Figure 16.53 Serpentine chlorination reactor for wastewater effluent disinfection.
664
BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CONTROL
residuals are both unwarranted and undesired. Yet another important aspect of ozone use
is that it must be produced on-site, using electrical hardware that is not all that simple and
at a cost that is considerably higher than that of chlorine.
A wide range of nonoxidizing organic and inorganic chemic als are used for, or are able
to provide, disinfecting effects, including aldehydes (formaldehyde and glutaraldehyde),
phenolics, alchohols (ethanol and isoproponal), cationic detergents, nitrites, and heavy
metals (e.g., mercury, silver nitrate, tin, arsenic, copper). Although most of these chem i-
cals have little relevance for the disinfection of waters, wastewaters, or sludges, there are
two noteworthy exceptions. Specifically, silver-impregnated filters are sometimes mar-
keted for point-of-use water conditioning devices, such as those that are sometimes
screwed onto the outlets of sink faucets. In this instance, the silver is intended to be slowly
leached from the filter medium (typically, activated carbon) at a rate that, hopefully, will
retard the opportunistic formation of microbial biofilms intent on using sorbed organics as
their energy source. A second nonoxidizing chemical disinfectant option is that of using
cationic detergents in the form of quaternary ammonium com pounds (formed as organic
salts of ammonium chloride and commonly referred to as quats). One such common
application is that of controlling biofilm growth on cooling tower surfaces, in which
aggressive (i.e., oxidative) disinfection agents such as chlorine, bromine, and ozone
would unacceptably attack exposed wood or metal heat-transfer surface s.
One of the unique disinfection features of the bactericidal quaternary compounds is
that they have a distinctly higher level of effectiveness with many gram-positive bacteria,
probably due to the added depth and complexity of their membr ane structure. Conversely,
gram-negative cells as a whole are often similarly considered to be somewhat more resis-
tant, perhaps due to the added depth and complexity of their membrane structure. Indeed,
Pseudomonas probably tops the list in terms of durability, under conditions that would foil
the vast majority of other cells (e.g., growth in distilled water). Similarly, gram-positive
Mycobacteria species, as well as spore formers, also tend to exhibit this resistant nature
when challenged with quaternary disinfectants, apparently based on the prot ective capa-
city of their respective outer cell coatings. Finally, given their chemical nature, these quat
compounds also bear a unique sensitivity to inactivation when exposed to complexing
soaps, detergents, and organic materials.
The fourth and final disinfection mechanism is that of alter ing and disrupting a cell’s
genetic makeup so that the cell is prevented from reproducing even though it may still
have the energy to do so. This disinfection effect is largely associated with the use of
ultraviolet irradiation, and its consequent high-energy cross-linking of adjacent nitrogen
base groups poised side by side at various points within stranded DNA (see the thymine
dimer reaction in Figure 16.54). The resulting impact of polymerizing DNA and forma-
tion of thymine dimers follow much the same path, such that cells are effectively steri-
lized by this UV exposure.
The range of wavelengths associated with ultraviolet irradiation actual ly has consider-
able breadth, from 4 to 300 nm, but the highest level of absorbance by DNA appears to
fall nearly coincident with the maximal output value (i.e., at approximately 254 nm) for
the emission spectrum of light emitted by mercury-vapor light bulbs. The standard engi-
neering application of UV irradi ation involves an array of mercury bulbs placed inside an
irradiation chamber, through which the water or wastewater is passed, with each such
tube being jacketed inside a UV-transparent quartz jacket. These tubes are aligned verti-
cally or horizontally in a fashion where the hydromechanics of the operation provides
maximal opportunity for exposure of cells flowing through the chamber while obviating
WATER AND WASTEWATER DISINFECTION TREATMENT 665
short-circuiting pathways that would degrade process efficiency. Figure 16.55 depicts
one such array used for disinfection of a wastewater effluent immediately prior to dis-
charge. UV irradiation with mercury bulbs offers an effective m eans of sterilization
for those engineering applications involving fairly clear or optically transparent
waters and wastewaters. Conversely, sludge is not amenable to UV disinfection
given the unacceptably shallow degree of light penetration into this material.
N
N
Thymine dimer
NH
O
H
CH
3
O
NH
O
H
CH
3
O
PHOSPHATE
PHOSPHATE
SUGAR
SUGAR
PHOSPHATE
Figure 16.54 Thymine dimer formation along DNA strand produced by polymerizing ionizing or
UV irradiation.
Figure 16.55 Ultraviolet irradiation reactor for wastewater effluent disinfection.
666
BIOLOGICAL APPLICATIONS FOR ENVIRONMENTAL CONTROL

Ionizing radiation using x-ray and gamma-ray beams with even higher energy levels
than that of UV irradiation, can also be used in water, wastewater, and sludge disinfection.
These ionizing mechanisms displace electrons during beam bombardment (i.e., at which
point they are said to ionize), and in the presence of oxygen these displaced electrons elec-
trochemically form a type of free radical (called hydroxyl radicals) that is highly toxic to
microbial cells. Free radicals are highly reactive, having essentially no activation energy
for their reaction. Given the acutely reactive nature of these radicals, they readily attack
and destroy hydrogen bo nds, double bonds, and ring structures essential to the metabolic
utility of various cellular molecules. Yet another operative mechanism, and perhaps the
key factor behind disinfection with ionizing irradiation, is that of a polymerizing impact
(e.g., DNA thymine dimerization) whereby the biochemical effectiveness of complex
molecules is degraded or termin ated. This technology bears a degree of complexity and
technical hazard that is not typically appropriate for most municipal applications, how-
ever, so that only a limited number of sites, the majority of which involve sludge disin-
fection presently rely on its use. On the other hand, ionizing radiation is used widely for
the disinfection of pharmaceuticals and of disposable dental and medical supplies (e.g.,
syringes, gloves).
In reviewing the various options for disinfection, the mere fact that there is such a
diverse range of disinfecting mechanisms and associated effects demonstrates that there
is no perfect solution for all engineering applications. Excluding heat treatment and osmo-
tic pressure, which have little if any pragmatic utility for large-scale engineered disinfec-
tion, the remaining options exhibit unique differences in their associated costs, technical
complexity, residual impacts, environmental sensitivity, and relative performance, such
that an appropriate assessment of many such site-specific issues have to be completed
prior to making a final decision.
As for defining the precise goal of disinfection, the normal benchmark for potable
water is that of zero residual fecal coliform presence. In the case of wastewater disinfec-
tion, though, effluent standards tend to be targeted for a somewhat higher level of residual
bacterial presence in a fashion that implicitly reflects the latter phenomenon of microbial
resistance. Compared against the typical levels of bacterial presence within raw, undisin-
fected was tewaters (such as those shown in le 16.15), therefore, effluent criteria for fecal
coliform usually tend to be somewhat more tolerant (i.e., typically at 200 fecal
coliform colony forming units per 100 mL). However, compliance with this sort of
effluent fecal coliform standard would still require a sizable four- to five-log reduction
(i.e., based on influent fecal coliform densities in the range 10
6
to 10
7
cells per 100 mL).
Extending beyond the theoretical aspects of these disinfection mechanisms there are
pragmatic uncertainties and inherent regulatory concerns tied to the nature and degree
TABLE 16.15 Bacterial Levels Present in Representative Wastewaters
Observed Bacterial Densities (10
6
viable cells/liter)
———————————————————————————————————————
Wastewater Total Coliform Fecal Coliform Fecal Streptococci FC/FS Ratio
A 172 172 40 4.3
B 330 109 24.7 4.4
C 19.4 3.4 0.64 5.3
D 63 17.2 2 8.6
Source: Adapted from Water Environment Federation (1996); Crites and Tchobanoglous (1998).
WATER AND WASTEWATER DISINFECTION TREATMENT
667
